Xudong Guo, Guoqiang Yang, Yi Li. Development of Extreme Ultraviolet Photoresists[J]. Laser & Optoelectronics Progress, 2022, 59(9): 0922004
Search by keywords or author
- Laser & Optoelectronics Progress
- Vol. 59, Issue 9, 0922004 (2022)
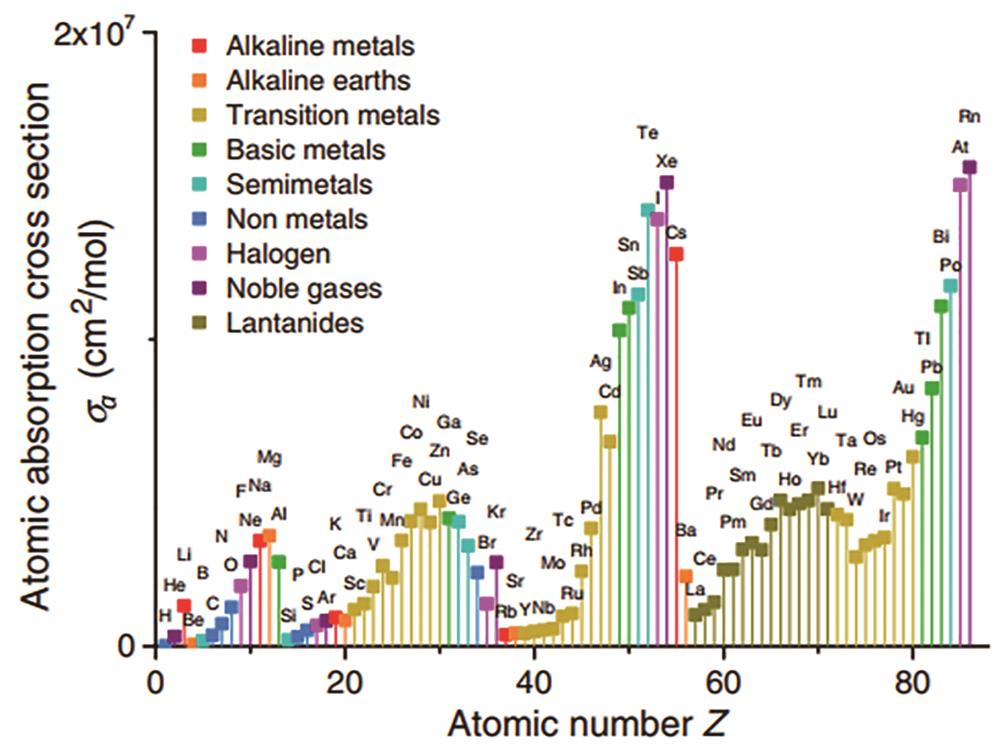
![Atomic absorption cross sections at EUV of different elements[9]](/richHtml/lop/2022/59/9/0922004/img_01.jpg)
Fig. 1. Atomic absorption cross sections at EUV of different elements[9]
![Mechanism of CAR[24]](/richHtml/lop/2022/59/9/0922004/img_02.jpg)
Fig. 2. Mechanism of CAR[24]
Fig. 3. Mechanism of the PMMA photochemical reaction[26]
Fig. 4. EUV lithography patterns of PMMA photoresist[30]
Fig. 7. Non-CARs based on Poly-p-hydroxystyrene derivatives[39-40]. (a) Matrix materials with photosensitive groups; (b)-(d) Matrix materials with olefinic or alkynyl groups; (e) free radical initiators; (f) (g) multi-mercapto crosslinkers
Fig. 8. Non-CARs with side-linked sulfonium ions [42]
Fig. 9. Matrix materials of ESCAP photoresist and their acid-catalyzed reaction[44]
Fig. 10. EUV lithography patterns of EUV-2D and MET-1K[49]. (a) (b) EUV-2D; (c) (d) MET-1K
Fig. 11. Polymethacrylate photoresists with side-linked leaving groups containing oxygen[50]
Fig. 12. Low activation energy photoresists and their acid-catalyzed reaction[51]
Fig. 13. EUV patterns of KRS photoresists[54]. (a) 35 nm linewidth, 1:1 duty cycle; (b) 28.3 nm linewidth, 1:4 duty cycle
Fig. 14. Polymeric photoresists with side-linked photoacid generators. (a) Cation[55]; (b) anion[56]
Fig. 15. Process flow of PSCARs[59]
Fig. 16. Mechanism of the generation of photosenitizers from their precursos in PSCARs[61]
Fig. 17. Mechanism of photochemisry reaction in PSCARs[61]
Fig. 18. Comparison of roughness between polymer photoresists and single molecule resin photoresists[24]
Fig. 19. Model of single-molecule resin CAR[68]
Fig. 20. Dendritic single-molecule resin with triphenyl core[70]
Fig. 22. TAS-tBoc-Ts single-molecule resin photoresist and its lithography mechanism[75]
Fig. 23. TAS-tBoc-Ts single-molecule resin[76]. (a) Structure; (b) mechanism
Fig. 25. Early calixarene photoresists. (a) Photoresists in Ref. [80]; (b) photoresists in Ref. [81]
Fig. 26. Calixarene photoresists [84]
Fig. 27. Noria Photoresists[86]
Fig. 29. Polymeric photoresist with silicon-containing side group[92]
Fig. 30. Polymeric photoresist with silicon- or boron-containing side group[93]
Fig. 31. Metal nanoparticle photoresists [97]
Fig. 32. Schematic of the ligand-displacement patterning mechanism for negative-tone pattern formation[107]
Fig. 33. Mechanism for the particle size increase of the negative-tone nanoparticle photoresists[108]
Fig. 34. Mechanism for solubility switching reactions induced by electron beam irradiation[109]
Fig. 35. Metal nanoparticle photoresists with polymeric ligands containing sulfurium [110]. (a) Structure; (b) patterns
Fig. 36. Tin-oxo cluster photoresists[111]. (a) Structure; (b) patterns
Fig. 37. Structure and EUV patterns of Zn-nTA cluster[117]
Fig. 38. Structure of Zn(MA)(TFA) clusters[118]
Fig. 39. Structure and photolithography patterns of polymeric photoresists with cobalt[119]
Fig. 40. Bismuth oligomer and their photolithography patterns[120]
Fig. 41. Polymeric photoresist with ferrocene and sulfonium and its photolighography patterns[121]
Fig. 42. Oxalic acid complexes of palladium and platinum and their photoreaction mechanism[122]
Fig. 43. JP-20 and its photolithography patterns[123]
Fig. 44. Photoresists with six-coordinated compounds of group VIII elements and their photolithography patterns[126]
Fig. 45. Single-molecule resin photoresists and their photolithography patterns. (a) Bisphenol A type[128]; (b) spirobifluorene type[129]
Set citation alerts for the article
Please enter your email address



