Zhongti Sun, Xiwen Chen, Wanjian Yin. Comprehensive first-principles studies on phase stability of copper-based halide perovskite derivatives AlCumXn (A = Rb and Cs; X = Cl, Br, and I)[J]. Journal of Semiconductors, 2020, 41(5): 052201
Search by keywords or author
- Journal of Semiconductors
- Vol. 41, Issue 5, 052201 (2020)
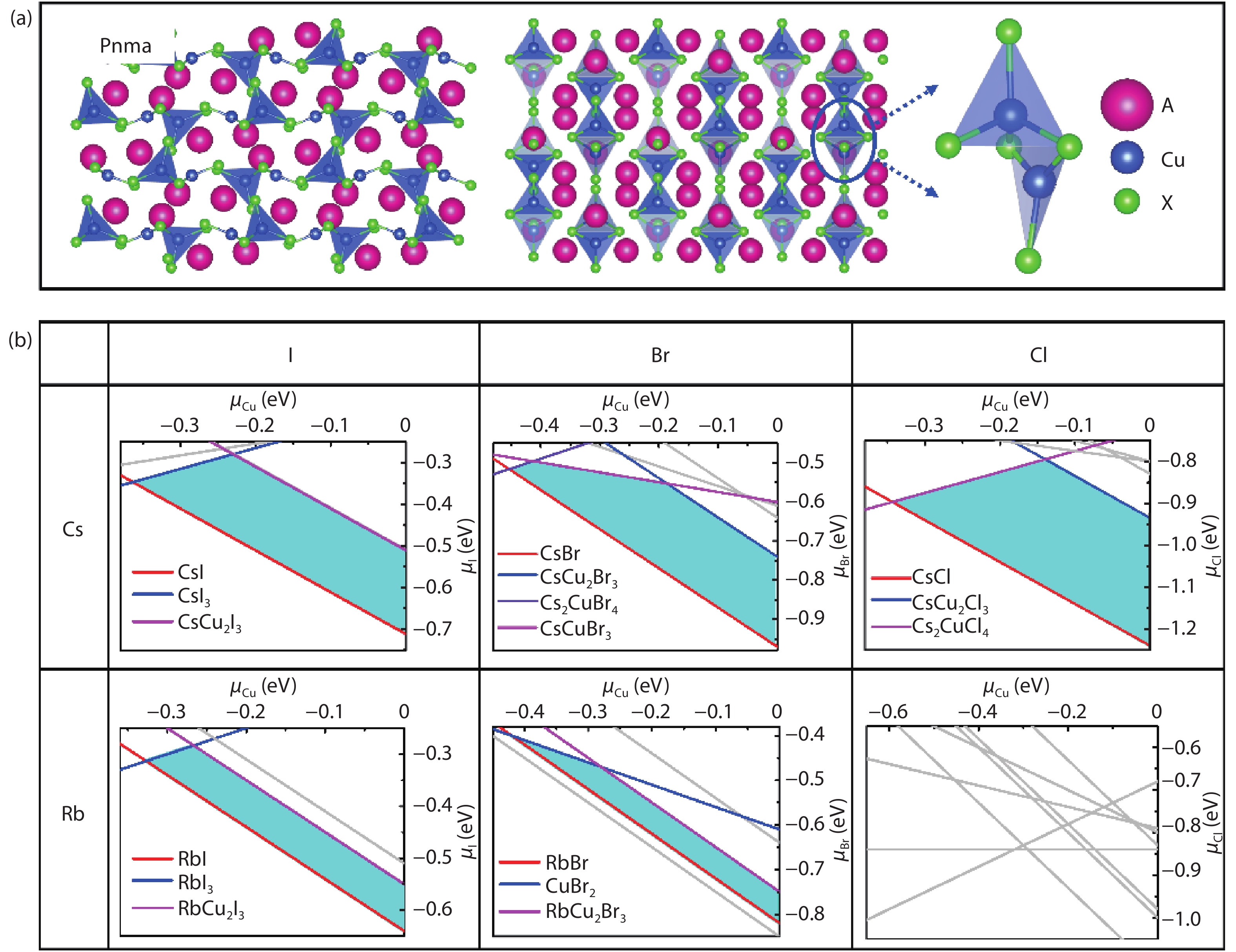
![(Color online) (a) The optimized structure of CHPs with 325-type, they own the isostructural model with space group of Pnma, isolated [Cu2X5]3– anion unit is composed of two types of Cu+ sites, a trigonal site and a tetragonal site. (b) Calculated phase stability regions versus μCu and μX (referring to the chemical potential of Cu and X from that of their elementary substance) from left to right for the X site of I, Br, and Cl element and up to down for Cs and Rb element on A site, respectively. The cyan polygon region represents thermodynamic stability growth region, which is encircled by possible competing phases using colored line (red, blue, violet, and pink). A, Cu, and X atoms are in purple, coral and brown, respectively.](/richHtml/jos/2020/41/5/052201/img_1.jpg)
Fig. 1. (Color online) (a) The optimized structure of CHPs with 325-type, they own the isostructural model with space group of Pnma, isolated [Cu2X5]3– anion unit is composed of two types of Cu+ sites, a trigonal site and a tetragonal site. (b) Calculated phase stability regions versus μ Cu and μ X (referring to the chemical potential of Cu and X from that of their elementary substance) from left to right for the X site of I, Br, and Cl element and up to down for Cs and Rb element on A site, respectively. The cyan polygon region represents thermodynamic stability growth region, which is encircled by possible competing phases using colored line (red, blue, violet, and pink). A, Cu, and X atoms are in purple, coral and brown, respectively.
![(Color online) (a) The structure model of CHPs with 123-type, they all own octahedral structure with the space group of Cmcm (No. 63), composed of edge-sharing [CuX4] tetrahedron 1D chain. (b) Calculated thermodynamic stability regions of ACu2X3 (A = Rb and Cs; X = Cl, Br, and I) CHPs versus μCu and μX (deviation of actual chemical potential of Cu and X from that of their elementary substance). The above from left to right is the CHP for the X site of I, Br, and Cl element, and up to down is for Cs and Rb element on A site, respectively. The cyan polygon region represents thermodynamic stable interval, which is surrounded by competing phases using colored line (red, blue, orange, violet, and pink). A, Cu, and X atoms are in purple, coral and brown, respectively.](/richHtml/jos/2020/41/5/052201/img_2.jpg)
Fig. 2. (Color online) (a) The structure model of CHPs with 123-type, they all own octahedral structure with the space group of Cmcm (No. 63), composed of edge-sharing [CuX4] tetrahedron 1D chain. (b) Calculated thermodynamic stability regions of ACu2X3 (A = Rb and Cs; X = Cl, Br, and I) CHPs versus μ Cu and μ X (deviation of actual chemical potential of Cu and X from that of their elementary substance). The above from left to right is the CHP for the X site of I, Br, and Cl element, and up to down is for Cs and Rb element on A site, respectively. The cyan polygon region represents thermodynamic stable interval, which is surrounded by competing phases using colored line (red, blue, orange, violet, and pink). A, Cu, and X atoms are in purple, coral and brown, respectively.
Fig. 3. (Color online) (a) The structure model of CHPs with 213-type, they possess isostructural model with Pnma symmetry, which is composed of 1D [Cu–X] tetrahedron chain with isolated alkali metal cation (Rb+ , Cs+ ). (b) Calculated phase stability regions in cyan polygon of A2CuX3 (A = Cs and Rb; X = I, Br, and Cl) CHPs versus μ Cu and μ X (deviation of actual chemical potential of Cu and X from that of their elementary substance). The cyan polygon region indicates thermodynamic phase stability growth interval, which is encompassed by competing phases with colored line (red, blue, and pink). A, Cu, and X atoms are in purple, coral and brown, respectively.
Fig. 4. (Color online) (a) The structure model of 459-type CHPs with space group of Pc, they own isolated [Cu5X9]4– anion with three types of Cu+ ions, a tetrahedral site, a trigonal site and a 2-fold coordination site. (b) Calculated thermodynamic stability regions of 459-type CHPs against μ Cu and μ X (deviation of actual chemical potential of Cu and X from that of their elementary substance). The cyan polygon region reveals thermodynamic stable growth interval and each colored line corresponds to one most probable competing phase. A, Cu, and X atoms are in purple, coral and brown, respectively.
Fig. 5. (Color online) (a) The structure model of 214-type CHPs, most of them possess octahedral space group Pnma with isolated [Cu–X] tetrahedron. But for Rb2CuCl4, it owns Cmca symmetry with 2D [Cu2Cl4]2– layers, induced by [Cu-Cl] octahedron Jahn-Teller distortion. (b) Calculated thermodynamic stability regions of A2CuX4 (A = Rb and Cs; X = Cl, Br, and I) halide perovskites versus μ Cu and μ X (deviation of actual chemical potential of Cu and X from that of their elementary substance). The horizontal and vertical axis is from I to Cl element and from Cs to Rb element, respectively. The cyan polygon region shows thermodynamic stability growth interval, which is encircled by most probable competing phases with colored lines (red, blue, violet, and pink). A, Cu, and X atoms are in purple, coral and brown, respectively.
Fig. 6. (Color online) (a) The structure model of 113-type CHPs, even though they possess different structures, they all have the same [CuX6] octahedron unit via face- and corner-sharing. (b) Calculated thermodynamic stability regions of ACuX3 (A = Rb and Cs; X = Cl, Br, and I) versus μ Cu and μ X (deviation of actual chemical potential of Cu and X from that of their elementary substance). The horizontal and vertical axis is from I to Cl element and from Cs to Rb element, respectively. The cyan polygon region represents thermodynamic stable growth interval, which is surrounded by most probable competing phases using colored line (red, blue, orange, violet, and pink). A, Cu, and X atoms are in purple, coral and brown, respectively.
Fig. 7. (Color online) (a) The structure model of 327-type CHPs, they all own tetragonal space group Ccca with isolated [Cu2X7]3– anion composing of elongated [Cu–X] octahedron unit. (b) Calculated thermodynamic stability regions of 327-type CHPs versus μ Cu and μ X (deviation of actual chemical potential of Cu and X from that of their elementary substance). The cyan polygon region represents phase stability growth condition, which is encircled by competing phases using colored line (red, blue, violet, and pink). A, Cu, and X atoms are in purple, coral and brown, respectively.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 1. The Space group of existing CHPs with various types encompassing 325-, 123-, 213-, 459-, 214-, 113-, and 327-type from inorganic crystal structure database (ICSD). ‘√’ and blank grid symbol indicates the existing and non-existing phase in experiment, respectively.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Calculated decomposition energies together with ODP in the CHPs Al Cum Xn (A = Rb and Cs; X = Cl, Br, and I; l , m , and n are integers).
|
Table 3. The relative energy per formula unit (eV/f.u.) of 214-type CHPs including Cs2CuI4, Rb2CuI4, and Rb2CuBr4 in the different space group encompassing Pnma and Cmca, which are all non-existing phase in the ICSD.
|
Table 4. The relative energy per formula unit (eV/f.u.) of 113-type CHP including CsCuI3, RbCuI3, and RbCuBr3 in the different space group containing C2221, P6122, Pbcn, which are non-existing phase in the ICSD.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 5. Summary for the stability region and decomposition energy of ODP in the different type CHPs Al Bm Xn (A = Rb and Cs; B = Cu; X = Cl, Br, and I; l , m , and n are integers; named as lmn-type). For each type CHP, number 1 and 2 represent decomposition energy and stability region, respectively. ‘√’ and ‘×’ symbols indicate ‘stable’ and ‘non-stable’ phase, respectively. Yellow square shows that this type is existing phase from inorganic Crystal Structure Database (ICSD).
Set citation alerts for the article
Please enter your email address



