- Journal of Semiconductors
- Vol. 41, Issue 5, 052201 (2020)
Abstract
1. Introduction
Artificial lighting accounts for one-fifth of global electricity consumption, with a half of that amount consumed by inefficient incandescent and fluorescent emission sources[
Recently, Cu-based halide perovskites and their derivatives (CHPs) have been emerging and attracted increasing attention because of the replacement of Pb with abundant, economic and environment-friendly Cu element[
Apart from 325- and 214-type CHPs which have been reported in lighting application, other phases such as 123-, 113-, 213-, 327-, and 459-type, have also been reported in experiments[
In this paper, first-principles calculations are performed to comprehensively investigate the thermodynamic stability of CHPs (AlCumXn, A = Rb and Cs; X = Cl, Br, and I) based on thermodynamic equilibrium growth condition. In total, there are 42 CHPs compounds with seven kinds of stoichiometric ratio including A3Cu2X5, ACu2X3, A2CuX3, A4Cu5X9, A2CuX4, ACuX3, and A3Cu2X7, as shown in Table 1. For chemical environments and stability calculations, all possible competing phases including compositional elements Rb, Cs, Cu, Cl, Br, I, binary compounds and ternary compounds from inorganic crystal structure database (ICSD) have been considered. We have found that most of existing AlCumXn phases in experiments have a stable growth region and positive decomposition energy in our calculations, which confirms the reliability of our calculations. Moreover, we also acquire ten new and stable phases encompassing Rb3Cu2I5, Rb3Cu2Br5, RbCu2Cl3, Rb2CuI3, Rb2CuBr4, RbCuBr3, Rb3Cu2Br7, Cs3Cu2Br7, Cs3Cu2Cl7, and Cs4Cu5Cl9, which are not yet reported experimentally. Therefore, our work provides prospective guidance for experiment to synthesize the above phases, amplifying the scope of phases of CHPs.
2. Computational details
All of the calculations were executed with spin-unrestricted density functional theory method, as implemented in the Vienna ab-initio simulation package (VASP)[
To evaluate the thermodynamic stability of CHPs AlCumXn (A = Rb and Cs; X = Cl, Br, and I) with different types, we first calculate the chemical potential range for equilibrium growth of compound to identify the proper chemical potentials for synthesizing particular compounds in experiments[
where μα is the chemical potential of constituent element α referring to the stable solid/gas in the growth conditions. ΔH is the formation enthalpy, AlCumXn and
Then, the thermodynamic stability of CHPs AlCumXn was furtherly confirmed through decomposition energy calculation based on optimal decomposition pathway (ODP) using linear programming method. Specific details are as follows:
where the xi is the molar fraction of possible competing phases, unknown variables. Eq. (4) is the decomposition pathway of host compounds, Eq. (5) is the decomposition energy between existing competing phases and host phase, Eq. (6) is the mass-conservation constraints for all atomic species, Eq. (7) is the minimum value of xi. The linear programming approach ensures that the calculated decomposition energy is based on the optimal decomposition pathway. If the value of ΔHd is a positive number, then this decomposition reaction is an endothermic reaction, indicating that this compound is thermodynamically stable.
3. Results and discussions
3.1. A3Cu2X5 phase
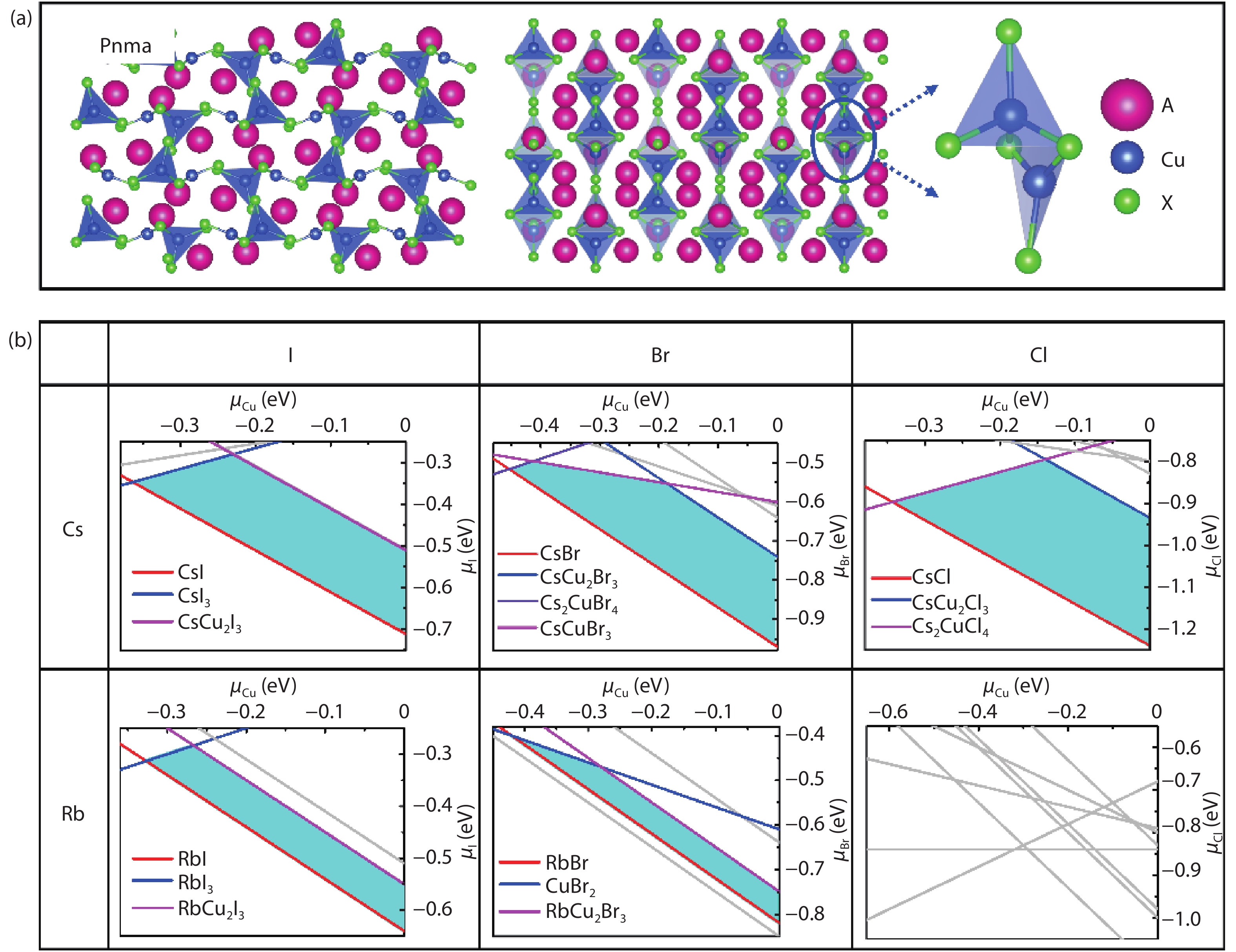
A3Cu2X5 phase exhibits orthorhombic crystal structure with space group of Pnma (No. 62) as shown in Fig. 1(a). It has two types of Cu+ ion sites, a tetrahedral site and a trigonal site, each of which constitutes spatially isolated [Cu2X5]3– (X = Cl, Br, I), encircled by A+ (A = Rb, Cs) ions. Thermodynamic stability of six kinds of 325-type CHPs are evaluated based on thermodynamic equilibrium growth conditions and decomposition energy with ODP as shown in Fig. 1(b) and Table 2, respectively. Except for Rb3Cu2Cl5, all five compounds possess stability region in cyan polygon. In consistent with experiments, Cs3Cu2X5 (X = Cl, Br, I) have been synthesized in experiments and Cs3Cu2I5 has been applied to the luminescent equipment with high PLQY[
![]()
Figure 1.(Color online) (a) The optimized structure of CHPs with 325-type, they own the isostructural model with space group of Pnma, isolated [Cu2X5]3– anion unit is composed of two types of Cu+ sites, a trigonal site and a tetragonal site. (b) Calculated phase stability regions versus
3.2. ACu2X3 phase
ACu2X3 phase exhibits orthorhombic space group of Cmcm (No. 63), the 1D chain with two [Cu–X] tetrahedra through edge-sharing in a row, isolated by A+ ion, as shown in Fig. 2(a). Fig. 2(b) shows the stability region of ACu2X3 phase in chemical potential map. The results show that all the ACu2X3 (A = Rb and Cs; X = Cl, Br, and I) compounds have thermodynamic stability region with cyan polyhedron, surrounded by the boundary composed of competing phases, which is in agreement with existing experiments that five of them have been synthesized successfully in experiments[
![]()
Figure 2.(Color online) (a) The structure model of CHPs with 123-type, they all own octahedral structure with the space group of Cmcm (No. 63), composed of edge-sharing [CuX4] tetrahedron 1D chain. (b) Calculated thermodynamic stability regions of ACu2X3 (A = Rb and Cs; X = Cl, Br, and I) CHPs versus
3.3. A2Cu1X3 phase
In contrast from the ACu2X3 phase with wider 1D chain by two Cu+ ions in one row, the A2CuX3 phase with the symmetry of Pnma (No. 62) also owns a 1D chain with only one Cu+ ion via vertex-sharing one line in Fig. 3(a). Their phase stability regions versus the chemical potential of Cu and X element are shown in Fig. 3(b). Only Rb2CuI3 and Rb2CuBr3 possess slim stability region with cyan polyhedron. Meanwhile, Rb2CuBr3 phase has been synthesized successfully in experiments[
![]()
Figure 3.(Color online) (a) The structure model of CHPs with 213-type, they possess isostructural model with Pnma symmetry, which is composed of 1D [Cu–X] tetrahedron chain with isolated alkali metal cation (Rb+ , Cs+ ). (b) Calculated phase stability regions in cyan polygon of A2CuX3 (A = Cs and Rb; X = I, Br, and Cl) CHPs versus
3.4. A4Cu5X9 phase
A4Cu5X9 compound is also a class of complex Cu-based compounds in Fig. 4(a). The crystal structure with the space group of Pc (No. 7) owns three types of Cu+ site, a tetragonal site, a trigonal site, and a 2-fold coordination site, which forms 0D isolated [Cu5X9]4– anion, isolated by A+ ions. Their phase stability regions versus the chemical potential of Cu and X element are shown in Fig. 4(b). Only two compounds, Rb4Cu5Cl9 and Cs4Cu5Cl9 own slim stability region with cyan polygon, surrounded by competing phases. The decomposition energies are 21 and 8 meV/atom for Rb4Cu5Cl9 and Cs4Cu5Cl9, respectively, in Table 2. Other 459-type CHP have negative ΔHd with the ODP. It is observed that the ODP for Rb4Cu5Cl9 phase is a disproportionation reaction, i.e., Rb4Cu5Cl9 → 12/5Cu + 1/5Rb2CuCl3 + 6/5Rb3Cu2Cl7, while for the Cs4Cu5Cl9, it is a non-redox reaction, i.e., Cs4Cu5Cl9 → 3/4Cs3Cu2Cl5 + 7/4CsCu2Cl3. Experimentally, Rb4Cu5Cl9 perovskite had been able to synthesize successfully, matches well with our predictions[
![]()
Figure 4.(Color online) (a) The structure model of 459-type CHPs with space group of Pc, they own isolated [Cu5X9]4– anion with three types of Cu+ ions, a tetrahedral site, a trigonal site and a 2-fold coordination site. (b) Calculated thermodynamic stability regions of 459-type CHPs against
3.5. A2CuX4 compounds
Since all the above CHPs own monovalent Cu+ ion on B site, their maximum coordination number is four, induced by higher 3d10 energy level and smaller ion radius of Cu, in consistent with the report by Xiao et al.[
![]()
Figure 5.(Color online) (a) The structure model of 214-type CHPs, most of them possess octahedral space group Pnma with isolated [Cu–X] tetrahedron. But for Rb2CuCl4, it owns Cmca symmetry with 2D [Cu2Cl4]2– layers, induced by [Cu-Cl] octahedron Jahn-Teller distortion. (b) Calculated thermodynamic stability regions of A2CuX4 (A = Rb and Cs; X = Cl, Br, and I) halide perovskites versus
3.6. ACuX3 compounds
Due to the divalent Cu2+ ions, the crystal structure of ACuX3 compounds also own elongated [Cu–X] octahedra via corner- and face-sharing connection, forming spatially isolated 1D coplanar chain and 3D network through vertex-sharing, surrounded by A+ ion in Fig. 6(a). Experimentally, CsCuBr3, CsCuCl3, and RbCuCl3 compounds have been synthesized successfully, where CsCuBr3 phase with space group of C2221 symmetry (No. 20) owns 3D network via vertex-sharing connection by coplanar double [CuX6] octahedron unit, while CsCuCl3 and RbCuCl3 phases possess 1D chain through face-sharing [CuX6] octahedron, they have different space group P6122 (No. 178) and Pbcn (No. 60), respectively[
![]()
Figure 6.(Color online) (a) The structure model of 113-type CHPs, even though they possess different structures, they all have the same [CuX6] octahedron unit via face- and corner-sharing. (b) Calculated thermodynamic stability regions of ACuX3 (A = Rb and Cs; X = Cl, Br, and I) versus
3.7. A3Cu2X7 phase
Last but not least, A3Cu2X7 phase with Cu2+ ion on B site owns spatially isolated 2D [Cu2Cl7]3– anion layer with double [Cu–X] octahedron layer by A+ ions, disparity with Rb2CuCl4 by single [Cu–Cl] octahedron layer, as illustrated in Fig. 7(a). Their stability growth regions in cyan polygon are also assessed against μCu and μX in Fig. 7(b). Except for Cs(Rb)3Cu2I7 compounds, others possess stability region with cyan polygon, consistent with the predictions of decomposition energy in Table 2. Experimentally, Rb3Cu2Cl7 compound has been synthesized successfully, our prediction signifies the experimental discovery[
![]()
Figure 7.(Color online) (a) The structure model of 327-type CHPs, they all own tetragonal space group Ccca with isolated [Cu2X7]3– anion composing of elongated [Cu–X] octahedron unit. (b) Calculated thermodynamic stability regions of 327-type CHPs versus
Table 5 summarizes the phase stability cases encompassing phase stability region and decomposition energy of ODP, whether or not they exist at room temperature. Most of existing phases are stable according to our predictions, except for Rb2CuCl4 and RbCuCl3 perovskites because they have more binary and ternary secondary phases and computational errors. Surprisingly, we also discover 10 novel CHPs with specific stability region and positive decomposition energy with ODP (i.e., Rb3Cu2I(Br)5, RbCu2Cl3, Rb2CuI3, Rb2CuBr4, RbCuBr3, Rb3Cu2Br7, Cs3Cu2Br(Cl)7 and Cs4Cu5Cl9) which have not yet been reported in experiment. Our results offer importance guidance to synthesize these phases, consequently broadening the range of existing CHPs.
4. Conclusion
In summary, we have systematically studied the stability of all ternary CHPs considering thermodynamic equilibrium growth conditions and decomposition energies. They all own lower electronic dimensionality including 2D layered, 1D chained and 0D isolated unit, surrounded by A+ ions. The coordination number of monovalent Cu (Cu+) in the CHP is less than 4, namely, 2-fold, trigonal, tetragonal site. The vast majority of CHPs with Cu2+ ion possess elongated octahedron induced by Jahn-Teller distortion. Most of existing CHPs are predicted to be stable, which is in consistent with the experiment. Furthermore, we discovered ten novel phases with specific stability region and positive decomposition energy with ODP via element exchange method, i.e. Rb3Cu2I(Br)5, RbCu2Cl3, Rb2CuI3, Rb2CuBr4, RbCuBr3, Rb3Cu2Br7, Cs3Cu2Br(Cl)7 and Cs4Cu5Cl9, which are not yet reported in experiment. Our predictions may provide insights for experimentalists to synthesize more novel inorganic CHPs, and will therefore tremendously expand the scope of existing CHP with promising applications.
Acknowledgements
The authors acknowledge funding support from National Natural Science Foundation of China (grant No. 11674237 and 51602211); National Key Research and Development Program of China (grant No. 2016YFB0700700); Natural Science Foundation of Jiangsu Province of China (grant No. BK20160299); the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD); and China Post-doctoral Foundation (grant No. 7131705619). The theoretical work was carried out at National Supercomputer Center in Tianjin and the calculations were performed on TianHe-1(A).
References
[1] Y Sun, N C Giebink, H Kanno et al. Management of singlet and triplet excitons for efficient white organic light-emitting devices. Nature, 440, 908(2006).
[2] J Luo, X Wang, S Li et al. Efficient and stable emission of warm-white light from lead-free halide double perovskites. Nature, 563, 541(2018).
[3] Z K Tan, R S Moghaddam, M L Lai et al. Bright light-emitting diodes based on organometal halide perovskite. Nat Nanotechnol, 9, 687(2014).
[4] W J Yin, T Shi, Y Yan. Unique properties of halide perovskites as possible origins of the superior solar cell performance. Adv Mater, 26, 4653(2014).
[5] H Cho, S H Jeong, M H Park et al. Overcoming the electroluminescence efficiency limitations of perovskite light-emitting diodes. Science, 350, 1222(2015).
[6] J Li, S G R Bade, X Shan et al. Single-layer light-emitting diodes using organometal halide perovskite/poly(ethylene oxide) composite thin films. Adv Mater, 27, 5196(2015).
[7] M I Saidaminov, J Almutlaq, S Sarmah et al. Pure Cs4PbBr6: highly luminescent zero-dimensional perovskite solids. ACS Energy Lett, 1, 840(2016).
[8] J H Cha, J H Han, W Yin et al. Photoresponse of CsPbBr3 and Cs4PbBr6 perovskite single crystals. J Phys Chem Lett, 8, 565(2017).
[9] M De Bastiani, I Dursun, Y Zhang et al. Inside perovskites: quantum luminescence from bulk Cs4PbBr6 single crystals. Chem Mater, 29, 7108(2017).
[10] D Cortecchia, H A Dewi, J Yin et al. Lead-free MA2CuCl
[11] H Yang, Y Zhang, J Pan et al. Room-temperature engineering of all-inorganic perovskite nanocrsytals with different dimensionalities. Chem Mater, 29, 8978(2017).
[12] J Yang, P Zhang, S H Wei. Band structure engineering of Cs2AgBiBr6 perovskite through order–disordered transition: a first-principle study. J Phys Chem Lett, 9, 31(2017).
[13] A M Elseman, A E Shalan, S Sajid et al. Copper-substituted lead perovskite materials constructed with different halides for working (CH3NH3)2CuX4-based perovskite solar cells from experimental and theoretical view. ACS Appl Mater Interfaces, 10, 11699(2018).
[14] T Jun, K Sim, S Iimura et al. Lead-free highly efficient blue-emitting Cs3Cu2I5 with 0D electronic structure. Adv Mater, 30, 1804547(2018).
[15] S Hull, P Berastegui. Crystal structures and ionic conductivities of ternary derivatives of the silver and copper monohalides — II: ordered phases within the (AgX)
[16] Z Xiao, K Du, W Meng et al. Chemical origin of the stability difference between copper(I)- and silver(I)-based halide double perovskites. Angew Chem Int Ed, 129, 12275(2017).
[17] P Yang, G Liu, B Liu et al. All-inorganic Cs2CuX4 (X = Cl, Br, and Br/I) perovskite quantum dots with blue-green luminescence. Chem Commun, 54, 11638(2018).
[18] L Helmholz, R F Kruh. The crystal structure of cesium chlorocuprate, Cs2CuCl4, and the spectrum of the chlorocuprate ion. J Am Chem Soc, 74, 1176(1952).
[19] F Aguado, F Rodríguez, R Valiente et al. Three-dimensional magnetic ordering in the Rb2CuCl4 layer perovskite—structural correlations. J Phys Condens Matter, 16, 1927(2004).
[20] A R Lim, S H Kim. Study of the structural phase transitions in RbCuCl3 and CsCuCl3 single crystals with the electric-magnetic-type interactions using a 87Rb and 133Cs nuclear magnetic resonance spectrometer. J Appl Phys, 101, 083519(2007).
[21] Y Kousaka, T Koyama, M Miyagawa et al. Crystal growth of chiral magnetic material in CsCuCl3. J Phys Conf Ser, 502, 012019(2014).
[22] G Kresse, J Furthmüller. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys Rev B, 54, 11169(1996).
[23] P E Blöchl. Projector augmented-wave method. Phys Rev B, 50, 17953(1994).
[24] J P Perdew, K Burke, M Ernzerh of. Generalized gradient approximation made simple. Phys Rev Lett, 77, 3865(1996).
[25] C Persson, Y J Zhao, S Lany et al. n-type doping of CuInSe2 and CuGaSe2. Phys Rev B, 72, 035211(2005).
[26] X G Zhao, D Yang, Y Sun et al. Cu–In halide perovskite solar absorbers. J Am Chem Soc, 139, 6718(2017).
Set citation alerts for the article
Please enter your email address



