- Journal of Semiconductors
- Vol. 41, Issue 9, 091709 (2020)
Abstract
1. Introduction
The realization of a zero-carbon society to address the energy and environmental issues has remained a great technical challenge. The utilization of solar energy, an inexhaustible natural energy source, is the ultimate option remaining to tackle the increasing global energy demands of modern society[
![]()
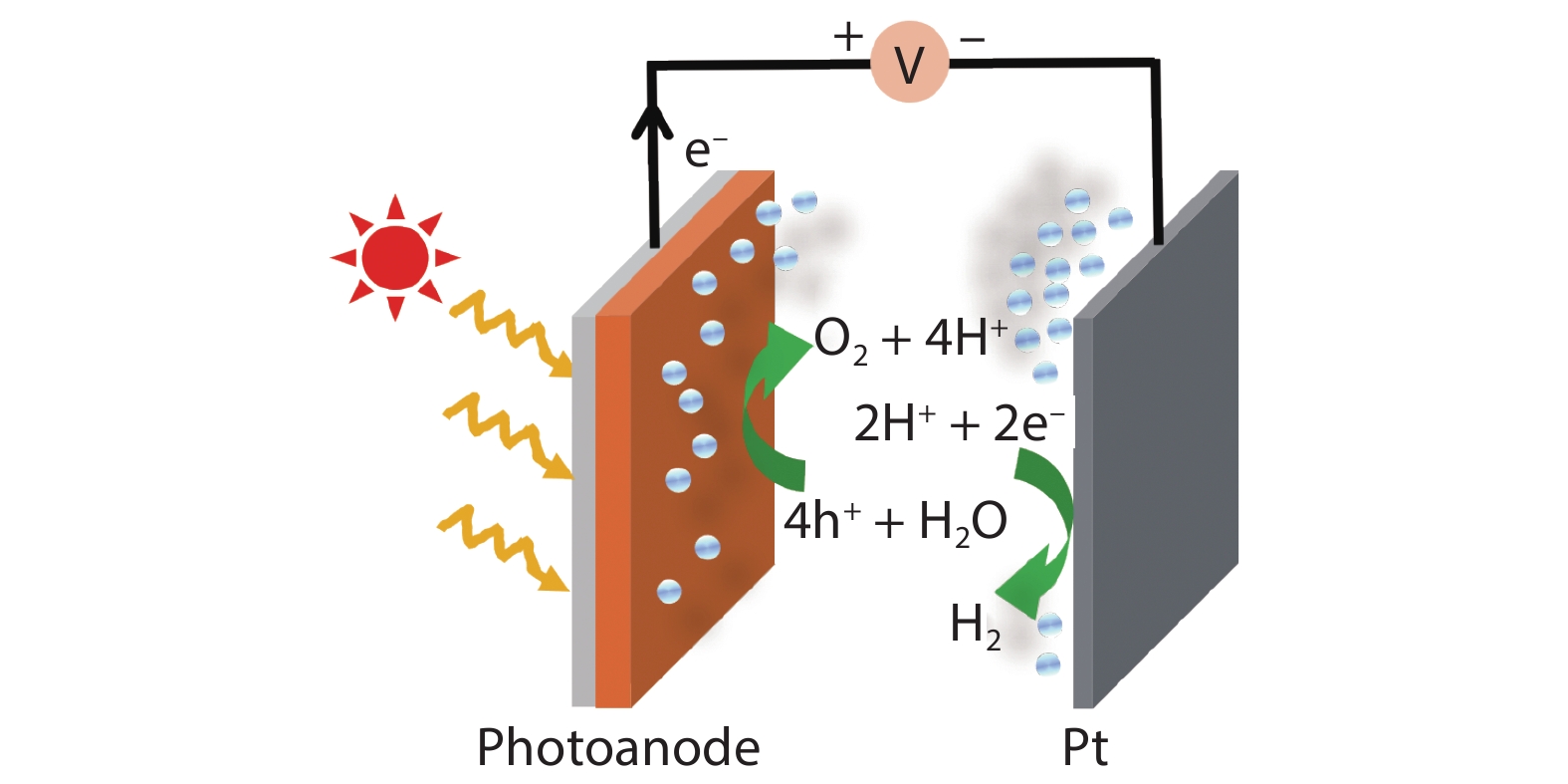
Figure 1.(Color online) Schematic of water splitting in photoanode-based PEC cell.
Among the metal oxide photoanode materials (Fig. 2), hematite (α-Fe2O3) is a typical metal oxide photoanode, owing to its intrinsic advantages of nontoxicity, favorable valence band position, good stability in aqueous solution, and narrow bandgap. Hematite has a band gap of about 2.1 eV, and thus a theoretical solar-to-hydrogen (STH) efficiency high as 15.4% can be achieved on hematite photoanode, which is higher than the commercial requirement STH efficiency (10%)[
![]()
Figure 2.(Color online) Ultimate photocurrent density and corresponding maximum STH of some semiconductors.
2. Advantages and limitations of hematite photoanode for solar water splitting
It is generally accepted that the water oxidation efficiency of photoanodes is affected by three synergetic processes: light absorbing; the separation of photogenerated electron–hole pairs (charge separation efficiency), photogenerated holes reacting with water molecules at photoanode/electrolyte interface (charge injection efficiency). For the hematite photoanode with a band gap of ~2.1 eV, its theoretical water oxidation photocurrent under standard solar illumination is 12.5 mA/cm2[
3. Typical strategies towards improving the charge transfer of hematite photoanode
To erase or attenuate above-mentioned issues that restrict the charge separation and transfer efficiency of hematite photoanode, several effective strategies are developed. For example (a) Nanostructuring of hematite increases the effective surface area and reduces the holes diffusion length. (b) Doping of hematite improves its conductivity, extends its holes’ lifetime, as well as reduces the electron–hole pairs recombination. (c) Loading of cocatalysts on hematite boosts its OER kinetics and improves the charge separation and transfer for water oxidation. (d) Surface passivating of hematite weakens the surface charge recombination. (e) Building heterojunctions enhance the charge separation and transfer efficiency of hematite through the heterojunction potential difference. The typical works and findings that related to above modified strategies for hematite photoanode are summarized as follows.
3.1. Nanostructuring
The short length of holes-diffusion (2–4 nm) in hematite critically suppresses the development of thick hematite film for effective light harvesting. As depicted in Fig. 3, although planar Fe2O3film can realize a long light penetration depth, it simultaneously causes severe recombination of photogenerated holes during water oxidation, when the diffusion length of holes exceeds 10 nm film thick. As such, the photocurrent of planar hematite electrodes is not still improved by increasing the film thickness. To overcome the contradiction, hematite photoanodes with porous structures or 1D nanostructures have been prepared by morphology controlling methods. These nanostructured hematite film compared with planar hematite films have following potential advantages (as nanostructured Fe2O3shown in Fig. 3): (1) Controlled film thickness for completing light absorption; (2) The smaller diameter of porous structures or 1D nanostructure for facilitating holes to reach electrolyte interface; (3) Larger specific surface area for the water molecules adsorption, dehydrogenation and activation[
![]()
Figure 3.(Color online) Comparison of electron–hole recombination in planar and 1D Fe2O3 nanorods arrays electrode.
![]()
Figure 4.(Color online) (a–c) SEM and (d) TEM images of obtained Fe2O3 by a facile rapid dehydration strategy (RD-Fe2O3). (e) Current density–voltage curves of obtained Fe2O3 by a conventional temperature-rising route (C-Fe2O3) and RD-Fe2O3 collected at 10 mV/s in 1.0 M KOH aqueous electrolyte under AM 1.5G illumination and in the dark. The solid and dashed lines represent the data collected under back (solid lines) and front (dashed lines) illuminations, respectively. Reproduced from Ref. [
In terms of charge separation and transfer efficiencies, as everyone knows that 1D nanostructures exhibit superior performances in PEC devices compared to other nanoarchitecture arrangements (e.g., 2D-nanostructures or 3D-nanostructures), but in special cases, these nanostructures are also beneficial to charge transport. For example when 2D-nanostructures (such as nanoflakes, nanoplatelets, etc.) perpendicularly oriented to the electrode, this arrangement can enhance charge transport through the directional transport of charge to the substrate, and reduce the charge recombination using the ultrathin thickness of 2D nanomaterials[
The section review shows the progress in morphology from thicker planar films to various 1D nanostructures, and then to recently special 2D or 3D nanostructure to improve the tradeoff between poor carrier collection and poor light absorption of hematite. It indicates nanostructure strategy is unquestionably well substantiated to promote the charge separation and transfer efficiency and thus enhancing PEC performance of hematite photoanode. So, continuing to design and fabricate peculiar and new nanostructures for high charge separation efficiency of photoelectrode is one of the key research goals.
3.2. Doping
When considering hematite for solar PEC water splitting, suitable elements doping is considered as an effective strategy to alleviate its bulk carrier recombination, since doping can improve the charge carrier mobility and concentration. In the recent reports, a beneficial effect of various dopants (such as Ti4+[
Due to the disadvantages of mono-doping, two- and multi-ion doping have caught extensive attention. Recent studies have been demonstrated that hematite co-doped with two cations has higher activity than that doped with single cation because two ions doping could significantly reduce the recombination centers and effectively improve the charge migration efficiency[
![]()
Figure 5.(Color online) (a) Mott–Schottky (M–S) plots measured in 1 M NaOH solution. Conditions: 1 kHz frequency. (b)
As we summarized above examples about hematite doping, regardless of n-type metal/non-transition metal doping, or mono-doping/multi-doping, these doping attempts are aim to modify the intrinsically limited conduction property of hematite[
3.3. Surface modification for charge separation
Besides doping and nanostructure engineering strategies, surface modification is also an effectual method for the improvement of the charge separation and transfer of hematite[
![]()
Figure 6.(Color online) Comparison of the band structure of an n-type semiconductor photoanode in the presence and the absence of passivation layers inside a PEC cell. (a) High charge recombination at surface defects and inefficient water oxidation by the photogenerated holes. (b) Use of an OER catalyst layer, which promotes facile hole transfer across the interface to the electrolyte for improving water oxidation. (c) Use of thin non-catalytic passivation layers which suppress surface recombination and improve water oxidation.
3.3.1. Cocatalyst
To modify the charge separation and injection of hematite photoanodes, various cocatalysts have been loaded on the hematite to improve its surface reaction kinetics for water oxidation. Typical OER cocatalysts are noble catalysts (such as, Pt, Ru) and metal oxides (such as, IrO2, RuO2). Even though the noble metal-based cocatalysts are efficient and stable under working conditions, there are not suitable to large-scale application in PEC devices owing to their high-cost and rarity. Therefore, cobalt-based and nickel-based compounds, chalcogenides have been developed as noble metal-free cocatalysts for the surface modification of hematite photoanode based on their high catalytic activity of water oxidation, low-cost and low-toxicity.
Among the noble metal-free cocatalysts, the typical compounds are cobalt oxides and amorphous cobalt–phosphate (Co–Pi)[
![]()
Figure 7.(Color online) (a)
In recent years, Ni-based catalysts, such as NiOOH, Ni-Bi, and Ni-Co double hydroxides, have also been investigated to enhance the surface activity of hematite photoanodes. Kelleyet al.reported NiO/α-Fe2O3 electrodes for PEC water splitting using atomic layer deposition method for the preparation of NiO. Compared to the untreated α-Fe2O3, they found that the as-deposited NiO on α-Fe2O3 could be converted into Ni(OH)2 during PEC water oxidation conditions, which resulted in the photocurrent onset potential of α-Fe2O3decreased by 300 mV and the photocurrent density increased by 200% at 1.23 V vs. RHE[
![]()
Figure 8.(Color online) (a) Scheme of charge transfer from Fe2O3 to H2O through Ni(OH)2 and/or IrO2. (b) Chronoamperometry measurement of Ti-Fe2O3, Ti-Fe2O3/Ni(OH)2, and Ti-Fe2O3/Ni(OH)2/IrO2 under a stepped potential. Reproduced from Ref. [
Besides Co-, and Ni-based catalysts possess high activity to OER, several other catalysts (e.g., FeOOH[
![]()
Figure 9.(Color online) (a) High-resolution TEM images of Fe2O3/FeB photoanode. (b) Charge separation and (c) injection efficiency at 1.23 V vs. RHE for the Fe2O3 and Fe2O3/FeB photoanodes. Reproduced from Ref. [
3.3.2. Surface passivation
Surface passivation is another efficient surface modification method to improve charge transfer and separation of hematite photoanode for solar water oxidation by removing unfavorable surface states of semiconductor, which often act as recombination centers competing intensively with charge transfer from the semiconductor to the electrolyte. Since some researchers had discovered surface states are located at just slightly positive than water oxidation potential (1.23 V vs. RHE) but more negative than valence band with two measures in neutral and basic conditions[
3.4. Building heterojunctions for charge transport and separation
Building heterojunctions is also one of the most common strategies to tackle the rapid bulk charge recombination and thus to increase PEC performance by increasing the charge spatial separation. So far, the models of heterojunction primarily include conventional type-II heterojunction, p–n heterojunction, Z-scheme photocatalytic system, semiconductor-metal heterojunction, and so on (as depicted schematically in Fig. 10). The four different heterojunctions have various mechanisms on enhancing charge separation which mainly depend on the electronic properties of the partner materials. For conventional type-II heterostructure, the conduction band (CB) and the valence band (VB) levels of semiconductor A are higher than the corresponding levels of the semiconductor B. Thus, the photogenerated electrons will transfer to semiconductor B, while the photogenerated holes will migrate to semiconductor A under light irradiation, resulting in a spatial separation of electron–hole pairs. For p–n heterojunctions, an additional internal electric field form at the heterojunction interface which lead to a band bending compared to conventional type-II heterostructure, electrons have a tendency to flow from the higher to the lower lying conduction band, while holes will follow the opposite direction. For Z-scheme photocatalytic systems, the photo-induced electrons on the semiconductor with a lower CB potential will combine with the holes on another semiconductor with a higher VB potential, and leave the electrons and holes persevere of the strong redox ability in reduction evolving and oxidation-evolving semiconductors, respectively, thus leading to superior charge separation. For semiconductor-metal type heterojunction, the Schottky barrier which is obtained by interfacing a semiconductor with a metal, is employed to drive electron moving from the material with the higher Fermi level to that with a lower one, until steady-state equilibrium is reached.
![]()
Figure 10.(Color online) Schematic illustration of (a) the type-II heterostructure, (b) p–n heterostructure, and (c) Z-scheme system without electron-mediators band alignments, and the correspondingly possible separation and transfer process of photoinduced electron–hole pairs of semiconductor photocatalysts. Reproduced from Refs. [
The conventional type-II and p–n heterojunction are common configuration to facilitate charge separation and transfer as well as the promotion of PEC water splitting performance due to their band matching effect or the formation of additional electric field at the interface of p–n junction. Various hematite-based heterojunction photoelectrode structures such as TiO2/Fe2O3, WO3/Fe2O3, MgFe2O4/Fe2O3, and Fe2O3/BiVO4, etc[
From charge transfer mechanism of the aforementioned two-types heterojunction standpoint, although the conventional type-II and p–n heterojunctions make the photogenerated electrons and holes spatially isolate, which greatly inhibits their undesirable recombination, the disadvantage is that the redox ability of photogenerated electrons and holes is weakened after charge transfer because the VB potential of semiconductor A is less positive than that of semiconductor B and the CB potential of semiconductor A is less negative than that of semiconductor B. Hence, it is difficult for the present heterojunction-type photocatalytic system to simultaneously possess the high charge-separation efficiency and strong redox ability. Thus, the development of a new-type photocatalytic system is urgently needed to solve the aforementioned problems.
The artificial Z-scheme photocatalytic system have attracted an ever-growing number of scientists to this field since the concept of "Z-scheme" is proposed by Bard et al. in 1979[
![]()
Figure 11.(Color online) Schematic for the energy band structure of the Fe2O3-NA/RGO/BiV1–
Other hematite-based heterojunctions were combined with nanocarbons as an electron conducting scaffold, such as reduced graphene oxide[
![]()
Figure 12.(Color online) (a) TiSi2 nanonet-based hematite nanostructure is essentially a core/shell arrangement where the core is the nanonet for effective charge collection and the shell is hematite for photocatalytic functionalities. The electronic band structure is shown in the enlarged cross-sectional view. (b) Low- and (c) high-magnification transmission electron microscopy (TEM) images showing the conformal coverage and crystallinity of hematite. (d)
As discussed above, one strategy fabricating heterojunction structures can enhance charge separation and transfer of electrodes. The main idea of heterojunction is to use electronic properties of additional material components to improve charge separation. So, further development of new materials for the design and fabrication of high-quality hematite-based heterojunction is one of the key research goals. Moreover, although Z-scheme photocatalytic system is more beneficial to spatially separate the electrons–holes due to its unique charge separation and transfer mechanism than other heterojunctions (e.g., conventional type-II heterojunction, and p–n heterojunction, etc.), the transfer pathway of photogenerated charge at the heterojunction interface and charge-migration kinetics in the Z-scheme heterojunction do not achieve a deeper understanding. Therefore, further investigation of these issues is important for confirming the formation of different types of heterojunction photocatalysts, and further advancements in theoretical calculations are highly desirable to shed some light on the true picture of the photocatalytic processes in the heterojunction photocatalysts.
4. Other strategies
Apart from above mentioned strategies, there are several other approaches have been reported to modify the hematite photoanode for solar water oxidation. Itoh, Liang and Bockris et al.have approved that the poor water oxidation activity of hematite photoanode depends on the enhanced bulk recombination that induced by the hematite/FTO substrate interface effect[
In addition, hematite-cocatalyst interface engineering is also a critical method for boosting the charge transfer from hematite to cocatalyst. In this approach, the cocatalysts usually coupled with other interlayers (such as hole-transport, hole-storage, electron-blocking layer), and the activity and stability of hematite photoanode can be obviously improved by the resultant interface engineering. For example, Ni-based complex could act as a holes-storage layer, and Ir-based complex could work as an OER catalyst, thus significantly enhanced photocurrent is achieved on the Ir-based complex/Ni-based complex/hematite photoanode[
5. Summary and Perspectives
In summary, the strategies developed for the modification of charge separation and transfer of hematite photoanode were summarized in the present review to help readers to get insight into the modifications progress for metal oxide photoanodes. The advantages and limitations of hematite photoanode for solar water splitting were firstly presented. Based on their function of improving hematite photoanode performance for solar water oxidation, several typical strategies including of nanostructuring, doping, surface modification and junction building are systematically categorized and introduced.
PEC water splitting is a promising pathway to produce hydrogen fuel using solar energy. Although the solar-to-fuel efficiency of PEC water splitting still some way off the requirements of commercialization, the progress in the hematite photoanode in the last ten years is heartening. In our opinion, the following research directions are important to develop high performance hematite photoanode. (1) The development of effective combination of experimental investigation and theoretical simulation to understand the charge separation and transfer in modified hematite photoanodes. (2) The systematical investigation of water oxidation mechanism on hematite photoanodes to guide developing new modification methods.
Acknowledgements
The work is supported by National Natural Science Foundation of China (41702037, 41831285, and 21773114).
References
[1] Z Wang, C Li, K Domen. Recent developments in heterogeneous photocatalysts for solar-driven overall water splitting. Chem Soc Rev, 48, 2109(2019).
[2] Z Zou, J Ye, K Sayama et al. Direct splitting of water under visible light irradiation with an oxide semiconductor photocatalyst. Nature, 414, 625(2001).
[3] A Fujishima, K Honda. Electrochemical photolysis of water at a semiconductor electrode. Nature, 238, 37(1972).
[4] Y Ma, X L Wang, Y S Jia et al. Titanium dioxide-based nanomaterials for photocatalytic fuel generations. Chem Rev, 114, 9987(2014).
[5] S C Wang, P Chen, Y Bai et al. New BiVO4 dual photoanodes with enriched oxygen vacancies for efficient solar-driven water splitting. Adv Mater, 30, 1800486(2018).
[6] W Wang, X M Xu, W Zhou et al. Recent progress in metal-organic frameworks for applications in electrocatalytic and photocatalytic water splitting. Adv Sci, 4, 1600371(2017).
[7] A G Tamirat, J Rick, A A Dubale et al. Using hematite for photoelectrochemical water splitting: A review of current progress and challenges. Nanoscale Horiz, 1, 243(2016).
[8] M Tahir, L Pan, F Idrees et al. Electrocatalytic oxygen evolution reaction for energy conversion and storage: A comprehensive review. Nano Energy, 37, 136(2017).
[9] I Roger, M A Shipman, M D Symes. Earth-abundant catalysts for electrochemical and photoelectrochemical water splitting. Nat Rev Chem, 1, 0003(2017).
[10] S H Shen, S A Lindley, X Y Chen et al. Hematite heterostructures for photoelectrochemical water splitting: Rational materials design and charge carrier dynamics. Energy Environ Sci, 9, 2744(2016).
[11] L M Carneiro, S K Cushing, C Liu et al. Excitation-wavelength-dependent small polaron trapping of photoexcited carriers in
[12] T Wang, W J Luo, X Wen et al. Nonequilibrium Ti4+ doping significantly enhances the performance of Fe2O3 photoanodes by quenching. ChemNanoMat, 2, 652(2016).
[13] K Sivula, Formal F Le, M Grätzel. Solar water splitting: Progress using hematite (
[14] D A Wheeler, G M Wang, Y C Ling et al. Nanostructured hematite: Synthesis, characterization, charge carrier dynamics, and photoelectrochemical properties. Energy Environ Sci, 5, 6682(2012).
[15] S R Pendlebury, A J Cowan, M Barroso et al. Correlating long-lived photogenerated hole populations with photocurrent densities in hematite water oxidation photoanodes. Energy Environ Sci, 5, 6304(2012).
[16] M Barroso, S R Pendlebury, A J Cowan et al. Charge carrier trapping, recombination and transfer in hematite (
[17] G G Bessegato, T T Guaraldo, M V B Zanoni et al. Enhancement of photoelectrocatalysis efficiency by using nanostructured electrodes. Mod Electrochem Methods Nano, Surface Corros Sci, 11, 27(2014).
[18] A G Tamirat, W N Su, A A Dubale et al. Photoelectrochemical water splitting at low applied potential using a NiOOH coated codoped (Sn, Zr)
[19] L C Jia, K Harbauer, P Bogdanoff et al.
[20] G M Wang, Y C Ling, X H Lu et al. A mechanistic study into the catalytic effect of Ni(OH)2 on hematite for photoelectrochemical water oxidation. Nanoscale, 5, 4129(2013).
[21] J Y Kim, G Magesh, D H Youn et al. Single-crystalline, wormlike hematite photoanodes for efficient solar water splitting. Sci Rep, 3, 2681(2013).
[22] C D Bohn, A K Agrawal, E C Walter et al. Effect of tin doping on
[23] B Klahr, S Gimenez, F Fabregat-Santiago et al. Water oxidation at hematite photoelectrodes: The role of surface states. J Am Chem Soc, 134, 4294(2012).
[24] U Bjoerksten, J Moser, M Graetzel. Photoelectrochemical studies on nanocrystalline hematite films. Chem Mater, 6, 858(1994).
[25] J Vura-Weis, C M Jiang, C Liu et al. Femtosecond M2, 3-edge spectroscopy of transition-metal oxides: Photoinduced oxidation state change in
[26] A G Joly, J R Williams, S A Chambers et al. Carrier dynamics in
[27] M P Dare-Edwards, J B Goodenough, A Hamnett et al. Electrochemistry and photoelectrochemistry of iron(III) oxide. J Chem Soc, Faraday Trans 1, 79, 2027(1983).
[28] K Sivula. Metal oxide photoelectrodes for solar fuel production, surface traps, and catalysis. J Phys Chem Lett, 4, 1624(2013).
[29] H Dotan, O Kfir, E Sharlin et al. Resonant light trapping in ultrathin films for water splitting. Nat Mater, 12, 158(2013).
[30] J T Li, S K Cushing, P Zheng et al. Plasmon-induced photonic and energy-transfer enhancement of solar water splitting by a hematite nanorod array. Nat Commun, 4, 2651(2013).
[31] X G Wen, S H Wang, Y Ding et al. Controlled growth of large-area, uniform, vertically aligned arrays of
[32] C C Li, A Li, Z B Luo et al. Inside back cover: Surviving high-temperature calcination: ZrO2 -induced hematite nanotubes for photoelectrochemical water oxidation. Angew Chem Int Ed, 56, 4361(2017).
[33] M Y Li, Y Yang, Y C Ling et al. Morphology and doping engineering of Sn-doped hematite nanowire photoanodes. Nano Lett, 17, 2490(2017).
[34] C J Jia, L D Sun, Z G Yan et al. Iron oxide tube-in-tube nanostructures. J Phys Chem C, 111, 13022(2007).
[35] U Cvelbar, Z Q Chen, M K Sunkara et al. Spontaneous growth of superstructure
[36] A Z Liao, H C He, L Q Tang et al. Quasi-topotactic transformation of FeOOH nanorods to robust Fe2O3 porous nanopillars triggered with a facile rapid dehydration strategy for efficient photoelectrochemical water splitting. ACS Appl Mater Interfaces, 10, 10141(2018).
[37] J S Chen, X W Lou. Anatase TiO2 nanosheet: An ideal host structure for fast and efficient lithium insertion/extraction. Electrochem Commun, 11, 2332(2009).
[38] S Hoang, S P Berglund, R R Fullon et al. Chemical bath deposition of vertically aligned TiO2 nanoplatelet arrays for solar energy conversion applications. J Mater Chem A, 1, 4307(2013).
[39] L Wang, C Y Lee, A Mazare et al. Enhancing the water splitting efficiency of Sn-doped hematite nanoflakes by flame annealing. Chem Eur J, 20, 77(2014).
[40] M Ji, J G Cai, Y R Ma et al. Controlled growth of ferrihydrite branched nanosheet arrays and their transformation to hematite nanosheet arrays for photoelectrochemical water splitting. ACS Appl Mater Interfaces, 8, 3651(2016).
[41] H Han, F Riboni, F Karlicky et al. A-Fe2O3/TiO2 3D hierarchical nanostructures for enhanced photoelectrochemical water splitting. Nanoscale, 9, 134(2017).
[42] C W Cheng, H J Fan. Branched nanowires: Synthesis and energy applications. Nano Today, 7, 327(2012).
[43] H N Chen, S H Yang. Hierarchical nanostructures of metal oxides for enhancing charge separation and transport in photoelectrochemical solar energy conversion systems. Nanoscale Horiz, 1, 96(2016).
[44] Y G Li, X L Wei, B W Zhu et al. Hierarchically branched Fe2O3@TiO2nanorod arrays for photoelectrochemical water splitting: Facile synthesis and enhanced photoelectrochemical performance. Nanoscale, 8, 11284(2016).
[45] Z B Luo, T Wang, J J Zhang et al. Cover picture: Dendritic hematite nanoarray photoanode modified with a conformal titanium dioxide interlayer for effective charge collection. Angew Chem Int Ed, 56, 12791(2017).
[46] A W Pu, J J Deng, M Li et al. Coupling Ti-doping and oxygen vacancies in hematite nanostructures for solar water oxidation with high efficiency. J Mater Chem A, 2, 2491(2014).
[47] D P Cao, W J Luo, M X Li et al. A transparent Ti4+ doped hematite photoanode protectively grown by a facile hydrothermal method. CrystEngComm, 15, 2386(2013).
[48] J C Launay, G Horowitz. Crystal growth and photoelectrochemical study of Zr-doped
[49] M A Lukowski, S Jin. Improved synthesis and electrical properties of Si-doped
[50] Y C Ling, G M Wang, D A Wheeler et al. Sn-doped hematite nanostructures for photoelectrochemical water splitting. Nano Lett, 11, 2119(2011).
[51] H W Chang, Y M Fu, W Y Lee et al. Visible light-induced electronic structure modulation of Nb- and Ta-doped
[52] Y S Hu, A Kleiman-Shwarsctein, A J Forman et al. Pt-doped
[53] A Annamalai, H H Lee, S H Choi et al. Sn/be sequentially co-doped hematite photoanodes for enhanced photoelectrochemical water oxidation: Effect of Be2+ as co-dopant. Sci Rep, 6, 23183(2016).
[54] S Shen, n J Chen, M Wang et al. Titanium dioxide nanostructures for photoelectrochemical applications. Prog Mater Sci, 98, 299(2018).
[55] S Li, J J Cai, Y L Liu et al. Tuning orientation of doped hematite photoanodes for enhanced photoelectrochemical water oxidation. Sol Energy Mater Sol Cells, 179, 328(2018).
[56] M L Zhang, W J Luo, Z S Li et al. Improved photoelectrochemical responses of Si and Ti codoped
[57] J J Cai, H Chen, C X Liu et al. Engineered Sn- and Mg-doped hematite photoanodes for efficient photoelectrochemical water oxidation. Dalton Trans, 49, 11282(2020).
[58] F Amano, B Ohtani, H Yoshida. Role of doped titanium species in the enhanced photoelectrochemical properties of iron oxide films: Comparison between water oxidation and iodide ion oxidation. J Electroanal Chem, 766, 100(2016).
[59] R Liu, Z Zheng, J Spurgeon et al. Enhanced photoelectrochemical water-splitting performance of semiconductors by surface passivation layers. Energy Environ Sci, 7, 2504(2014).
[60] L F Xi, S Y Chiam, W F Mak et al. A novel strategy for surface treatment on hematite photoanode for efficient water oxidation. Chem Sci, 4, 164(2013).
[61] D K Lee, K S Choi. Enhancing long-term photostability of BiVO4 photoanodes for solar water splitting by tuning electrolyte composition. Nat Energy, 3, 53(2018).
[62] J R Ran, J Zhang, J G Yu et al. Earth-abundant cocatalysts for semiconductor-based photocatalytic water splitting. Chem Soc Rev, 43, 7787(2014).
[63] J R Xiao, L L Fan, F G Zhao et al. Kinetic analysis of the synergistic effect of NaBH4 treatment and Co–Pi coating on Fe2O3 photoanodes for photoelectrochemical water oxidation. J Catal, 381, 139(2020).
[64] D Chen, Z F Liu, Z G Guo et al. 3D branched Ca-Fe2O3/Fe2O3 decorated with Pt and Co–Pi: Improved charge-separation dynamics and photoelectrochemical performance. ChemSusChem, 12, 3286(2019).
[65] B Klahr, S Gimenez, F Fabregat-Santiago et al. Photoelectrochemical and impedance spectroscopic investigation of water oxidation with “Co–Pi”-coated hematite electrodes. J Am Chem Soc, 134, 16693(2012).
[66] D K Zhong, D R Gamelin. Photoelectrochemical water oxidation by cobalt catalyst (“Co–Pi”)/
[67] G M Carroll, D R Gamelin. Kinetic analysis of photoelectrochemical water oxidation by mesostructured Co–Pi/
[68] M W Kanan, J Yano, Y Surendranath et al. Structure and valency of a cobalt–phosphate water oxidation catalyst determined by
[69] J G McAlpin, Y Surendranath, M Dincǎ et al. EPR evidence for Co(IV) species produced during water oxidation at neutral pH. J Am Chem Soc, 132, 6882(2010).
[70] S C Riha, B M Klahr, E C Tyo et al. Atomic layer deposition of a submonolayer catalyst for the enhanced photoelectrochemical performance of water oxidation with hematite. ACS Nano, 7, 2396(2013).
[71] L F Xi, P D Tran, S Y Chiam et al. Co3O4-decorated hematite nanorods as an effective photoanode for solar water oxidation. J Phys Chem C, 116, 13884(2012).
[72] K M H Young, T W Hamann. Enhanced photocatalytic water oxidation efficiency with Ni(OH)2 catalysts deposited on
[73] Y R Hong, Z L Liu, S F B S A Al-Bukhari et al. Effect of oxygen evolution catalysts on hematite nanorods for solar water oxidation. Chem Commun, 47, 10653(2011).
[74] Z L Wang, G J Liu, C M Ding et al. Synergetic effect of conjugated Ni(OH)2/IrO2 cocatalyst on titanium-doped hematite photoanode for solar water splitting. J Phys Chem C, 119, 19607(2015).
[75] M Wang, J Q Wang, C Xi et al. A hydrogen-deficient nickel–cobalt double hydroxide for photocatalytic overall water splitting. Angew Chem Int Ed, 59, 11510(2020).
[76] J Y Kim, D H Youn, K Kang et al. Highly conformal deposition of an ultrathin FeOOH layer on a hematite nanostructure for efficient solar water splitting. Angew Chem Int Ed, 128, 11012(2016).
[77] Q Yu, X G Meng, T Wang et al. Hematite films decorated with nanostructured ferric oxyhydroxide as photoanodes for efficient and stable photoelectrochemical water splitting. Adv Funct Mater, 25, 2686(2015).
[78] L Liardet, J E Katz, J S Luo et al. An ultrathin cobalt–iron oxide catalyst for water oxidation on nanostructured hematite photoanodes. J Mater Chem A, 7, 6012(2019).
[79] J W Jang, C Du, Y F Ye et al. Enabling unassisted solar water splitting by iron oxide and silicon. Nat Commun, 6, 7447(2015).
[80] A Z Liao, H C He, Z W Fan et al. Facile room-temperature surface modification of unprecedented FeB co-catalysts on Fe2O3 nanorod photoanodes for high photoelectrochemical performance. J Catal, 352, 113(2017).
[81] A Z Liao, R T Chen, F T Fan et al. Integration of Fe
[82] O Zandi, T W Hamann. Enhanced water splitting efficiency through selective surface state removal. J Phys Chem Lett, 5, 1522(2014).
[83] Y Yang, M Forster, Y C Ling et al. Acid treatment enables suppression of electron-hole recombination in hematite for photoelectrochemical water splitting. Angew Chem Int Ed, 55, 3403(2016).
[84] J Y Kim, J W Jang, D H Youn et al. A stable and efficient hematite photoanode in a neutral electrolyte for solar water splitting: Towards stability engineering. Adv Energy Mater, 4, 1400476(2014).
[85] M Barroso, C A Mesa, M Grätzel et al. From the cover: Chemical approaches to artificial photosynthesis. Proc Natl Acad Sci USA, 109, 15560(2012).
[86] G J Liu, J Y Shi, F X Zhang et al. A tantalum nitride photoanode modified with a hole-storage layer for highly stable solar water splitting. Angew Chem Int Ed, 53, 7295(2014).
[87] Q F Gui, Z Xu, H F Zhang et al. Enhanced photoelectrochemical water splitting performance of anodic TiO2 nanotube arrays by surface passivation. ACS Appl Mater Interfaces, 6, 17053(2014).
[88] J J Kelly. The influence of surface recombination and trapping on the cathodic photocurrent at p-type III–V electrodes. J Electrochem Soc, 129, 730(1982).
[89] L Steier, I Herraiz-Cardona, S Gimenez et al. Understanding the role of underlayers and overlayers in thin film hematite photoanodes. Adv Funct Mater, 24, 7681(2014).
[90] Formal F Le, N Tétreault, M Cornuz et al. Passivating surface states on water splitting hematite photoanodes with alumina overlayers. Chem Sci, 2, 737(2011).
[91] P Y Tang, J Arbiol. Engineering surface states of hematite based photoanodes for boosting photoelectrochemical water splitting. Nanoscale Horiz, 4, 1256(2019).
[92] P Zhou, J G Yu, M Jaroniec. All-solid-state Z-scheme photocatalytic systems. Adv Mater, 26, 4920(2014).
[93] H J Li, Y Zhou, W G Tu et al. State-of-the-art progress in diverse heterostructured photocatalysts toward promoting photocatalytic performance. Adv Funct Mater, 25, 998(2015).
[94] A J Cowan, J R Durrant. Long-lived charge separated states in nanostructured semiconductor photoelectrodes for the production of solar fuels. Chem Soc Rev, 42, 2281(2013).
[95] H L Wang, L S Zhang, Z G Chen et al. Semiconductor heterojunction photocatalysts: Design, construction, and photocatalytic performances. Chem Soc Rev, 43, 5234(2014).
[96] K Sivula, F L Formal, M Grätzel. WO3–Fe2O3 photoanodes for water splitting: A host scaffold, guest absorber approach. Chem Mater, 21, 2862(2009).
[97] P Zhao, C X Kronawitter, X F Yang et al. WO3–
[98] M T Mayer, C Du, D W Wang. Hematite/Si nanowire dual-absorber system for photoelectrochemical water splitting at low applied potentials. J Am Chem Soc, 134, 12406(2012).
[99] X Wang, K Q Peng, Y Hu et al. Silicon/hematite core/shell nanowire array decorated with gold nanoparticles for unbiased solar water oxidation. Nano Lett, 14, 18(2014).
[100] K J McDonald, K S Choi. Synthesis and photoelectrochemical properties of Fe2O3/ZnFe2O4 composite photoanodes for use in solar water oxidation. Chem Mater, 23, 4863(2011).
[101] Y M Wang, T Yu, X Y Chen et al. Enhancement of photoelectric conversion properties of SrTiO3/
[102] J T Li, F K Meng, S Suri et al. Photoelectrochemical performance enhanced by a nickel oxide –hematite p –n junction photoanode. Chem Commun, 48, 8213(2012).
[103] D K Bora, A Braun, R Erni et al. Hematite–NiO/
[104] A J Bard. Photoelectrochemistry and heterogeneous photo-catalysis at semiconductors. J Photochem, 10, 59(1979).
[105] Y Hou, F Zuo, A Dagg et al. Visible light-driven
[106] A Z Liao, Y Zhou, L X Xiao et al. Correction: Direct Z scheme-fashioned photoanode systems consisting of Fe2O3 nanorod arrays and underlying thin Sb2Se3 layers toward enhanced photoelectrochemical water splitting performance. Nanoscale, 11, 1451(2019).
[107] A Rehman, S Zulfiqar, I Shakir et al. Nanocrystalline hematite α-Fe2O3 synthesis with different precursors and their composites with graphene oxide. Ceram Int, 46, 8227(2020).
[108] W L Fu, K Liu, X Zou et al. Surface engineering of defective hematite nanostructures coupled by graphene sheets with enhanced photoelectrochemical performance. ACS Sustain Chem Eng, 7, 12750(2019).
[109] S A Carminati, A do Nascimento Barbosa, A L de Freitas et al. Unraveling the role of single layer graphene as overlayer on hematite photoanodes. J Catal, 372, 109(2019).
[110] H W Lan, Y J Xia, K Feng et al. Co-doped carbon layer to lower the onset potential of hematite for solar water oxidation. Appl Catal B, 258, 117962(2019).
[111] V Strauss, M Anderson, C X Wang et al. Carbon nanodots as feedstock for a uniform hematite-graphene nanocomposite. Small, 14, 1803656(2018).
[112] Z R Xie, H L Tan, X M Wen et al. The importance of the interfacial contact: Is reduced graphene oxide always an enhancer in photo(electro)catalytic water oxidation. ACS Appl Mater Interfaces, 11, 23125(2019).
[113] H L Tan, H A Tahini, X M Wen et al. Interfacing BiVO4 with reduced graphene oxide for enhanced photoactivity: A tale of facet dependence of electron shuttling. Small, 12, 5295(2016).
[114] M T Mayer, Y J Lin, G B Yuan et al. Forming heterojunctions at the nanoscale for improved photoelectrochemical water splitting by semiconductor materials: Case studies on hematite. Acc Chem Res, 46, 1558(2013).
[115] S Rai, A Ikram, S Sahai et al. Morphological, optical and photoelectrochemical properties of Fe2O3–GNP composite thin films. RSC Adv, 4, 17671(2014).
[116] Y J Lin, S Zhou, S W Sheehan et al. Nanonet-based hematite heteronanostructures for efficient solar water splitting. J Am Chem Soc, 133, 2398(2011).
[117] T Hisatomi, H Dotan, M Stefik et al. Enhancement in the performance of ultrathin hematite photoanode for water splitting by an oxide underlayer. Adv Mater, 24, 2699(2012).
[118] Formal F Le, M Grätzel, K Sivula. Controlling photoactivity in ultrathin hematite films for solar water-splitting. Adv Funct Mater, 20, 1099(2010).
[119] F L Souza, K P Lopes, E Longo et al. The influence of the film thickness of nanostructured
[120] D Wang, X T Zhang, P P Sun et al. Enhanced photoelectrochemical water splitting on hematite thin film with layer-by-layer deposited ultrathin TiO2 underlayer. Int J Hydrog Energy, 39, 16212(2014).
[121] D P Cao, W J Luo, J Y Feng et al. Cathodic shift of onset potential for water oxidation on a Ti4+doped Fe2O3 photoanode by suppressing the back reaction. Energy Environ Sci, 7, 752(2014).
[122] H W Gao, C Liu, H E Jeong et al. Plasmon-enhanced photocatalytic activity of iron oxide on gold nanopillars. ACS Nano, 6, 234(2012).
[123] S Ramadurgam, T G Lin, C Yang. Aluminum plasmonics for enhanced visible light absorption and high efficiency water splitting in core-multishell nanowire photoelectrodes with ultrathin hematite shells. Nano Lett, 14, 4517(2014).
Set citation alerts for the article
Please enter your email address



