Hongtao Ren, Yachao Liu, Lei Zhang, Kai Liu. Synthesis, properties, and applications of large-scale two-dimensional materials by polymer-assisted deposition[J]. Journal of Semiconductors, 2019, 40(6): 061003
Search by keywords or author
- Journal of Semiconductors
- Vol. 40, Issue 6, 061003 (2019)
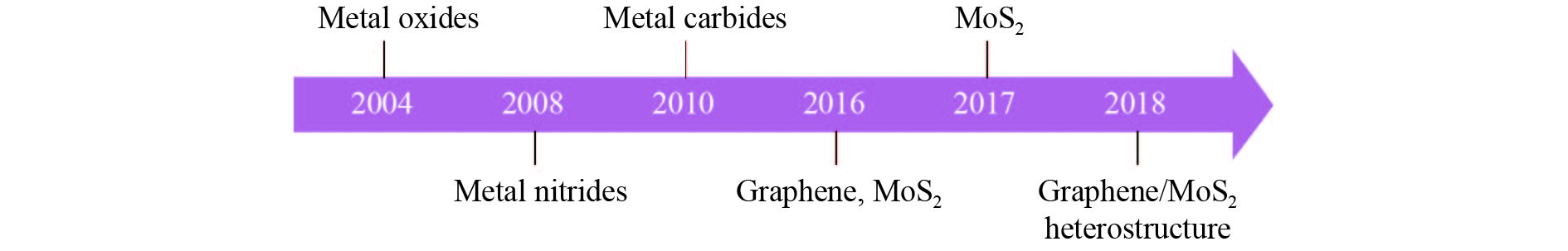
Fig. 1. (Color online) Timeline showing key development by polymer-assisted deposition. Metal oxides; metal nitrides; metal carbides; glassy-graphene; MoS2; MoS2/glassy-graphene heterostructure.
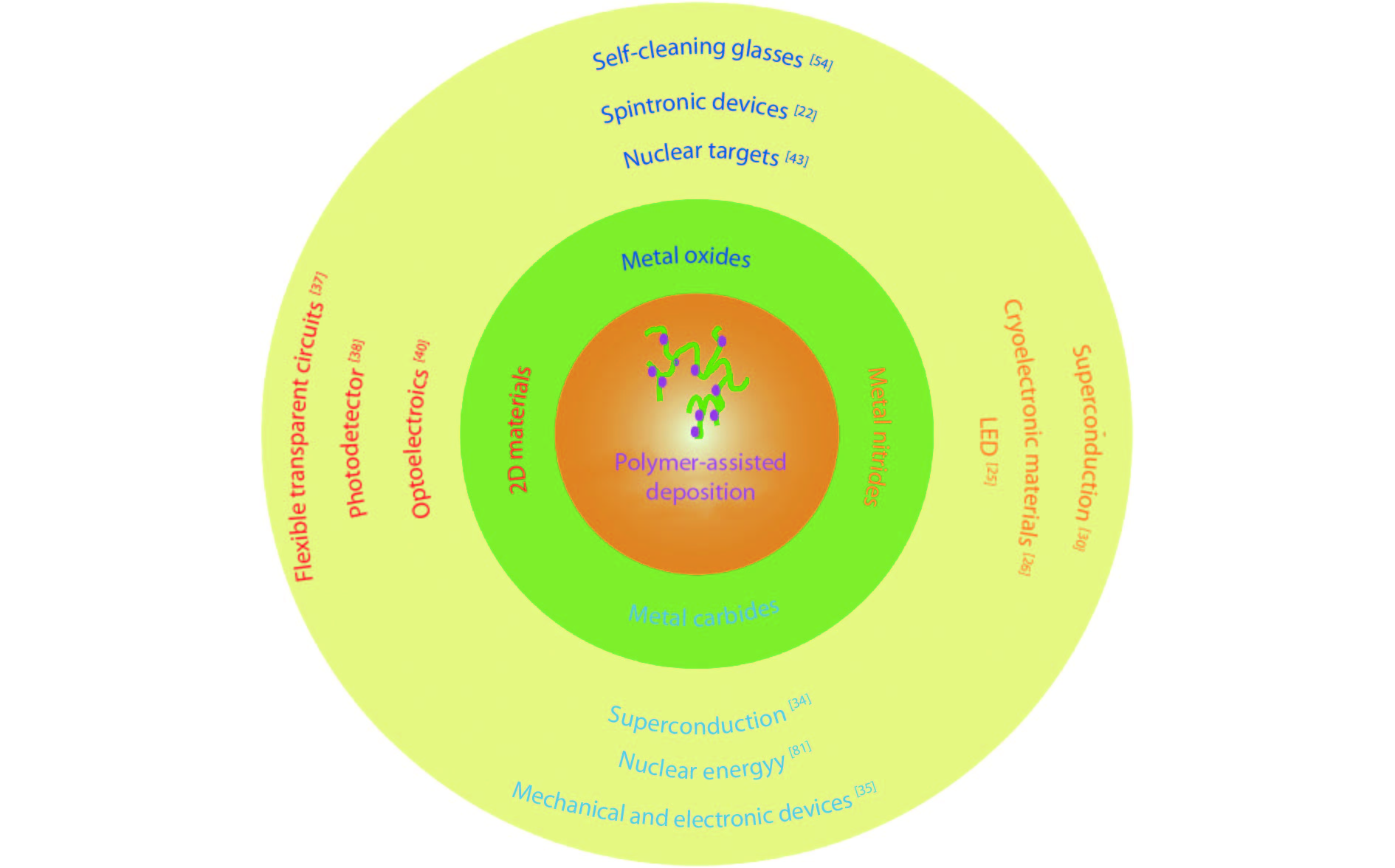
Fig. 2. (Color online) Application of as-grown thin films by PAD.
Fig. 3. (Color online) Schematic illustration of the main processing steps used to grow thin films by PAD.
Fig. 4. (Color online) Evolution from glassy carbon to glassy-graphene and graphene[37 ].
Fig. 5. (Color online) Preparation of glassy graphene-based circuits and the flexibility test[37 ].
Fig. 6. (Color online) Thickness-dependent bandgap tunable MoS2 thin films for optoelectronics[40 ].
Fig. 7. (Color online) Wafer-scale synthesis of MoS2 thin films via polymer-assisted deposition[39 ].
Fig. 8. (Color online) Formation of large-area web buckles[16 ].
Fig. 9. (Color online) Schematic of MGH preparation and 3D view of the transparent photodetector, photoresponsivity and time-resolved photoresponse of photodetectors under different illuminations[38 ].
|
Table 1. Elements in the magenta boxes coordinated with polymer to form a stable complex. The elements shown in red font were bound with the polymer in the previous reports.
|
Table 2. Summary of various metal elements binded by polymers.
|
Table 3. Comparison of three different methods to synthesize MoS2 and MoS2 field-effect transistor structures.

Set citation alerts for the article
Please enter your email address




