Wenzhen Zou, Chu Zhang, Hongmin Jiang, Liguo Gao, Meiqiang Fan, Tingli Ma. Application of Transition Metal Doping in Perovskite Photovoltaic Devices[J]. Laser & Optoelectronics Progress, 2023, 60(9): 0900002
Search by keywords or author
- Laser & Optoelectronics Progress
- Vol. 60, Issue 9, 0900002 (2023)
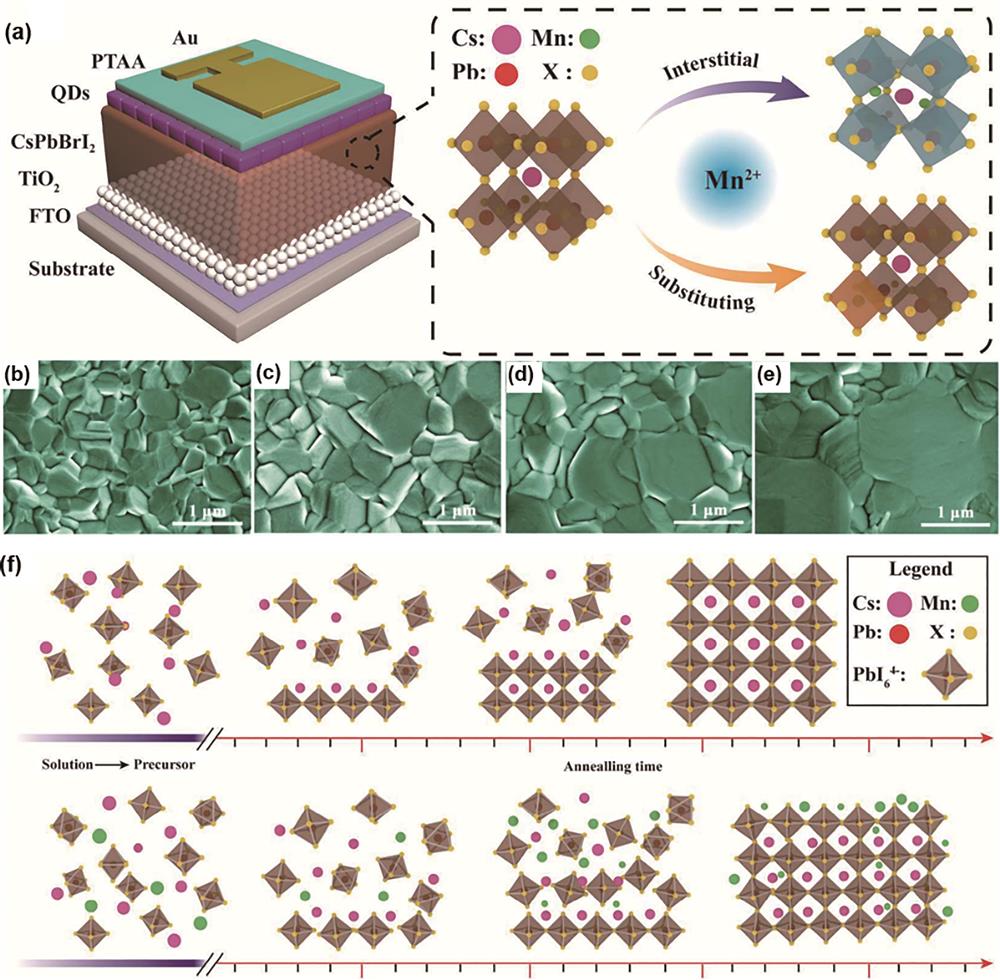
![Mn-doped CsPbI2Br[28]. (a) Schematic structure of device and description of Mn2+ doping modes: interstitial and substitution; scanning electron microscope (SEM) images of (b) original chlorinated paraffins and (c) 0.5%, (d) 1%, and (e) 2% MnCl2; (f) schematic diagram of grain growth driven by surface passivated MnCl2](/richHtml/lop/2023/60/9/0900002/img_01.jpg)
Fig. 1. Mn-doped CsPbI2Br[28]. (a) Schematic structure of device and description of Mn2+ doping modes: interstitial and substitution; scanning electron microscope (SEM) images of (b) original chlorinated paraffins and (c) 0.5%, (d) 1%, and (e) 2% MnCl2; (f) schematic diagram of grain growth driven by surface passivated MnCl2
![Mn-dopded CsPbI3[29]. (a) Structure and crystal junction diagram of Mn-doped CsPbI3 perovskite solar cells; (b) X-ray diffraction (XRD) patterns of Mn-doped PVK films; (c) partial XRD diffraction peak amplified at 14.36°; (d) energy dispersing X-ray spectroscopy (EDS) for 2% Mn-doped perovskite thin films; (e)~(g) X-ray photoelectron spectroscopy (XPS) for Cs, Pb and I in control film and 2% Mn-doped PVK film; (h) optimal current density and voltage (J-V) characteristic curves of PSC based on Mn-doped CsPbI3 in forward and reverse measurements](/richHtml/lop/2023/60/9/0900002/img_02.jpg)
Fig. 2. Mn-dopded CsPbI3[29]. (a) Structure and crystal junction diagram of Mn-doped CsPbI3 perovskite solar cells; (b) X-ray diffraction (XRD) patterns of Mn-doped PVK films; (c) partial XRD diffraction peak amplified at 14.36°; (d) energy dispersing X-ray spectroscopy (EDS) for 2% Mn-doped perovskite thin films; (e)~(g) X-ray photoelectron spectroscopy (XPS) for Cs, Pb and I in control film and 2% Mn-doped PVK film; (h) optimal current density and voltage (J-V) characteristic curves of PSC based on Mn-doped CsPbI3 in forward and reverse measurements
Fig. 3. Influence of Ag ion doping on PVK[33]. (a) Layout diagram of planar junction solar cell; (b) photovoltaic performance of MAB0.1Pb0.9I3 system with different B elements; (c) UV spectra curves of MAPb1-xHgxI3 films at different Hg2+ doping concentrations; (d) PCE box diagram of solar cells based on MAPb1-xHgxI3; (e) SEM images of perovskite films with different Hg2+ doping concentrations
Fig. 4. Influence of Co ion doping on PVK[34]. (a) Transmission electron microscope (TEM) cross-section image of MA(31Pb∶1Co)I3 thin film; (b)~(d) composite elemental maps performed with energy dispersive X-Ray spectroscopy (EDX) in scanning transmission electron microscopy (STEM) mode. Individual elemental maps of (b) Pb, (c) I, and (e) Co indicate that elements are distributed homogeneously throughout film thickness; (f) SEM images of MAPbI3 and MA(Pb∶Co)I3 thin films; (g) XPS images of MAPbI3 and MA(31Pb∶1Co)I3 thin films; (h) forward and reverse J-V diagrams of solar cell measurements, and average performance of optimized MA(Pb∶Co)I3 solar cells; (i) energy level diagram of MA (Pb∶Co)I3 relative to MAPbI3
Fig. 5. Influence of Cd ion doping on PVK[31]. (a) Lattice relaxation mechanism. a-c are schematic diagram illustrating a local strain, which can be reduced by forming b point defects or c introducing small ions. d is schematic diagram shows strain in (002) plane, which is reduced by introduction of small B/X ions. e is B/X covalent candidate ions; (b) top view SEM images of perovskite thin films on TiO2/ITO substrates and cross-sectional SEM images of perovskite solar cells; (c) PCE statistics for 30 PSCs of each component; (d) evolution of PCE in solar cells as six devices age in ambient air (50 per cent relative humidity); (e) in air environment and (f) in nitrogen environment, unpackaged power factor correction circuit is operated under maximum power point conditions using an UV filter with cut-off wavelength of 420 nm
Fig. 6. Nb ion and Ta ion doping in TiO2[54]. (a) Conduction band diagram of dopant regulated battery; (b) energy graph image of Nb-TiO2 thin film with respect to vacuum level calculated based on UV-VIS spectrum and uninterruptible power source measurement results; (c) top view a and cross section image b of TiO2 films prepared by SEM, and top view of Nb-TiO2 thin films containing 1%, 3%, 5%, 7%, 10%, and 20% mole fraction Nb atoms as shown by c-h; (d) XRD patterns of undoped and doped TiO2 nanowire arrays with different Ta doping levels; (e) a and c are undoped TiO2 nanowire arrays, b and d are TEM and high-resolution transmission electron microscopy (HRTEM) images of 0.1-Ta-TiO2 nanowire arrays; Illustrations in c and d are corresponding fast Fourier transform (FFT) diffraction patterns
Fig. 7. Co ion doping in TiO2[61]. (a) MOF preparation process and perovskite solar cell structure; (b) J-V curves of solar cells with best performance using dye-sol TiO2 and co-doped TiO2 (mass fraction is 1%); (c) SEM images of dye-sol TiO2 and 1% mass fraction co-doped TiO2 on FTO coated glass; (d) EIS curves based on TiO2 and 1% mass fraction co-doped TiO2 thin films
Fig. 8. Ni ion doping in TiO2[66]. (a) Schematic diagram of solution treatment method for preparing Ni-TiO2 thin film; (b) schematic diagram of carbon base plane PSC structure and (c) high resolution cross-section SEM image; (d) energy level diagram; (e) steady-state luminescence spectra of ore on different substrates
Fig. 9. Co ion doping in SiO2[73]. (a) Band diagrams of SnO2, SnO2-CoCl2, and PVK layers and J-V curves of prepared PSCs; (b) cross-sectional SEM images of PSC based on SnO2-CoCl2 at a scale of 500 nm; (c) PSCs PCE histogram based on SnO2 and SnO2-CoCl2; (d) XPS image of Co 2p; XPS image of O 1s for (e) pristine SnO2 and (f) SnO2-CoCl2
Fig. 10. Pd ion doping in HTL[84]. (a) Schematic diagram of PSCs with HTL layer doped Pd nanosheets; (b) TEM image of Pd nanosheets; (c) energy level diagram of materials used in PSCs; (d) schematic of interface of PVK and HTM layers; (e) comparison of PCE distributions of 30 individual devices; (f) EIS plots of devices in dark at 0.8 mV forward bias voltage
Fig. 11. Ag ion doping in NiOx[94]. (a) Device structure; (b) SEM image of cross section of complete solar cell device and (c) Ag-doped lattice structure diagram; (d) Ag 3d XPS image of pristine NiOx thin film and Ag∶NiOx thin film; (e) UV-VIS absorption spectra and (f) time-resolved PL spectra of MAPbI3 thin films grown on pristine NiOx and Ag∶NiOx thin films; (g) J-V curves with reverse direction and (h) IPCE spectra of best performed device based PEDOT∶PSS, NiOx, and Ag∶NiOx as HTLs; (i) normalized PCE of PSCs based on PEDOT∶PSS, pristine NiOx, and Ag∶NiOx HTL as a function of storage time in ambient environment (30±2% humidity, T = 25 °C)
Fig. 12. Cr ion doping in NiOx[97]. (a) Cr/CuGaO2-CC/NiOx HTL device structure and (b) its SEM image; (c) PSC device energy level diagram; (d) SEM images of perovskite on NiOx and Cr/CuGaO2-CC/NiOx HTL, respectively; (e) PL spectrograms of perovskites on NiOx and Cr/CuGaO2-CC/NiOx HTL
|
Table 1. Doping situation of transition metal in each layer of PSCs
Set citation alerts for the article
Please enter your email address



