- Journal of Semiconductors
- Vol. 43, Issue 2, 021701 (2022)
Abstract
1. Introduction
With the rapid development of the world economy and the increase of the population, the energy crisis and environmental pollution have gradually become the key challenges for the sustainable development of mankind. As a powerful and sustainable strategy, photocatalysis has been regarded as one of the most promising energy conversion processes to utilize green and inexhaustible solar energy for exploiting ideal fuels and simultaneously promoting environmental remediation[
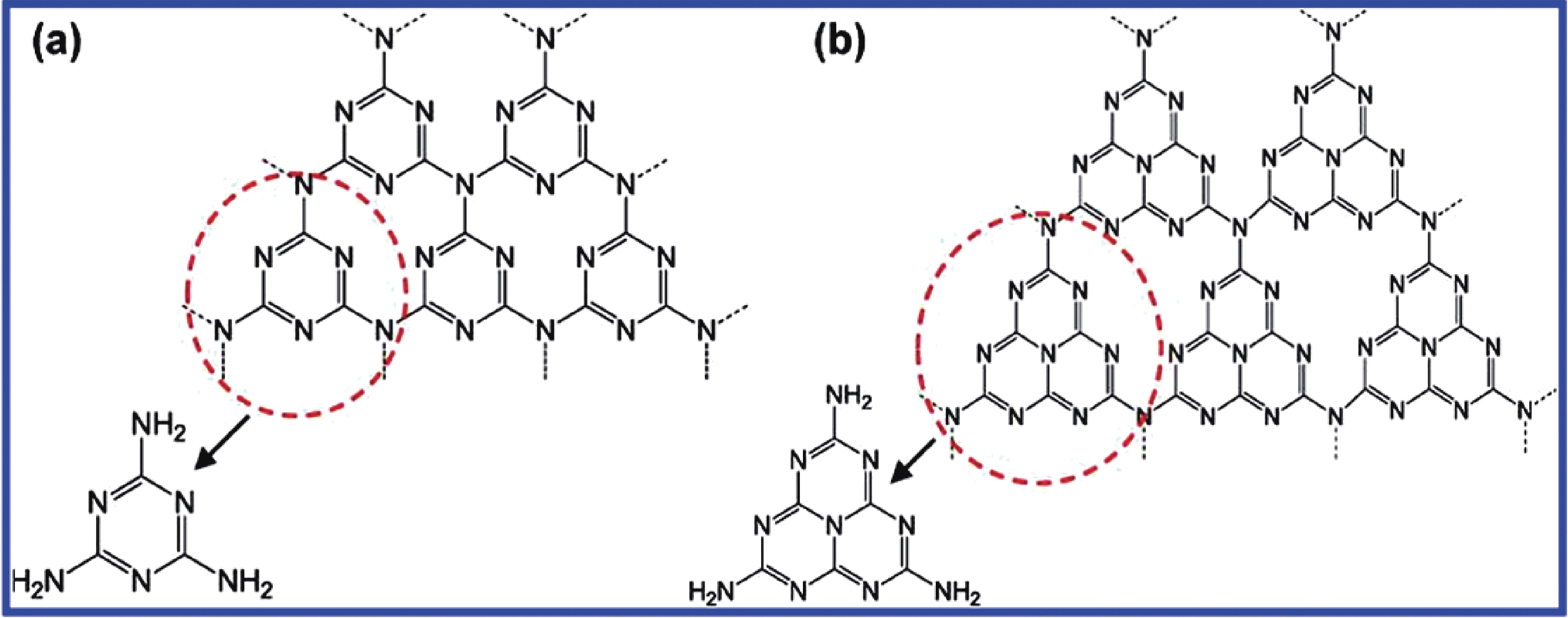
In the development of robust visible-light-responsive photocatalysts for utilizing solar energy, graphitic carbon nitride (g-C3N4), a metal-free polymeric semiconductor with triazine or heptazine as a basic structural unit (Fig. 1)[
![]()
Figure 1.(Color online) The schematic structure of triazine (a) and tri-s-triazine (heptazine) (b) in g-C3N4. Reprinted from Ref. [
Up to now, although many interesting reviews on the strategies to improve the photocatalytic property of g-C3N4 have been reported, a crucial review focusing on the electronic structure tuning of g-C3N4 is lacking to provide the researchers with a panorama of the latest advances in this field. Herein, we present a comprehensive and updated review on the most recent progress in the electronic structure tuning of g-C3N4 for highly efficient photocatalysis. The diverse design and regulation strategies for optimizing the electronic structure of g-C3N4 are elaborated on and the effect of the electronic structure tuning on the properties and photocatalytic activities of g-C3N4 are also summarized. Moreover, this review will present some novel insights on the crucial challenges, opportunities, and inspiring perspectives for the design of highly efficient g-C3N4-based photocatalysts, which should be a guide for future research in this hot area.
2. Role of electronic structure modulation in optimizing the photocatalytic activity of g-C3N4
It is a well-known fact that modulating the electronic structure of g-C3N4 serves as a key strategy to optimize not only light absorption ability but also the redox reaction kinetics. Precisely, according to semiconductor theory, a semiconductor material can only absorb the photons whose energy is equal to or greater than its bandgap energy. As shown in Fig. 2, when it was used as a photocatalyst and exposed to sunlight, the electrons can be excited to the higher conduction band (CB) and participate in reduction reactions, resulting in the formation of holes in the valence band (VB) where they can take part in oxidation reactions. Usually, if a semiconductor has a large bandgap energy (>2.8 eV), it can only absorb solar light in the ultraviolet (UV) region (λ < 420 nm), which is only about 5% of the total solar spectrum. While if a semiconductor material has a small bandgap energy (0 < Eg < 2.8 eV), its absorbable spectrum can reach the visible region (420 < λ < 780 nm) or near-infra-red (NIR) region (780 < λ < 2500 nm), which accounts for about 43% and 52% of the total solar spectrum, respectively [
![]()
Figure 2.(Color online) The process of overall solar water splitting over a semiconductor photocatalyst.
3. Strategies of electronic structure tuning
The photocatalytic properties of materials are directly related to their electronic structures, which determine the spectral response properties and chemical kinetics of photocatalysts. In recent years, with the development of materials science and engineering, electronic structure tuning has received a lot of attention, especially in the field of photocatalysis. Ever since it has been used as a photocatalyst, tremendous effort has been devoted to designing and optimizing the photocatalytic performances of g-C3N4. The approaches, including vacancy modification, doping, crystallinity modulation and synthesis of new molecular structure, are the main strategies to tune the electronic structure of g-C3N4.
3.1. Vacancy modification
Recent studies found that defect engineering by the introduction of carbon and/or nitrogen vacancy defects into the motif of g-C3N4 has a pronounced effect on its electronic structure and photocatalytic performance, which is mainly due to that the VB and CB of g-C3N4 are composed of nitrogen PZ orbitals and carbon PZ orbitals, respectively[
In order to investigate the effect of band structure changing, induced by vacancy defects on light absorption of g-C3N4, Yang and coworkers synthesized a N-vacancy-rich g-C3N4 through a hard template method with a high pressure and high temperature (HPHT) technique. The measurements showed that the bandgap energy of the resulted sample was significantly narrowed and reduced from 2.97 to 1.88 eV due to the up-shifted VB and down-shifted CB edge potential. As a result, the light-responding range of N-vacancy-mediated g-C3N4 was greatly extended and showed a maximum absorption of 660 nm[
![]()
Figure 3.(Color online) (a) The UV–vis absorption spectra, (b) converted Kubelka–Munk vs. light energy plots and (c) XPS valence band spectra of CN and CNQs. (d) The schematic band structures of CN and CNQ 680. Reprinted from Ref. [
Generally, the C or N defects can be successfully introduced into the framework when the bulk g-C3N4 is thermally treated under H2, Ar, NH3 or N2 atmosphere[
To meet this challenge, many new defect engineering strategies that can control the concentration of the defect have been developed. As shown in Fig. 4, our group has designed a novel and facile urea and KOH-assisted thermal polymerization (UKATP) strategy for the preparation of defect-modified thin-layered and porous g-C3N4 (DTLP-CN), wherein the thickness of g-C3N4 was dramatically decreased, and nitrogen vacancies, cyano groups and mesopores were simultaneously introduced. Especially, the roles of N defects and structures of g-C3N4 can be targeted, regulated and optimized by changing the mass ratio of precursors. Consequently, the band structure and charge carrier transportation of g-C3N4 were well-optimized, and the photocatalytic hydrogen evolution performance of the DTLP-CN was significantly improved more than 48.5 times with the average apparent quantum yield (AQY) of as high as 0.82% at 500 nm[
![]()
Figure 4.(Color online) Schematic illustration of synthesis methods of DTLP-CN via thermal polymerization of melamine, urea, and KOH. Reprinted from Ref. [
3.2. Doping
Owing to regulating the physical-chemical property of semiconductors effectively, doping is widely used to optimize the photocatalysts for efficient solar energy conversion. Recently, doping has been proven as a valid method to regulate the electronic structure of g-C3N4, which can be facilely realized by incorporating foreign elements or a structure-matching organic group into the framework of g-C3N4[
Many non-metal elements, such as O, B, P, S, F, Br and I, have been incorporated into the motif of g-C3N4[
![]()
Figure 5.(Color online) (a) Schematic structure of the O-doped g-C3N4-based photocatalyst. (b) Band structure diagrams of g-C3N4 and O-doped g-C3N4. (c) Schematic of the fabrication of BDCNN originated from CNN and (d) the charge-transfer process in BDCNN-based heterojunction upon light irradiation. Reprinted from Refs. [
Similar to the non-metal doping, the introduction of metal atoms such as Na, K, Fe, Co, Cu, Mn, Zn, Ni, and Ag into the framework of g-C3N4 is also a successful strategy to modulate the band structure, extend the light absorption and improve the photocatalytic performance of g-C3N4[
![]()
Figure 6.(Color online) (a) UV–vis diffuse reflectance spectra, (b) the band gap from (
From the above studies, we can conclude that the electronic structure and photocatalytic properties of g-C3N4 can be modulated by doping of non-metal or metal elements, however, there are still many problems to be solved, such as the presence of surface trapping center, doping site, lower oxidizing and reducing ability. Additionally, many defects may originate from the excessive doping of metals and non-metals, which may reduce the separation performance of charge carriers because of doping asymmetry[
3.3. Crystallinity modulation
The performances of g-C3N4, such as band structure, charge carrier migration, absorptivity and photoelectronic characteristics, can be effectively optimized by regulating its crystallinity, thus, the degree of crystallinity is closely related to the photoelectrochemical property of semiconductor[
In order to understand the effect of crystallinity on the electronic structure and photocatalytic performance of g-C3N4, Zhang and coworkers proposed a facile ion thermal strategy to enhance the crystallinity of g-C3N4 where most of the photons can be used to drive photocatalytic reactions. The measurements demonstrated that this thermal condensation in the molten salt method can result in highly crystalline g-C3N4 with a maximum π–π layer stacking distance of 0.292 nm. Moreover, the band gap can be regulated from 2.74 to 2.56 eV by the addition of oxamide. As a result, the visible-light response range of CN-OA-m is extended to 700 nm and its H2 evolution activity is dramatically improved[
![]()
Figure 7.(Color online) (a) UV–visible diffuse reflectance spectrum (DRS) and (b) HOMO and LUMO positions of CN, CN-LiNa, CN-NaK, and CN-LiK. (c) UV–vis DRS and (b) bandgap structures for CN, crystalline CN, CCN and crystalline CCN. Reprinted from Refs. [
3.4. Development of new molecular structure
Carbon nitride is usually composed of triazine building units, which forms melon or crystallinity-modulated poly triazine imide (PTI) and poly heptazine imide (PHI)[
![]()
Figure 8.(Color online) Schematic illustrations of basic structural units of polymeric carbon nitride with different C and N stoichiometric ratios: (a) triazine-based graphitic carbon nitride, (b) heptazine-based graphitic carbon nitride, (c, d) polymeric C3N5, (e) C3N6, (f) C3N7, and (g) C3N3. Reprinted from Ref. [
Recently, C4N has attracted significant attention as a new class of low-band-gap polymeric carbon nitride owning to its excellent physiochemical properties for efficient solar energy conversions. Li and coworkers firstly fabricated the exfoliated C4N nanosheets for highly efficient oxygen reduction via a top-down method. The obtained C4N demonstrated a small band gap of 1.41 eV and an extended absorption band at around 500 nm due to n→π* transition[
![]()
Figure 9.(Color online) (a) Synthesis scheme of C3N5. (b) UV–Vis DRS for C3N5 compared with bulk g-C3N4. (c) Steady-state PL spectra of melem, g-C3N4 and C3N5. Reprinted from Ref. [
4. Conclusions and perspectives
In this review, the strategies and recent progresses in the electronic structure tuning of g-C3N4 for highly efficient photocatalysis are summarized, which is critical for highly efficient solar energy conversion, such as water splitting, organism degradation, and CO2 reduction, etc. Specifically, the regulation strategies based on vacancy modification, doping, crystallinity modulation and molecular structure construction are elaborated in detail. Thus, with the rational designing and modifying of the electronic band structure of g-C3N4, the light harvesting, charge separation and photocatalytic properties of g-C3N4 would be dramatically enhanced. This review provides a multi-angle cognition to cater to actual production demand in the field of solar energy conversion.
Despite plenty of progress on electronic structure having been made, there are some trends or challenges of pivotal issues, which are elaborated on in the following:
(1) As summarized above, various strategies have been demonstrated to tune the electronic band structure of g-C3N4 so as to improve light harvesting and accelerate photo-generated carriers transfer kinetics for enhanced photocatalysis. Nevertheless, the approaches that synergistically use different regulatory strategies to precisely regulate the different features of g-C3N4 need to be researched further.
(2) With the development of g-C3N4 research, the strategies that combine the regulation of electronic structure with the construction of g-C3N4-based heterostructure have attracted great attention. However, the synergetic effect and interaction mechanism of the different components have not been clearly elucidated. Therefore, much attention on new experimental methods and theoretical calculations should be paid to clarify these problems.
(3) Although it is believed that g-C3N4 has strong chemical and thermal stability, the existing photocatalytic decays are often ignored when the electronic structure is modulated. Therefore, the stability of modified g-C3N4 should be of concern for future research.
Acknowledgements
This work was mostly supported by the National Natural Science Foundation of China (Nos. 21975245, 51972300, 61674141, 12004094, and 21976049), the Key Research Program of Frontier Science, CAS (QYZDB-SSW-SLH006), the National Key Research and Development Program of China (Nos. 2017YFA0206600 and 2018YFE0204000), the Strategic Priority Research Program of Chinese Academy of Sciences (XDB43000000), the Natural Science Foundation of Hebei Province (F2019402063), the Youth Foundation of Hebei Province Department of Education (QN2019326), the Science and Technology Research and Development Program of Handan city (21422111246), and the Key Project of Handan University (2018101). K.L. acknowledges the support from the Youth Innovation Promotion Association, Chinese Academy of Sciences (2020114). Y. H. also acknowledges the support from the Doctoral Special Fund Project of Hebei University of Engineering.
References
[1] L Ai, R Shi, J Yang et al. Efficient combination of g-C3N4 and CDs for enhanced photocatalytic performance: a review of synthesis, strategies, and applications. Small, 17, 2007523(2021).
[2] J Y Y Loh, N P Kherani, G A Ozin. Persistent CO2 photocatalysis for solar fuels in the dark. Nat Sustain, 4, 466(2021).
[3] M S Nasir, G Yang, I Ayub et al. Recent development in graphitic carbon nitride based photocatalysis for hydrogen generation. Appl Catal B, 257, 117855(2019).
[4] Y Huang, J Liu, Y Deng et al. The application of perovskite materials in solar water splitting. J Semicond, 41, 011701(2020).
[5] M Ma, Y Huang, J Liu et al. Engineering the photoelectrochemical behaviors of ZnO for efficient solar water splitting. J Semicond, 41, 091702(2020).
[6] Y Yang, H Tan, B Cheng et al. Near-infrared-responsive photocatalysts. Small Methods, 5, 2001042(2021).
[7] J Zhang, J Cui, S Eslava. Oxygen evolution catalysts at transition metal oxide photoanodes: their differing roles for solar water splitting. Adv Energy Mater, 11, 2003111(2021).
[8] Y Huang, J Liu, D Cao et al. Separation of hot electrons and holes in Au/LaFeO3 to boost the photocatalytic activities both for water reduction and oxidation. Int J Hydrogen Energy, 44, 13242(2019).
[9] G Liao, Y Gong, L Zhang et al. Semiconductor polymeric graphitic carbon nitride photocatalysts: the “holy grail” for the photocatalytic hydrogen evolution reaction under visible light. Energy Environ Sci, 12, 2080(2019).
[10] W J Ong, L L Tan, Y H Ng et al. Graphitic carbon nitride (g-C3N4)-based photocatalysts for artificial photosynthesis and environmental remediation: are we a step closer to achieving sustainability. Chem Rev, 116, 7159(2016).
[11] X Wang, K Maeda, A Thomas et al. A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat Mater, 8, 76(2009).
[12] A Savateev, I Ghosh, B König et al. Photoredox catalytic organic transformations using heterogeneous carbon nitrides. Angew Chem Int Ed, 57, 15936(2018).
[13] L Lin, Z Yu, X Wang. Crystalline carbon nitride semiconductors for photocatalytic water splitting. Angew Chem Int Ed, 58, 6164(2019).
[14] V W H Lau, B V Lotsch. A Tour-guide through carbon nitride-land: structure- and dimensionality-dependent properties for photo(electro)chemical energy conversion and storage. Adv Energy Mater, 2101078(2021).
[15] H Che, G Che, P Zhou et al. Nitrogen doped carbon ribbons modified g-C3N4 for markedly enhanced photocatalytic H2-production in visible to near-infrared region. Chem Eng J, 382, 122870(2019).
[16] A Kumar, P Raizada, A Hosseini-Bandegharaei et al. C-, N-Vacancy defect engineered polymeric carbon nitride towards photocatalysis: viewpoints and challenges. J Mater Chem A, 9, 111(2021).
[17] H Yu, R Shi, Y Zhao et al. Alkali-assisted synthesis of nitrogen deficient graphitic carbon nitride with tunable band structures for efficient visible-light-driven hydrogen evolution. Adv Mater, 29, 1605148(2017).
[18] X Pan, M Q Yang, X Fu et al. Defective TiO2 with oxygen vacancies: synthesis, properties and photocatalytic applications. Nanoscale, 5, 3601(2013).
[19] P Yang, H Zhuzhang, R Wang et al. Carbon vacancies in a melon polymeric matrix promote photocatalytic carbon dioxide conversion. Angew Chem Int Ed, 58, 1134(2018).
[20] F Li, X Yue, D Zhang et al. Targeted regulation of exciton dissociation in graphitic carbon nitride by vacancy modification for efficient photocatalytic CO2 reduction. Appl Catal B, 292, 120179(2021).
[21] Z Yang, D Chu, G Jia et al. Significantly narrowed bandgap and enhanced charge separation in porous, nitrogen-vacancy red g-C3N4 for visible light photocatalytic H2 production. Appl Surf Sci, 504, 144407(2020).
[22] P Niu, M Qiao, Y Li et al. Distinctive defects engineering in graphitic carbon nitride for greatly extended visible light photocatalytic hydrogen evolution. Nano Energy, 44, 73(2018).
[23] P Niu, L C Yin, Y Q Yang et al. Increasing the visible light absorption of graphitic carbon nitride (melon) photocatalysts by homogeneous self-modification with nitrogen vacancies. Adv Mater, 26, 8046(2014).
[24] P Zhou, F Lv, N Li et al. Strengthening reactive metal-support interaction to stabilize high-density Pt single atoms on electron-deficient g-C3N4 for boosting photocatalytic H2 production. Nano Energy, 56, 127(2019).
[25] Y Huang, J Liu, C Zhao et al. Facile synthesis of defect-modified thin-layered and porous g-C3N4 with synergetic improvement for photocatalytic H2 production. ACS Appl Mater Interfaces, 12, 52603(2020).
[26] L Duan, G Li, S Zhang et al. Preparation of S-doped g-C3N4 with C vacancies using the desulfurized waste liquid extracting salt and its application for NO
[27] D Zhang, Y Guo, Z Zhao. Porous defect-modified graphitic carbon nitride via a facile one-step approach with significantly enhanced photocatalytic hydrogen evolution under visible light irradiation. Appl Catal B, 226, 1(2018).
[28] P Hu, C Chen, R Zeng et al. Facile synthesis of bimodal porous graphitic carbon nitride nanosheets as efficient photocatalysts for hydrogen evolution. Nano Energy, 50, 376(2018).
[29] X Wang, J Meng, X Zhang et al. Controllable approach to carbon-deficient and oxygen-doped graphitic carbon nitride: robust photocatalyst against recalcitrant organic pollutants and the mechanism insight. Adv Funct Mater, 31, 2010763(2021).
[30] Y Zhou, L Zhang, W Wang. Direct functionalization of methane into ethanol over copper modified polymeric carbon nitride via photocatalysis. Nat Commun, 10, 506(2019).
[31] Y Wang, S Z F Phua, G Dong et al. Structure tuning of polymeric carbon nitride for solar energy conversion: from nano to molecular scale. Chem, 5, 2775(2019).
[32] S Cao, J Low, J Yu et al. Polymeric photocatalysts based on graphitic carbon nitride. Adv Mater, 27, 2150(2015).
[33] Z Zhou, Y Zhang, Y Shen et al. Molecular engineering of polymeric carbon nitride: advancing applications from photocatalysis to biosensing and more. Chem Soc Rev, 47, 2298(2018).
[34] Z Lin, X Wang. Nanostructure engineering and doping of conjugated carbon nitride semiconductors for hydrogen photosynthesis. Angew Chem Int Ed, 52, 1735(2013).
[35] Z A Lan, G Zhang, X Wang. A facile synthesis of Br-modified g-C3N4 semiconductors for photoredox water splitting. Appl Catal B, 192, 116(2016).
[36] Y Deng, J Liu, Y Huang et al. Engineering the photocatalytic behaviors of g/C3N4-based metal-free materials for degradation of a representative antibiotic. Adv Funct Mater, 30, 2002353(2020).
[37] J Ran, T Y Ma, G Gao et al. Porous P-doped graphitic carbon nitride nanosheets for synergistically enhanced visible-light photocatalytic H2 production. Energy Environ Sci, 8, 3708(2015).
[38] D Zhao, C L Dong, B Wang et al. Synergy of dopants and defects in graphitic carbon nitride with exceptionally modulated band structures for efficient photocatalytic oxygen evolution. Adv Mater, 31, 1903545(2019).
[39] C Feng, L Tang, Y Deng et al. Synthesis of leaf-vein-like g-C3N4 with tunable band structures and charge transfer properties for selective photocatalytic H2O2 evolution. Adv Funct Mater, 30, 2001922(2020).
[40] D Zhao, Y Wang, C L Dong et al. Boron-doped nitrogen-deficient carbon nitride-based Z-scheme heterostructures for photocatalytic overall water splitting. Nat Energy, 6, 388(2021).
[41] T Xiong, W Cen, Y Zhang et al. Bridging the g-C3N4 interlayers for enhanced photocatalysis. ACS Catal, 6, 2462(2016).
[42] M Zhang, X Bai, D Liu et al. Enhanced catalytic activity of potassium-doped graphitic carbon nitride induced by lower valence position. Appl Catal B, 164, 77(2015).
[43] S Hu, X Chen, Q Li et al. Fe3+ doping promoted N2 photofixation ability of honeycombed graphitic carbon nitride: The experimental and density functional theory simulation analysis. Appl Catal B, 201, 58(2017).
[44] Z Li, C Kong, G Lu. Visible photocatalytic water splitting and photocatalytic two-electron oxygen formation over Cu- and Fe-doped g-C3N4. J Phys Chem C, 120, 56(2016).
[45] W Yan, L Yan, C Jing. Impact of doped metals on urea-derived g-C3N4 for photocatalytic degradation of antibiotics: Structure, photoactivity and degradation mechanisms. Appl Catal B, 244, 475(2019).
[46] Z Ding, X Chen, M Antonietti et al. Synthesis of transition metal-modified carbon nitride polymers for selective hydrocarbon oxidation. ChemSusChem, 4, 274(2011).
[47] S Cao, Q Huang, B Zhu et al. Trace-level phosphorus and sodium co-doping of g-C3N4 for enhanced photocatalytic H2 production. J Power Sources, 351, 151(2017).
[48] G Dong, K Zhao, L Zhang. Carbon self-doping induced high electronic conductivity and photoreactivity of g-C3N4. Chem Commun, 48, 6178(2018).
[49] S Hu, J Zhu, L Wu et al. Effect of fluorination on photocatalytic degradation of rhodamine B over In(OH)
[50] L Lin, Z Lin, J Zhang et al. Molecular-level insights on the reactive facet of carbon nitride single crystals photocatalysing overall water splitting. Nat Catal, 3, 649(2020).
[51] G Zhang, G Li, Z A Lan et al. Optimizing optical absorption, exciton dissociation, and charge transfer of a polymeric carbon nitride with ultrahigh solar hydrogen production activity. Angew Chem Int Ed, 56, 13445(2017).
[52] G Zhang, L Lin, G Li et al. Ionothermal synthesis of triazine–heptazine-based copolymers with apparent quantum yields of 60 % at 420 nm for solar hydrogen production from “Sea Water”. Angew Chem Int Ed, 57, 9372(2018).
[53] Y Xu, X He, H Zhong et al. Solid salt confinement effect: An effective strategy to fabricate high crystalline polymer carbon nitride for enhanced photocatalytic hydrogen evolution. Appl Catal B, 246, 349(2019).
[54] J Yuan, Y Tang, X Yi et al. Crystallization, cyanamide defect and ion induction of carbon nitride: Exciton polarization dissociation, charge transfer and surface electron density for enhanced hydrogen evolution. Appl Catal B, 251, 206(2019).
[55] L Lin, W Ren, C Wang et al. Crystalline carbon nitride semiconductors prepared at different temperatures for photocatalytic hydrogen production. Appl Catal B, 231, 234(2018).
[56] D Vidyasagar, T Bhoyar, G Singh et al. Recent progress in polymorphs of carbon nitride: synthesis, properties, and their applications, macromol. Rapid Commun, 42, 2000676(2021).
[57] S Kumar, V R Battula, K Kailasam. Single molecular precursors for CxNy materials- Blending of carbon and nitrogen beyond g-C3N4. Carbon, 183, 332(2021).
[58] J Mahmood, E K Lee, M Jung et al. Nitrogenated holey two-dimensional structures. Nat Commun, 6, 6486(2015).
[59] J Xu, J Mahmood, Y Dou et al. 2D frameworks of C2N and C3N as new anode materials for lithium-ion batteries. Adv Mater, 29, 1702007(2017).
[60] Z Fang, Y Li, J Li et al. Capturing visible light in low-band-gap C4N-derived responsive bifunctional air electrodes for solar energy conversion and storage. Angew Chem Int Ed, 60, 17615(2021).
[61] P Kumar, E Vahidzadeh, U K Thakur et al. C3N5: A low bandgap semiconductor containing an azo-linked carbon nitride framework for photocatalytic, photovoltaic and adsorbent applications. J Am Chem Soc, 141, 5415(2019).
[62] S N Talapaneni, G P Mane, D H Park et al. Diaminotetrazine based mesoporous C3N6 with a well-ordered 3D cubic structure and its excellent photocatalytic performance for hydrogen evolution. J Mater Chem A, 5, 18183(2017).
[63] Y Li, C Mo, J Li et al. Pyrazine–nitrogen–rich exfoliated C4N nanosheets as efficient metal–free polymeric catalysts for oxygen reduction reaction. J Energy Chem, 49, 243(2020).
[64] J Zhang, B Jing, Z Tang et al. Experimental and DFT insights into the visible-light driving metal-free C3N5 activated persulfate system for efficient water purification. Appl Catal B, 289, 120023(2021).
[65] J Mahmood, E K Lee, M Jung et al. Two-dimensional polyaniline (C3N) from carbonized organic single crystals in solid state. PNAS, 113, 7414(2016).
Set citation alerts for the article
Please enter your email address



