Zhe Feng, Jun Qian. Advances on in vivo Fluorescence Bioimaging in the Second Near-Infrared Window[J]. Laser & Optoelectronics Progress, 2022, 59(6): 0617001
Search by keywords or author
- Laser & Optoelectronics Progress
- Vol. 59, Issue 6, 0617001 (2022)
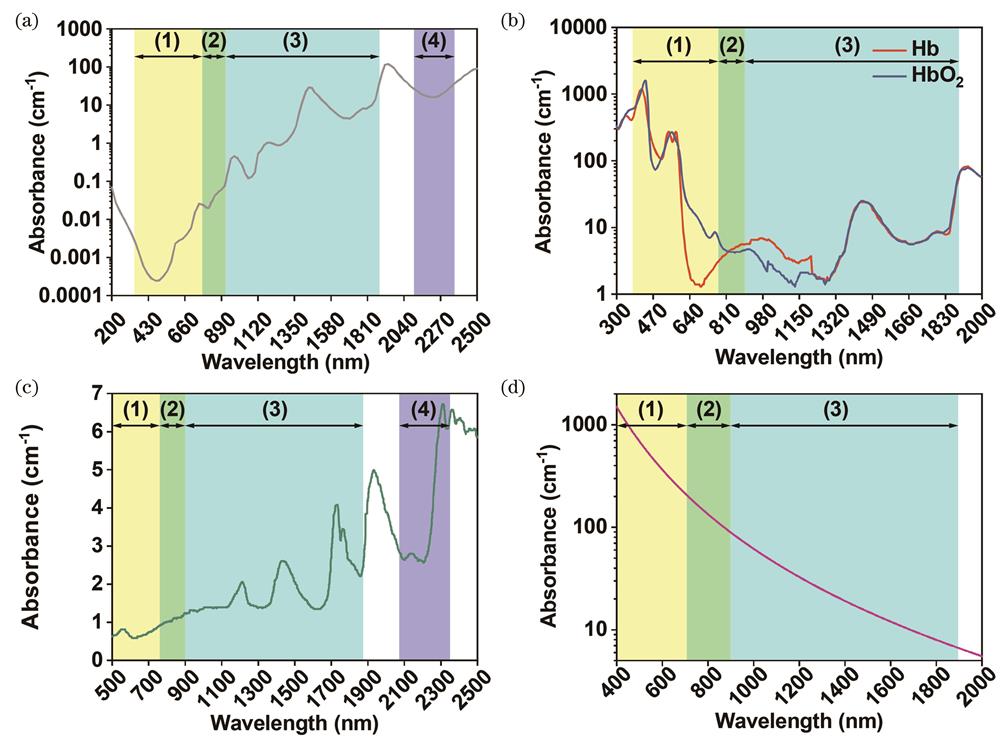
![Light absorption properties of several common biological components in the visible and near-infrared regions. (a) Absorption spectrum of water[13]; (b) absorption spectrum of hemoglobin/oxygenated hemoglobin[14]; (c) absorption spectrum of subcutaneous adipose tissue[15]; (d) absorption spectrum of melanin[16]](/richHtml/lop/2022/59/6/0617001/img_1.jpg)
Fig. 1. Light absorption properties of several common biological components in the visible and near-infrared regions. (a) Absorption spectrum of water[13]; (b) absorption spectrum of hemoglobin/oxygenated hemoglobin[14]; (c) absorption spectrum of subcutaneous adipose tissue[15]; (d) absorption spectrum of melanin[16]
![Light scattering characteristics of typical biological tissues in the visible and near-infrared regions. (a) Scattering spectrum of blood[14]; (b) scattering spectrum of subcutaneous adipose tissue[15]](/richHtml/lop/2022/59/6/0617001/img_2.jpg)
Fig. 2. Light scattering characteristics of typical biological tissues in the visible and near-infrared regions. (a) Scattering spectrum of blood[14]; (b) scattering spectrum of subcutaneous adipose tissue[15]
Fig. 3. Spontaneous fluorescence characteristics of biological tissues in the visible and near-infrared regions[18]. (a) Autofluorescence spectra of liver under the excitation of different wavelength light sources; (b) autofluorescence spectra of liver, spleen, and heart under 808 nm laser excitation; (c) enlarged spectra of Fig.3 (b) in 1300‒1750 nm
Fig. 4. Mechanism of near-infrared optical biological imaging[19]. (a) Schematic of light propagation in bio-tissue; (b) absorption spectrum of water and the imaging windows in the near-infrared region
Fig. 5. Several water-soluble molecular dyes with NIR-II fluorescence. (a) Chemical structure of ICG[22-24]; (b) chemical structure of MB[25]; (c)(d) two NIR-II fluorescent dyes with polymethylene structure[29-30]; (e)(f) two NIR-II fluorescent dyes with D-A-D structure[6, 31]
Fig. 6. Schematic of ACQ and AIE[45]. (a) Fluorescence quenching of fluorescein in the aggregate state due to π-π stacking; (b) emission enhancement of tetraphenylethene (TPE) in the aggregate state due to restriction of intramolecular motion (RIM)
Fig. 7. Excretable AIE fluorophores for deep-penetration fluorescence imaging in non-human primates[51]. (a) Biologically excretable OTPA-BBT dots with bright NIR-II/NIR-IIb emission; (b) NIR-II fluorescence monitoring of excretion in mice after intravenous injection of OTPA-BBT dots, scale bar is 10 mm; (c) deep cerebrovascular microscopy in the marmosets through thinned skull; (d) noninvasive NIR-IIb fluorescence gastrointestinal imaging in the marmosets with high spatial frequency, scale bar is 20 mm
Fig. 8. Two typical organic conjugated polymer fluorophores with bright NIR-II emission. (a) Chemical structure of pDA[60]; (b) chemical structure of L1057 polymer[64]
Fig. 9. Several typical semiconductor quantum dots with near-infrared emission[73]
Fig. 10. Rare-earth doped photoluminescence nanoparticles with near-infrared spectral emission properties. (a) Typical structure of the nanoparticles with NaYF4 Yb∶Ln as core and NaYF4 as shell[90]; (b) emission band of several typical near-infrared rare-earth irons[90]; (c) absorption and emission spectra of several typical near-infrared rare-earth ions[91]
Fig. 11. Typical NIR-II fluorescence in vivo imaging system based on the InGaAs array detector[19]. (a) Macro
Fig. 12. Wide-field NIR-II fluorescence functional in vivo microscopic imaging. (a) NIR-II fluorescence microscopic imaging of tumor vessel, scale bar is 100 µm[50]; (b) ICG-assisted NIR-II fluorescence microscopic cerebral vasculature imaging, scale bar is 100 µm[24]; (c) structural changes of brain blood vessels in one mouse before and after the MCAO model establishment, scale bar is 100 µm[26]; (d) blood flow rate monitoring in the mouse brain, scale bar is 50 µm[50]
Fig. 13. Laser scanning confocal NIR-II fluorescence microscopy. (a) Inverted confocal microscopy imaging system and partial ovarian imaging in mice[119]; (b) upright confocal microscope imaging system and the 3D imaging of mouse brain vessel[118]; (c) multi-direction adjustable upright confocal microscope imaging system suitable for large animals and the 3D imaging of monkey brain vessel[120]
Fig. 14. Two-photon microscopy (2PM) with NIR-II excitation and NIR-II detection[125]. (a) 2PM system with NIR-II point detection and the images of brain vessels, scale bar is 50 μm; (b) 2PM system with NIR-II area detection and the images of brain vessels, scale bar is 50 μm
Fig. 15. Multicolor NIR-II fluorescence functional imaging. (a) Absorption spectra of three fluorescent dyes and the three-color imaging of mice using different excitation wavelengths[129]; (b) images of the three fluorophores using different excitation sources and filters, and the three-color imaging of the mice with blood vessels, lymph nodes, and ureters[132]
Fig. 16. NIR-II photoluminescence imaging system. (a) Time-resolved NIR-II optical imaging system based on the chopper[108]; (b) NIR-II fluorescence lifetime microscopic imaging system based on the TCSPC technique[118]
Fig. 17. Multi-channel NIR-IIb photoluminescence in vivo imaging[7]. (a) Schematic of two-channel NIR-IIb photoluminescence imaging using the PbS/CdS quantum dots and Er3+-doped nanoparticles as probes; (b) two-color NIR-IIb optical imaging of the colon tumor mouse
Fig. 18. NIR-II fluorescence + X multi-channel in vivo imaging. (a) NIR-II fluorescence/photoacoustic two-channel in vivo imaging[142]; (b) NIR-II fluorescence/X-ray CT two-channel in vivo imaging, scale bar is 3.5 mm[145]; (c) NIR-II fluorescence (Ag2S quantum dots labeled)/dual bioluminescence (red firefly luciferase and Gauss luciferase labeled) three-channel in vivo imaging[146]
Fig. 19. Multifunctional diagnosis and treatment platform guided by NIR-II fluorescence imaging. (a) Tumor cell-derived exosome (EXO) assisted photothermal therapy[148]; (b) hypoxia-tolerant photodynamic theranostics of tumor[150]; (c) dual photodynamic and immune therapy of tumor[151]; (d) precious surgery of human liver tumor[152]
Set citation alerts for the article
Please enter your email address



