- Journal of Semiconductors
- Vol. 43, Issue 4, 041104 (2022)
Abstract
1. Introduction
With the climate issues caused by the rapid development of industry and the energy demand brought by the increase in population, people face the problems of energy shortage and global warming. The development of semiconductor photocatalysts has realized these important reaction processes[
At present, semiconductor catalysts have received extensive attention and rapid development because of their green and pollution-free catalytic process and the effective catalytic ability for slow and complex kinetic processes. Nevertheless, there are still some obstacles. For example, photogenerated carriers are difficult to excite, have a high recombination rate and low transfer efficiency, and have low reaction activity on the catalytic surface. To solve these problems, researchers have proposed strategies such as heterojunction engineering[
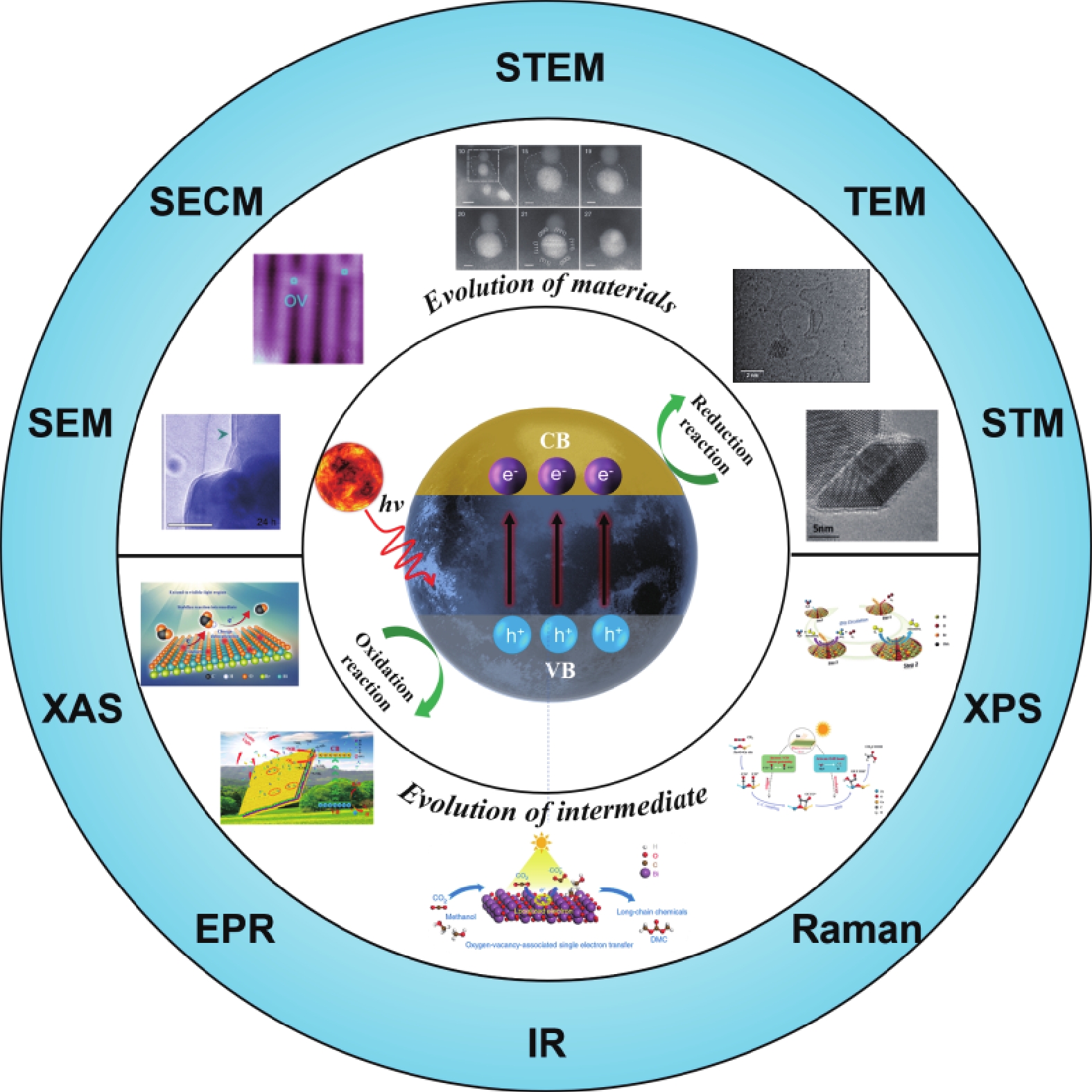
However, most of these studies focus on the changes of materials before and after the reaction, and the changes in the reaction process are not clear, which is of great significance to improve the stability of the catalyst. Therefore, it is necessary to further reveal the dynamic changes of materials and the changes of reaction intermediates at the surface and interface using in-situ or operando characterization methods and equipment, including microscopic imaging and spectral characterization (Fig. 1).
![]()
Figure 1.(Color online)
2. Semiconducting photocatalysis and in-situ characterization techniques
2.1. Principle of photocatalysis
The semiconductor is a kind of material with a unique energy band structure whose conductivity is between the conductor and insulator at room temperature. The special energy band structure of semiconductors consists of the valence band, conduction band, bandgap, and Fermi level (Fig. 2)[
![]()
Figure 2.(Color online) Energy band diagram and electron distribution of three typical types of semiconductors, including (a) intrinsic type, (b) negative-type, and (c) positive-type. (d) Mechanism displays of semiconductor photocatalysis.
A classical semiconductor photocatalytic process will undergo three critical processes (Fig. 2(d)): first, light absorption excites electron–hole pairs; second, the separation of photogenerated electron holes and transfer to the surface and interface of catalyst; third, the occurrence of reduction or oxidation half-reaction at the surface and interface of catalyst. In the photocatalytic process, some critical aspects need to be paid attention to. First, the semiconductor catalysts need to have a suitable energy band structure, including bandgap and energy level. A suitable energy gap can combine high carrier separation efficiency with excellent photocatalytic activity. Semiconductors with energy levels higher than redox potential are more conducive to photocatalytic reaction specifically, the low solar absorption caused by the wide bandgap is not favorable for separating electron–hole pairs. However, the narrow bandgap will produce electrons or holes with low reduction or oxidation potential, resulting in low photocatalytic activity. Therefore, bandgap and activity are two seemingly contradictory. Second, semiconductor catalysts need to have effective separation of photogenerated carriers and efficient surface transport to improve the activity and speed of photocatalytic reaction. Third, the catalysts need to have a relatively stable thermodynamic phase to improve the stability in the reaction processes[
In the process of the photocatalytic reaction, the catalyst material evolution will occur, such as atom migration and in-situ shelling, and a series of reaction intermediates will appear at the surface and interface of the catalyst. These evolutions have an important impact on the activity and stability of the catalytic reaction and the elaboration of the deeper reaction mechanism. Therefore, it needs to be characterized by matching in-situ techniques[
2.2. Characterization techniques to support and demonstrate
Modern advanced characterization techniques have been applied in the study of catalysts[
![]()
Figure 3.(Color online) Schematic diagram of the interaction between particles (electrons and photons) and materials.
X-ray photoelectron spectroscopy (XPS) is a comparatively surface-sensitive (about 10 nm depth) technology[
X-ray absorption spectroscopy (XAS) characterizes the material by measuring the absorption coefficient of X-ray[
Infrared (IR) spectroscopies use infrared light to stimulate materials and probes dipole moment changes by the vibrations and rotations of bonds in molecules, functional groups, and radicals[
Raman spectroscopy is another useful vibrational spectroscopy. This technique utilizes a visible light laser for excitation and measures the energy losses between incident photons and inelastically scattered photons, acquiring polarizability change in a sample under vibrational modes. Raman is very adept at identifying low-frequency modes, which are difficult to detect by traditional methods[
Electron paramagnetic resonance spectroscopy (EPR), also known as electron spin resonance (ESR), is a magnetic resonance technology to directly detect and study unpaired electron paramagnetic[
A transmission electron microscope (TEM) is commonly employed for direct observation of nanomaterials[
A scanning tunneling microscope (STM) uses the quantum tunnel effect to identify the atomic arrangement on the material's surface by measuring the tunneling current between the probe tip and the material surface[
Scanning electrochemical microscopy (SECM) works on electrochemical principles and measures the electrochemical currents given by the oxidation or reduction of substances within a microzone, which is ideal for exploring the heterogeneous catalysis process considering that it combines the spatial resolution of scanned probe microscopy with the in-situ manipulation of electrochemical techniques[
3. Evolution of catalytic materials
3.1. Single-component semiconductors
When a single-component semiconductor is employed as a photocatalytic model, all charge-kinetics processes stated above can occur on the same semiconductor catalyst. As a result, their photocatalytic active surface change is relatively simple, including some defect dynamics and surface structural evolution. This part mainly introduces some examples of using in-situ technologies to monitor the evolution in single-component semiconductor catalysts and aids in the insight into the mechanism.
Defects play a crucial role in photocatalytic reactions since they are highly connected with the surface-active site[
![]()
Figure 4.(Color online) (a)
Surface atom migration on specific facets of semiconductors often occurs under photocatalytic reactions, which may influence their catalysis and stability. Some works report that the inelastic scattering of electron beam in electron microscopy can bring about an effect similar to that of UV light[
With the advancement of in-situ TEM, it is possible to directly observe the chemical change process of catalyst surface during light irradiation rather than electron beam irradiation mentioned above[
![]()
Figure 5.(Color online) (a)
Since most characterizations for photocatalytic reactions of anatase TiO2 are limited to water vapor environment, Sui et al. established a novel method to examine photocatalytic water splitting using anatase TiO2 in a genuine reaction environment (Fig. 5(f))[
Yu et al. also obtained in-situ TEM observations of an unstable Cu2O catalyst during a photocatalytic reaction. Fig. 5(i) shows HRTEM images of Cu2O samples in continuous irradiation[
3.2. Multi-component semiconductor
To promote the photocatalytic efficiency of semiconductors, various studies forward from the single-component system to the multi-component hybrid system, like a metal–semiconductor hybrid, semiconductor–semiconductor hybrid, and so on[
Integration with metal can harness the charging kinetics of semiconductors. Especially some noble metals, such as Au, Ag, and Cu, which offer strong plasmonic properties, can improve the activity of semiconductors[
![]()
Figure 6.(Color online) (a) Schematic diagram of water reduction in Au-TiO2 with UV- (right) and visible-light (left) driven. (b)
Construction of heterojunctions in semiconductor–semiconductor photocatalysts is another way to steer the charging kinetics, such as extending the spectral range for light absorption and enhancing electron-hole separation[
For multi-component semiconductors, the interfacial alterations of multiple components are also significant in the catalytic process. Many atomic-scale evolutions of the metal particle, oxide surface, and interface during catalysis remain unanswered. In-situ TEM is the best method to visualize atomic-scale dynamics. When there is a strong metal-support interaction (SMSI), metal nanoparticles can be reversibly covered when the support undergoes reduction. Bokhoven et al., use an in-situ TEM to study an actual Pt/TiO2 catalyst under changing gas atmosphere (Fig. 6(f))[
4. Dynamic process of surface reaction intermediate
Accurate and intuitive observation of the dynamic process of reaction intermediates is helpful to reveal the mechanism of photocatalysis and design an advanced photocatalyst. In-situ monitoring provides an effective tool for detecting reaction intermediates in the whole photocatalytic process[
4.1. Water photosplitting including HER and OER
The water-splitting, including hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) (Fig. 7(a)), are key chemical reactions for green hydrogen production and energy conversion process[
![]()
Figure 7.(Color online)
To monitor the water-splitting HER mechanism under realistic working conditions, Guo et al. proposed an in-situ measurement system combining linear-sweep voltammetry curves and operando Raman spectra and a possible HER mechanism of MoS2xSe2(1−x) (Figs. 7(b)−7(d)). The spectra results show that H atoms are adsorbed to the active S/Se atoms (or atoms) to form intermediate MoS2-H/MoSe2-H and subsequent hydrogen desorption during the HER process[
In addition, in-situ diffuse reflectance infrared Fourier-transform (DRIFT) spectroscopy were performed to determine the possible reaction intermediates on the ZnIn2S4@MoS2 in photocatalytic HER[
By analyzing the variations of infrared peaks after illumination with in-situ Fourier-transform infrared (FTIR) technique, Ye et al. studied the process of the HER on ReS2/CdS under different sacrificial reagents, including Na2S-Na2SO3 and lactic acid[
Primary intermediates of ultraviolet light-induced OER on the TiO2 (rutile) particles were monitored with in-situ MIRIR spectroscopy, and the results support the previously proposed OER mechanism[
4.2. CO2 conversion
Solar CO2 conversion, including CO2 photoreduction and CO2 photofixation to valuable chemicals (Fig. 8(a)), play an important role in energy storage and global warming[
![]()
Figure 8.(Color online)
Based on defect engineering, Wu et al. designed a kind of BiOBr layers with enriching oxygen vacancies and adopted in-situ FTIR spectroscopy to monitor the reaction intermediates during CO2 photoreduction to CO[
Chen et al. explored that the important role of oxygen vacancy confined in Bi2O3 nanosheets in the conversion process of CO2 and elaborated the reaction mechanism of CO2 photofixation to long-chain chemicals by in-situ DRIFT spectroscopy[
With in-situ DRIFT spectroscopy, Wang et al. detected carbon-based species formed on the surface of S-scheme core-shell TiO2@ZnIn2S4 heterojunction photocatalyst during photocatalytic CO2 reduction reaction (Fig. 8(f))[
The photothermal effect is not emphasized in traditional photocatalytic systems. To reveal the CO2 reduction intermediates and to further address the role of photothermal during the photocatalytic reactions, in-situ FTIR experiments were conducted at different temperatures with the oxygen vacancy-rich Zn2GeO4 nanobelts[
4.3. N2 fixation
Ammonia, an important raw material for nitrogen fertilizer, is synthesized from N2 and H2 under high temperature and pressure using the Haber–Bosch process[
![]()
Figure 9.(Color online)
Li et al. developed a simple low-temperature synthesis method to obtain a series of Bi5O7Br nanostructures[
Wang et al. utilized in-situ DRIFT spectroscopy to investigate the evolution of N-related intermediates in the early stages on the Bi5O7Br-NT[
To directly observe the mechanistic process of N2 photoreduction, Li et al. used in-situ DRIFT spectroscopy to monitor the evolution channel of reaction intermediates on the BiOBr within a well-designed reaction cell[
4.4. Other catalytic processes
In addition to the above catalytic process, some other critical reactions[
NO from industrial emissions is considered an important contributor to air pollution and has adverse effects on human health[
Shang et al. designed a kind of oxygen vacancy rich-blue TiO2 photocatalyst for highly selective removal of NO and proved the reaction mechanism of selective NO removal[
![]()
Figure 10.(Color online)
In addition, the in-situ FTIR spectra were used to analyze the time-dependent NO oxidation process in the presence of Bi0OVs-(BiO)2CO3[
Adsorbed hydroxyl radical •OH(ads) is an important surface immediate for photocatalysis reaction of TiO2[
5. Conclusion and prospects
Despite these advances in the field of in-situ characterization of photocatalysis, there is still an urgent need to further develop advanced and robust techniques to track the evolution of materials and reaction intermediates to gain a deeper understanding of the material changes and reaction mechanisms in the process of photocatalytic reactions.
For in-situ spectroscopic characterization, it is necessary to accurately control the reaction temperature, pressure, and atmosphere, and reduce the influence of the reaction device, so as to realize in-situ monitoring under real reaction conditions. Therefore, rational design of the in-situ device is required, for example, using more accurate probing thermocouples as close to the reaction system as possible without affecting the reaction environment. Use inert materials to design the reaction device to avoid the impact on the reaction environment at high temperatures. In addition, transient spectroscopy techniques with higher temporal resolution should be emphasized to monitor the kinetic behavior of active sites and reaction intermediates.
For microscopic imaging technology, especially in-situ TEM, the pursuit of high resolution (including spatial resolution and time resolution) is an eternal topic. Since catalysis reactions often occur at breakneck speed, time resolution needs to be improved with a good image signal-to-noise ratio to capture the dynamics and identify more intermediates in catalysis reactions. The advancement of electron detection can be quite beneficial. Like the newly developed approach allows for the detection of a single electron without the need to convert it to photons. The in-situ TEM can be utilized to examine dynamics on a millisecond time scale with this feature. The ultrafast TEM uses a brief electron pulse with a high current density and has a time resolution of up to femtoseconds. Ultrafast electron diffraction, in particular, can be used to investigate catalytic reactions with a significantly better temporal resolution.
Meanwhile, in-situ TEM needs to strike a balance between radiation damage and image resolution. Radiation damage is mainly caused by high-energy electron beams. When a sample is not resistant to radiation, the greatest defense is to lower the energy of the electron beams. The electron beam can be reduced while keeping acceptable resolution using spherical aberration-corrected electron microscopy. Note that when EDS and EELS are adopted, samples must be exposed to relatively high-energy electrons. It can be considered to carry out EDS and EELS analysis after the imaging characterization, which can reduce the generation of false images and improve the accuracy of experiments. In addition, updating the reaction cell materials to increase chip mechanical strength and chemical resistance would result in reduced window bulging, reducing the thickness of the liquid or gas layer, and so boosting spatial resolution.
Finally, the in-situ characterization process should organically combine a variety of in-situ techniques, including spectroscopy, microscopic imaging, and performance monitoring systems, to achieve a comprehensive and multidimensional information tracking of catalyst materials and reaction intermediates during the photocatalytic reaction. In addition, it is also necessary to focus on the whole photocatalytic process from beginning to end to elaborate on the real reaction mechanism and provide a deeper perspective on the design of advanced catalysts.
Acknowledgements
The work was supported by the National Science Foundation of China (21875137, 51521004, and 51420105009), Innovation Program of Shanghai Municipal Education Commission (Project No. 2019-01-07-00-02-E00069), the 111 Project (Project No. B16032), and the fund from Center of Hydrogen Science and Joint Research Center for Clean Energy Materials at Shanghai Jiao Tong University for financial supports.
References
[1] K Maeda, K Teramura, D Lu et al. Photocatalyst releasing hydrogen from water. Nature, 440, 295(2006).
[2] Y C Zhang, N Afzal, L Pan et al. Structure-activity relationship of defective metal-based photocatalysts for water splitting: Experimental and theoretical perspectives. Adv Sci, 6, 1900053(2019).
[3] S L Foster, S I P Bakovic, R D Duda et al. Catalysts for nitrogen reduction to ammonia. Nat Catal, 1, 490(2018).
[4] Q L Xu, L Y Zhang, B Cheng et al. S-scheme heterojunction photocatalyst. Chem, 6, 1543(2020).
[5] Y G Chao, P Zhou, N Li et al. Ultrathin visible-light-driven Mo incorporating In2O3-ZnIn2Se4 Z-scheme nanosheet photocatalysts. Adv Mater, 31, 1807226(2019).
[6] Y Gu, A P Wu, Y Q Jiao et al. Two-dimensional porous molybdenum phosphide/nitride heterojunction nanosheets for pH-universal hydrogen evolution reaction. Angew Chem Int Ed, 60, 6673(2021).
[7] S Khan, M Je, N N T Ton et al. C-doped ZnS-ZnO/Rh nanosheets as multijunctioned photocatalysts for effective H2 generation from pure water under solar simulating light. Appl Catal B, 297, 120473(2021).
[8] L Ran, J G Hou, S Y Cao et al. Defect engineering of photocatalysts for solar energy conversion. Sol RRL, 4, 1900487(2020).
[9] M Liu, Y Chen, J Su et al. Photocatalytic hydrogen production using twinned nanocrystals and an unanchored NiS
[10] M Barawi, L Collado, M Gomez-Mendoza et al. Conjugated porous polymers: Ground-breaking materials for solar energy conversion. Adv Energy Mater, 11, 2101530(2021).
[11] J G Wang, Y J Chen, W Zhou et al. Cubic quantum dot/hexagonal microsphere ZnIn2S4 heterophase junctions for exceptional visible-light-driven photocatalytic H2 evolution. J Mater Chem A, 5, 8451(2017).
[12] H B Yu, J H Huang, L B Jiang et al. Enhanced photocatalytic tetracycline degradation using N-CQDs/OV-BiOBr composites: Unraveling the complementary effects between N-CQDs and oxygen vacancy. Chem Eng J, 402, 126187(2020).
[13] D D Gao, X H Wu, P Wang et al. Selenium-enriched amorphous NiSe1+
[14] S Bai, J Jiang, Q Zhang et al. Steering charge kinetics in photocatalysis: Intersection of materials syntheses, characterization techniques and theoretical simulations. Chem Soc Rev, 44, 2893(2015).
[15] F Y Chen, Z Y Wu, Z Adler et al. Stability challenges of electrocatalytic oxygen evolution reaction: From mechanistic understanding to reactor design. Joule, 5, 1704(2021).
[16] S R Zhang, L Nguyen, Y Zhu et al.
[17] F Zaera.
[18] der Wal L I van, S J Turner, J Zečević. Developments and advances in
[19] A Knop-Gericke, E Kleimenov, M Hävecker et al. X-ray photoelectron spectroscopy for investigation of heterogeneous catalytic processes. Adv Catal, 52, 213(2009).
[20] M H M Ahmed, R H Temperton, J N O'Shea. An
[21] P Zhang, Y K Li, Y S Zhang et al. Photogenerated electron transfer process in heterojunctions:
[22] S Bordiga, E Groppo, G Agostini et al. Reactivity of surface species in heterogeneous catalysts probed by
[23] F Zaera. New advances in the use of infrared absorption spectroscopy for the characterization of heterogeneous catalytic reactions. Chem Soc Rev, 43, 7624(2014).
[24] I E Wachs, C A Roberts. Monitoring surface metal oxide catalytic active sites with Raman spectroscopy. Chem Soc Rev, 39, 5002(2010).
[25] H Kim, K M Kosuda, R P van Duyne et al. Resonance Raman and surface- and tip-enhanced Raman spectroscopy methods to study solid catalysts and heterogeneous catalytic reactions. Chem Soc Rev, 39, 4820(2010).
[26] M G Bakker, B Fowler, M K Bowman et al. Experimental methods in chemical engineering: Electron paramagnetic resonance spectroscopy-EPR/ESR. Can J Chem Eng, 98, 1668(2020).
[27] J B Wu, H Shan, W L Chen et al.
[28] W Grogger, F Hofer, G Kothleitner et al. An introduction to high-resolution EELS in transmission electron microscopy. Top Catal, 50, 200(2008).
[29] F Besenbacher, J V Lauritsen, S Wendt. STM studies of model catalysts. Nano Today, 2, 30(2007).
[30] A Preet, T E Lin. A review: Scanning electrochemical microscopy (SECM) for visualizing the real-time local catalytic activity. Catalysts, 11, 594(2021).
[31] G X Zhuang, Y W Chen, Z Y Zhuang et al. Oxygen vacancies in metal oxides: Recent progress towards advanced catalyst design. Sci China Mater, 63, 2089(2020).
[32] H F Feng, Z F Xu, L Ren et al. Activating titania for efficient electrocatalysis by vacancy engineering. ACS Catal, 8, 4288(2018).
[33] T T Hou, Y Xiao, P X Cui et al. Operando oxygen vacancies for enhanced activity and stability toward nitrogen photofixation. Adv Energy Mater, 9, 1902319(2019).
[34] N Kolmakova, A Kolmakov. Scanning electron microscopy for
[35] G Möbus, Z Saghi, D C Sayle et al. Dynamics of polar surfaces on ceria nanoparticles observed
[36] M Bugnet, S H Overbury, Z L Wu et al. Direct visualization and control of atomic mobility at {100} surfaces of ceria in the environmental transmission electron microscope. Nano Lett, 17, 7652(2017).
[37] F Cavalca, A B Laursen, B E Kardynal et al.
[38] L X Zhang, B K Miller, P A Crozier. Atomic level
[39] Y Lu, W J Yin, K L Peng et al. Self-hydrogenated shell promoting photocatalytic H2 evolution on anatase TiO2. Nat Commun, 9, 2752(2018).
[40] S H Yu, Y H Jiang, Y Sun et al. Real time imaging of photocatalytic active site formation during H2 evolution by
[41] M Rycenga, C M Cobley, J Zeng et al. Controlling the synthesis and assembly of silver nanostructures for plasmonic applications. Chem Rev, 111, 3669(2011).
[42] G R Bamwenda, S Tsubota, T Nakamura et al. Photoassisted hydrogen production from a water-ethanol solution: A comparison of activities of Au–TiO2 and Pt–TiO2. J Photochem Photobiol A, 89, 177(1995).
[43] J B Priebe, M Karnahl, H Junge et al. Water reduction with visible light: Synergy between optical transitions and electron transfer in Au-TiO2 catalysts visualized by in situ EPR spectroscopy. Angew Chem Int Ed, 52, 11420(2013).
[44] K S Yang, Y R Lu, Y Y Hsu et al. Plasmon-induced visible-light photocatalytic activity of Au nanoparticle-decorated hollow mesoporous TiO2: A view by X-ray spectroscopy. J Phys Chem C, 122, 6955(2018).
[45] O S Ekande, M Kumar. Review on polyaniline as reductive photocatalyst for the construction of the visible light active heterojunction for the generation of reactive oxygen species. J Environ Chem Eng, 9, 105725(2021).
[46] Y Yuan, R T Guo, L F Hong et al. A review of metal oxide-based Z-scheme heterojunction photocatalysts: Actualities and developments. Mater Today Energy, 21, 100829(2021).
[47] T M Di, Q L Xu, W Ho et al. Review on metal sulphide-based Z-scheme photocatalysts. ChemCatChem, 11, 1394(2019).
[48] L M Sai, X Y Kong. Type II hybrid structures of TiO2 nanorods conjugated with CdS quantum dots: Assembly and optical properties. Appl Phys A, 114, 605(2014).
[49] Y Y Zhu, Y F Liu, Y H Lv et al. Enhancement of photocatalytic activity for BiPO4 via phase junction. J Mater Chem A, 2, 13041(2014).
[50] J W Xue, J Bao. Interfacial charge transfer of heterojunction photocatalysts: Characterization and calculation. Surf Interfaces, 25, 101265(2021).
[51] H Yang. A short review on heterojunction photocatalysts: Carrier transfer behavior and photocatalytic mechanisms. Mater Res Bull, 142, 111406(2021).
[52] L B Wang, B Cheng, L Y Zhang et al.
[53] A Beck, X Huang, L Artiglia et al. The dynamics of overlayer formation on catalyst nanoparticles and strong metal-support interaction. Nat Commun, 11, 3220(2020).
[54] J L Vincent, P A Crozier. Atomic level fluxional behavior and activity of CeO2-supported Pt catalysts for CO oxidation. Nat Commun, 12, 5789(2021).
[55] B H Simpson, J Rodríguez-López. Emerging techniques for the
[56] Y Nosaka, A Y Nosaka. Generation and detection of reactive oxygen species in photocatalysis. Chem Rev, 117, 11302(2017).
[57] J Y Liu, Z D Wei, W F Shangguan. Defects engineering in photocatalytic water splitting materials. ChemCatChem, 11, 6177(2019).
[58] P A Connor, K D Dobson, A J McQuillan. Infrared spectroscopy of the TiO2/aqueous solution interface. Langmuir, 15, 2402(1999).
[59] S Haschke, M Mader, S Schlicht et al. Direct oxygen isotope effect identifies the rate-determining step of electrocatalytic OER at an oxidic surface. Nat Commun, 9, 4565(2018).
[60] W Y Lin, H Frei. Photochemical and FT-IR probing of the active site of hydrogen peroxide in Ti silicalite sieve. J Am Chem Soc, 124, 9292(2002).
[61] X Rong, J Parolin, A M Kolpak. A fundamental relationship between reaction mechanism and stability in metal oxide catalysts for oxygen evolution. ACS Catal, 6, 1153(2016).
[62] O Zandi, T W Hamann. Determination of photoelectrochemical water oxidation intermediates on haematite electrode surfaces using operando infrared spectroscopy. Nat Chem, 8, 778(2016).
[63] Z Zou, J Ye, K Sayama et al. Direct splitting of water under visible light irradiation with an oxide semiconductor photocatalyst. Nature, 414, 625(2001).
[64] S H Guo, Y H Li, S W Tang et al. Monitoring hydrogen evolution reaction intermediates of transition metal dichalcogenides via operando Raman spectroscopy. Adv Funct Mater, 30, 2003035(2020).
[65] Y H Peng, M J Geng, J Q Yu et al. Vacancy-induced 2H@1T MoS2 phase-incorporation on ZnIn2S4 for boosting photocatalytic hydrogen evolution. Appl Catal B, 298, 120570(2021).
[66] L Q Ye, Z Y Ma, Y Deng et al. Robust and efficient photocatalytic hydrogen generation of ReS2/CdS and mechanistic study by on-line mass spectrometry and
[67] X Wang, X Wang, J Huang et al. Interfacial chemical bond and internal electric field modulated Z-scheme Sv-ZnIn2S4/MoSe2 photocatalyst for efficient hydrogen evolution. Nat Commun, 12, 4112(2021).
[68] R Nakamura, Y Nakato. Primary intermediates of oxygen photoevolution reaction on TiO2 (Rutile) particles, revealed by
[69] M Zhang, M de Respinis, H Frei. Time-resolved observations of water oxidation intermediates on a cobalt oxide nanoparticle catalyst. Nat Chem, 6, 362(2014).
[70] Q Ding, Y Liu, T Chen et al. Unravelling the water oxidation mechanism on NaTaO3-based photocatalysts. J Mater Chem A, 8, 6812(2020).
[71] F Fresno, S Galdón, M Barawi et al. Selectivity in UV photocatalytic CO2 conversion over bare and silver-decorated niobium-tantalum perovskites. Catal Today, 361, 85(2021).
[72] M Halmann. Photoelectrochemical reduction of aqueous carbon dioxide on p-type gallium phosphide in liquid junction solar cells. Nature, 275, 115(1978).
[73] M Marszewski, S W Cao, J G Yu et al. Semiconductor-based photocatalytic CO2 conversion. Mater Horiz, 2, 261(2015).
[74] H Rao, L C Schmidt, J Bonin et al. Visible-light-driven methane formation from CO2 with a molecular iron catalyst. Nature, 548, 74(2017).
[75] M Schreier, F Héroguel, L Steier et al. Solar conversion of CO2 to CO using Earth-abundant electrocatalysts prepared by atomic layer modification of CuO. Nat Energy, 2, 17087(2017).
[76] Y Wang, X Shang, J Shen et al. Direct and indirect Z-scheme heterostructure-coupled photosystem enabling cooperation of CO2 reduction and H2O oxidation. Nat Commun, 11, 3043(2020).
[77] Y Kou, Y Nabetani, M S Dai et al. Direct detection of key reaction intermediates in photochemical CO2 reduction sensitized by a rhenium bipyridine complex. J Am Chem Soc, 136, 6021(2014).
[78] L J Liu, Y Li. Understanding the reaction mechanism of photocatalytic reduction of CO2 with H2O on TiO2-based photocatalysts: A review. Aerosol Air Qual Res, 14, 453(2014).
[79] L J Liu, C Y Zhao, J T Miller et al. Mechanistic study of CO2 photoreduction with H2O on Cu/TiO2 nanocomposites by
[80] J Wu, X D Li, W Shi et al. Efficient visible-light-driven CO2 reduction mediated by defect-engineered BiOBr atomic layers. Angew Chem Int Ed, 57, 8719(2018).
[81] S Chen, H Wang, Z Kang et al. Oxygen vacancy associated single-electron transfer for photofixation of CO2 to long-chain chemicals. Nat Commun, 10, 788(2019).
[82] J C Zhu, W W Shao, X D Li et al. Asymmetric triple-atom sites confined in ternary oxide enabling selective CO2 photothermal reduction to acetate. J Am Chem Soc, 143, 18233(2021).
[83] X J Ren, M C Gao, Y F Zhang et al. Photocatalytic reduction of CO2 on BiOX: Effect of halogen element type and surface oxygen vacancy mediated mechanism. Appl Catal B, 274, 119063(2020).
[84] B A MacKay, M D Fryzuk. Dinitrogen coordination chemistry: The biomimetic borderlands. ChemInform, 35, 703(2004).
[85] H D Shen, M M Yang, L D Hao et al. Photocatalytic nitrogen reduction to ammonia: Insights into the role of defect engineering in photocatalysts. Nano Res, 275, 115(2021).
[86] J P Guo, P Chen. Catalyst: NH3 as an energy carrier. Chem, 3, 709(2017).
[87] A J Medford, M C Hatzell. Photon-driven nitrogen fixation: Current progress, thermodynamic considerations, and future outlook. ACS Catal, 7, 2624(2017).
[88] B M Hoffman, D Lukoyanov, Z Y Yang et al. Mechanism of nitrogen fixation by nitrogenase: The next stage. Chem Rev, 114, 4041(2014).
[89] H P Jia, E A Quadrelli. Mechanistic aspects of dinitrogen cleavage and hydrogenation to produce ammonia in catalysis and organometallic chemistry: Relevance of metal hydride bonds and dihydrogen. Chem Soc Rev, 43, 547(2014).
[90] H Yuzawa, T Mori, H Itoh et al. Reaction mechanism of ammonia decomposition to nitrogen and hydrogen over metal loaded titanium oxide photocatalyst. J Phys Chem C, 116, 4126(2012).
[91] H Hirakawa, M Hashimoto, Y Shiraishi et al. Photocatalytic conversion of nitrogen to ammonia with water on surface oxygen vacancies of titanium dioxide. J Am Chem Soc, 139, 10929(2017).
[92] C C Li, T Wang, Z J Zhao et al. Promoted fixation of molecular nitrogen with surface oxygen vacancies on plasmon-enhanced TiO2 photoelectrodes. Angew Chem, 130, 5376(2018).
[93] H Li, J Shang, Z H Ai et al. Efficient visible light nitrogen fixation with BiOBr nanosheets of oxygen vacancies on the exposed {001} facets. J Am Chem Soc, 137, 6393(2015).
[94] P S Li, Z A Zhou, Q Wang et al. Visible-light-driven nitrogen fixation catalyzed by Bi5O7Br nanostructures: Enhanced performance by oxygen vacancies. J Am Chem Soc, 142, 12430(2020).
[95] S Y Wang, X Hai, X Ding et al. Light-switchable oxygen vacancies in ultrafine Bi5O7Br nanotubes for boosting solar-driven nitrogen fixation in pure water. Adv Mater, 29, 1701774(2017).
[96] J H Yang, Y Z Guo, R B Jiang et al. High-efficiency “working-in-tandem” nitrogen photofixation achieved by assembling plasmonic gold nanocrystals on ultrathin titania nanosheets. J Am Chem Soc, 140, 8497(2018).
[97] F Rao, G Q Zhu, W B Zhang et al.
[98] H Shang, M Q Li, H Li et al. Oxygen vacancies promoted the selective photocatalytic removal of NO with blue TiO2 via simultaneous molecular oxygen activation and photogenerated hole annihilation. Environ Sci Technol, 53, 6444(2019).
[99] H Jin, R You, S Zhou et al.
[100] D Zigah, J Rodríguez-López, A J Bard. Quantification of photoelectrogenerated hydroxyl radical on TiO2 by surface interrogation scanning electrochemical microscopy. Phys Chem Chem Phys, 14, 12764(2012).
[101] L B Kreuzer, C K Patel. Nitric oxide air pollution: Detection by optoacoustic spectroscopy. Science, 173, 45(1971).
[102] S Jin, G H Dong, J M Luo et al. Improved photocatalytic NO removal activity of SrTiO3 by using SrCO3 as a new co-catalyst. Appl Catal B, 227, 24(2018).
[103] Y F Lu, Y Huang, Y F Zhang et al. Oxygen vacancy engineering of Bi2O3/Bi2O2CO3 heterojunctions: Implications of the interfacial charge transfer, NO adsorption and removal. Appl Catal B, 231, 357(2018).
[104] R Nakamura, A Imanishi, K Murakoshi et al.
Set citation alerts for the article
Please enter your email address



