Dongwang YANG, Tingting LUO, Xianli SU, Jinsong WU, Xinfeng TANG. Unveiling the Intrinsic Low Thermal Conductivity of BiAgSeS through Entropy Engineering in SHS Kinetic Process [J]. Journal of Inorganic Materials, 2021, 36(9): 991
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 36, Issue 9, 991 (2021)
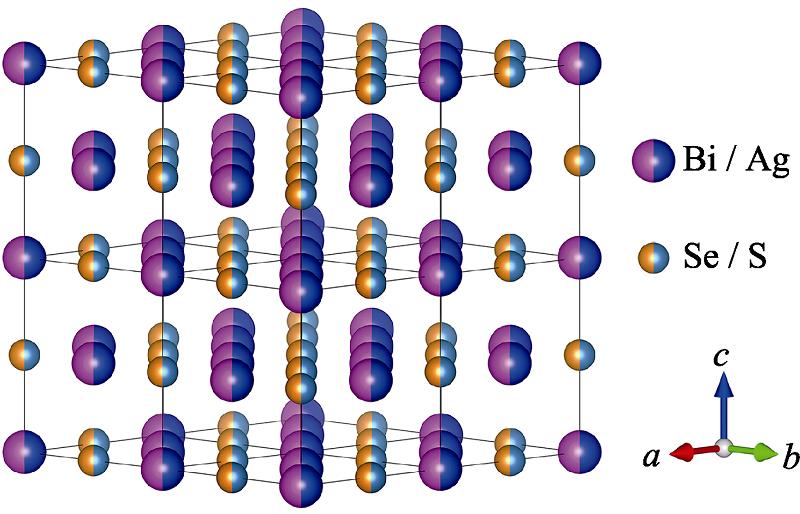
1. Crystal structure of BiAgSeS
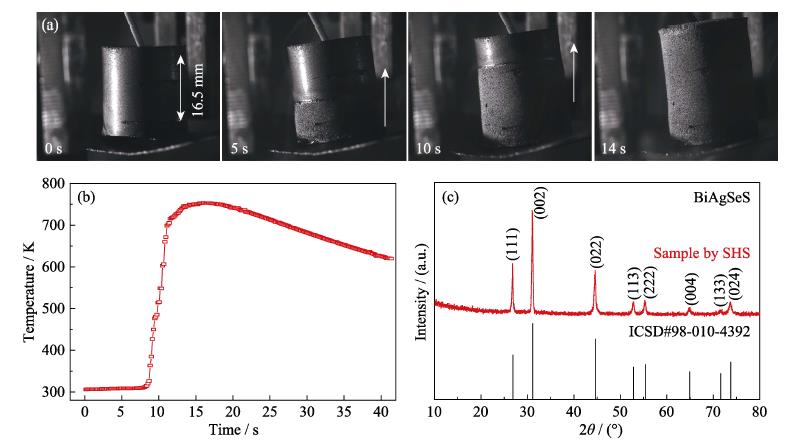
2. Photographs (a) for different stages of the SHS reaction process, temperature profile (b) of the SHS reaction, and XRD pattern (c) of the SHS product
3. Heat flow (a) at a heating rate of 85 K/min and a cooling rate of 50 K/min, and XRD patterns (b) of the products with temperature cutting off at 373, 400, 513 and 573 K
4. (a) Surface morphologies of the combustion wave quenching sample, (b) XRD patterns for typical regions, (c-f) FESEM images of microstructure and composition analysis from reactants to products (c) Mixture zone #1; (d) Preheating zone #2; (e) Reaction zone #3; (f) Product zone #4
5. Fracture surface morphologies of (a) MQ+PAS and (b) SHS+PAS BiAgSeS samples, and (c) secondary electron and (d) back scattering images of a polished surface of SHS+PAS sample
6. High temperature XRD patterns of (a) MQ+PAS and (b) SHS+PAS BiAgSeS bulk materials
7. DSC heat flow curves of MQ+PAS and SHS+PAS bulk samples at a heating and cooling rate of 5 K/min
8. Microstructures in BiAgSeS(a) Low-magnification TEM image revealing highly nanoscale distorted regions; (b) HRTEM image exhibiting highly atomic scale distortions; (c, e) HRTEM images depicting highly density of defects, including screw dislocation and strain-field domains; (d, e) Enlarged views of yellow marked regions in (c, e)
9. Thermoelectric performance of BiAgSeS samples(a) Electrical conductivity; (b) Seebeck coefficient; (c) Power factor; (d) Total thermal conductivity; (e) Lattice thermal conductivity; (f) Figure of merit ZT
S1. (a) Schematic diagram of the unstability of the crystal growth of BiAgSeS in SHS reaction; (b, c) the microstructure of SHS powder by FESEM
S2. Elemental distributions on the surface of “SHS+PAS” sample

Set citation alerts for the article
Please enter your email address



