Author Affiliations
1Faculty of Electronic Engineering Technology, Universiti Malaysia Perlis, 02600 Arau, Perlis, Malaysia2Faculty of Chemical Engineering Technology, Universiti Malaysia Perlis, 02600 Arau, Perlis, Malaysia3Institute of Nano Electronic Engineering, Universiti Malaysia Perlis, Perlis, Malaysia4Pure and Applied Physics, Ladoke Akintola University of Technology, Nigeria5Centre of Excellence for Nanobiotechnology and Nanomedicine (CoExNano), Faculty of applied Sciences, AIMST University, Semeling, 08100 Kedah, Malaysiashow less
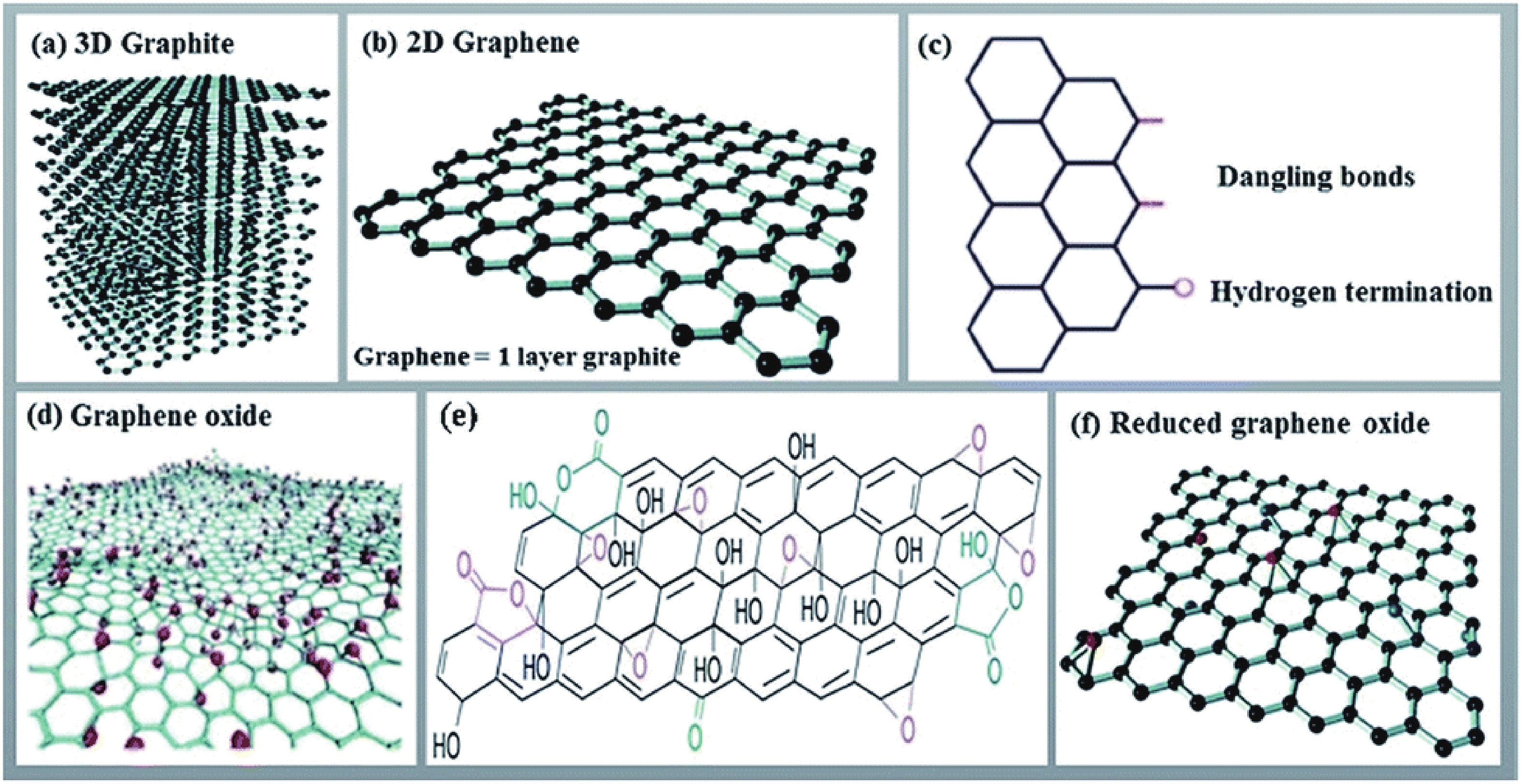
Fig. 1. (Color online) Structures of (a) 3D graphite, (b, c) 2D graphene and its edge, (d, e) graphene oxide, (f) reduced graphene oxide[22].
Fig. 2. (Color online) Top-down and bottom-up approaches for synthesis of graphene[23].
Fig. 3. Schematic diagram of the soluble salt assisted (Na2SO4) wet ball milling approach for synthesis of graphene nanosheet powder[36].
Fig. 4. (Color online) (a) Pure graphene. (b) Dry ice. (c) Edge-carboxylated graphite prepared by ball milling for 48 h. (d) Schematic view of physical cracking and edge-carboxylation of graphite by ball milling in the presence of dry ice, and protonation[38].
Fig. 5. (Color online) Schematic view: preparation of graphene oxide in laboratory designed ball mill[40].
Fig. 6. (Color online) Schematic view of tip sonication processing with parameters that influence graphene nanoplatelets dispersion in a liquid medium with obtained phenomena. (a) Fragmentation. (b) Exfoliation. (c) Defect[46].
Fig. 7. (Color online) Separation of graphitic oxide by sonication for 0.5 h.
Fig. 8. (Color online) Sonochemical synthesis of graphene oxide into graphene nanosheets in the presence NaOH[48].
Fig. 9. (Color online) Image of graphite flakes after electrochemical exfoliation. (b) Dispersed EG in DMF solution (concentration 2.5 mg/mL). (c) EG size on a bulk scale (163 g). (d) Diagrammatic representation of the principle of electrochemical exfoliation[53].
Fig. 10. (Color online) (a) Schematic view of the configuration used for face-to-face growth technique setup; (b) magnified view of the sample set up highlighted in panel (c) enlarged view of mounted SiC substrates highlighted by red lines in panel[64].
Fig. 11. (Color online) (a) Roll-to-roll process for the transfer of FLG from Ni foil to EVA/PET metal surface[76].
Fig. 12. (Color online) The graph of the sheet resistance versus the transmittance of the FLG/EVA/PET samples[76].
Fig. 13. (a) Pure VCCD-MWNT revealed the graphene helices released from the walls. (b) Milled for 1 h. (c, c’) Milled for 120 min[83].
Fig. 14. (Color online) Image of powder and the aqueous dispersion of graphene oxide (0.5 mg/mL) before (left) and after reduction (right)[104].
Fig. 15. SEM image of graphite-oxide[97].
Fig. 16. TEM images of graphite oxide[97].
Fig. 17. Plot of thermogravimetric analysis of (a) graphite oxide and (b) graphene[97]
Fig. 18. (Color online) Schematic view of the oxygen functionalities in GO and RGO[108].
| Metal surface | Pressure | Temp. (K) | Size & shape | H2/CH4(/Ar)
| Methods or annealing pretreatment | Mobility (cm2/(V·s))
| Growth time | Reference |
|---|
| Cu foil | LPCVD | 1308.15 | 0.5 × 10–3 m, dendrites
| 2/1.3 | Inside surface of copper-foil enclosures | 4000 (e) | | [87] (2011)
| | Cu foil | APCVD | 1323.15 | ~15 μm hexagonal
| H2/Ar, 10/300 CH4 in Ar 8 ppm
| H2/Ar, 10/ 300 sccm, 1323.15 K, 30 min annealing
| <103–104 | ~0.167 h | [88] (2011)
| | Cu foil | LPCVD | 1350.15 | ~2.3 × 10–3 m, ~4.5 × 10–6 m
| 70/0.15 | High pressure annealing (1500 torr, 500 sccm H2, 1350.15 K, electrochemical polishing
| ~11000 | 2.083 h | [89] (2012)
| | Ni (111) | UHV | 873.15–1073.15 | Millimeter size | Propylene ga(C3H6)
| Ni(111) hetero-epitaxially grown on MgO(111) | –
| 0.0833 h
| [90] (2011)
| | Cu foil | LPCVD | 1273.15 | 100 μm, six-lobed flower
| 12.5/1 | 0.667 h; vapor trapping | 4200; 20000 (hbn) | 0.5 h | [91] (2012)
| | Liquid foil | APCVD | 1433.15 | >100μm, hexagonal
| 300/6 | 200 sccm H2, 1373.15 K, 0.5 h
| 1000–2500 | 0.5 h, 10–50 μm/min
| [92] (2012)
| | Liquid foil | APCVD | 1363.15 | >200μm, hexagonal
| 80/10, CH4:Ar, 1.99
| 100 sccm (1.3 H2/Ar mix) 1090 °C, 0.5 h
| – | – | [93] (2012)
| | Cu foil | LPCVD | 1308.15 | Centimeter size | 10/0.1 | 0.1 torr H2, 1308.15, 0.5 h; 1 × 10–3 | 40000-65000 (1.7 K); 15000–30000 (r.t) | 12 h | [94] (2013)
| | Cu foil | APCVD | 1273.15 | 25 × 10–3 m diameter quartz
| | 10–15 sccm Ar, 600 sccm for H2, and 10–50 sccm for CH4 | 10–3700 (1273.15) | 0.333–0.16 h | [95] (2011)
| | Cu foil | LPCVD | 1308.15 | ~2 × 10–3 m
| 10/0.1 | Inside surface of Cu tube electroplating | 5200 | 6 h | [96] (2013)
| | Cu foil | LPCVD | 1273.15–
1318.15
| 0.25-inch-wide, 0.002 inch thick | 10/315 | 1010 °C and a pressure of 533.289 pascals with flows of 100 sccm H2 in both the inner tube were changed to 300 sccm H2 for the tube gap
| 25 mm/min (1273.15–
1318.15)
| 24 h | [97] (2015)
|
|
Table 1. Indicating the different graphene produced using CVD method.
| Source | Preparation | Morphology | Advantage | Properties | Application | Ref. |
|---|
| citrullus colocynthis (leaf extract)
| RGO was prepared from graphite powder using the modifiedhummers method | Stabilized
reduced graphene sheets
| Low cost, facile, green method for deoxygenation of GO. | Sharp diffraction peak
increase in interlayer spacing of GO
| Anticancer drugs | [110] (2017)
| | c. nucifera (cocos nucifera l.)
| Graphite oxide was prepared by oxidation of graphite with a mixture of sodium nitrate, concentrated ssulfuric acid and potassium chlorate | SEM and TEM images showed transparent and stable layers towards electron beam. AFM showed the bi-layer graphene. | Environmentally friendly
non-toxic reducing agent
| Low surface charge density | Biological materials | [111]
(2013)
| | Plants extracts (cherry, platanus, magnolia, persimmon, maple, pine and ginkgo). | Graphene oxide was prepared using the modified hummers method, which was followed by ultrasonication. | Reduced graphene oxide | Environmentally friendly | Increase in hydrophilicity which was caused by the reduction in polar functionality on the surface of the layers | Biomedical applications | [110] (2013)
| | Pomegranate juice | Improved hummers method was used to oxidize graphite for the synthesis of graphite oxide and followed by reduction of as-produced graphene oxide by pomegranate juice to form graphene nanosheets | Single or few layer graphene sheets | Facile and green method | Presence of several oxygen containing group in the presence of graphene oxide | Biological and optoelectronics. | [109]
(2014)
| | Ascorbic acid | Modified hummers method | Single layered graphene is 1 nm thick. | Low cost, green and efficient method, naturally available | Removal of oxygen functional group | Water purification | [111]
(2017)
| | Wild carrot root | Modified hummers method | Few layers graphene | Environmentally friendly reduction method, cost effectiveness, simple approach | Partial removal of oxygen functionality | Electronic devices | [112]
(2012)
| | Lime juice (citrus aurantifolia)
| The oxidization of graphite using hummers method to form GO and then the graphene oxide was reduced where lime was used as the natural reducing agents | Reduced graphene oxide | Low cost, environmentally benign method | The high intensity of the main peak in GO shows a sizeable number of oxygen containing groups, which occur after the deposition. | Biological materials | [113]
(2019)
| | magnifera indica | Mango leaves was cut down into tiny pieces (1–2 cm) and dipped in ethanol | Few layers graphene | Environmentally friendly, scalable, far and green method. | Biocompatible, photostable, excellent cellular uptake, good resolution | Biomedical nanotechnology applications | [114]
(2016)
|
|
Table 2. Different strategies on green synthesis of graphene.
| Method | Size | Advantage | Disadvantage | Application | Ref. |
|---|
| Epitaxial growth | 50 μm
| High quality, suitable for electronics | Highly expensive, low yield, wafer size, introduces voids in the transfer process | Field effect transistors, photodetectors | [115,116]
| | Chemical vapor deposition | 0.2–10 μm
| High quality and mass production, easy to transfer to other materials. | The use of harmful oxidizer or carboxylic acids, cost of the substrates may be high. The formation of graphene via high temperature on metal surface. | Electronics: light emitting diode, biosensors | [117,118]
| | Green synthesis | 200–800 nm | Low cost, facile (simple), Green method for deoxygenation of GO, reduces waste, the use of harmless solvent, suitable for large scale production of graphene nanoparticles, high temperature and pressure are not required, environmentally friendly | | Dye removal, electrochemical storage, Photocatalysis | [119,120]
| | Mechanical exfoliation | 5–10 nm | Cost effective, high quality graphene layers and laborsaving | Low yield, defects and in the flakes produce are inconsistent. | Space protection, energy | [121,122]
| | Electrochemical exfoliation | 2–3 nm | High quality single layer | Difficulty in removing the surfactants molecules, inconsistency in the produced graphene layer | Supercapacitors, batteries | [118,119]
|
|
Table 3. Different ways of synthesis for graphene.
![(Color online) Structures of (a) 3D graphite, (b, c) 2D graphene and its edge, (d, e) graphene oxide, (f) reduced graphene oxide[22].](/richHtml/jos/2022/43/6/061101/img_1.jpg)
![(Color online) Top-down and bottom-up approaches for synthesis of graphene[23].](/richHtml/jos/2022/43/6/061101/img_2.jpg)