Huayu Tao, Tianlin Wang, Danyang Li, Jie Xing, Gengwei Li. Preparation, properties, and applications of Bi2O2Se thin films: A review[J]. Journal of Semiconductors, 2023, 44(3): 031001
Search by keywords or author
- Journal of Semiconductors
- Vol. 44, Issue 3, 031001 (2023)
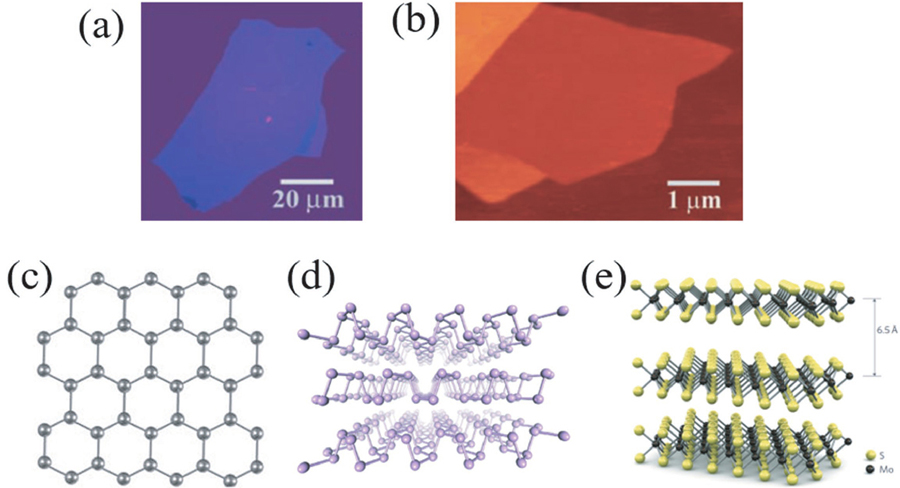
![(Color online) (a) Graphene optical photographs with a thickness of about 3 nm. (b) Atomic force microscope images of monolayer graphene. Reproduced with permission[9]. Copyright 2004, The American Association for the Advancement of Science. (c) Schematic diagram of the atomic structure of graphene. (d) Schematic diagram of black phosphorus atomic structure. Reproduced with permission[14]. Copyright 2014, Nature Publishing Group. (e) Schematic diagram of MoS2 atomic structure. Reproduced with permission[15]. Copyright 2011, Nature Publishing Group.](/richHtml/jos/2023/44/3/031001/031001_f1.jpg)
Fig. 1. (Color online) (a) Graphene optical photographs with a thickness of about 3 nm. (b) Atomic force microscope images of monolayer graphene. Reproduced with permission[9]. Copyright 2004, The American Association for the Advancement of Science. (c) Schematic diagram of the atomic structure of graphene. (d) Schematic diagram of black phosphorus atomic structure. Reproduced with permission[14]. Copyright 2014, Nature Publishing Group. (e) Schematic diagram of MoS2 atomic structure. Reproduced with permission[15]. Copyright 2011, Nature Publishing Group.
![(Color online) Preparation of two-dimensional films by CVD method. (a) CVD preparation diagram. Reproduced with permission[31]. Copyright 2017, American Chemical Society. (b) 2D Bi2O2Se crystal synthesized on mica. Reproduced with permission[11]. Copyright 2017, Nature Publishing Group. (c–f) Domain size and crystal phase transition of Bi2O2Se thin films. Reproduced with permission[31]. Copyright 2017, American Chemical Society. (g, h) SEM of both transverse Bi2O2Se and vertical triangular Bi2OxSe. Reproduced with permission41]. Copyright 2018, Wiley-VCH. (i) Improved preparation method. Reproduced with permission[32]. Copyright 2019, American Chemical Society. (j, k) Schematic of VS growth mechanism. Reproduced with permission[20]. Copyright 2019, Wiley-VCH. (l) SEM pictures of Bi2O2Se on STO. Reproduced with permission[19]. Copyright 2019, American Chemical Society. (m) Vertical growth of 2D Bi2O2Se films. Reproduced with permission[43]. Copyright 2019, Wiley-VCH.](/richHtml/jos/2023/44/3/031001/031001_f2.jpg)
Fig. 2. (Color online) Preparation of two-dimensional films by CVD method. (a) CVD preparation diagram. Reproduced with permission[31]. Copyright 2017, American Chemical Society. (b) 2D Bi2O2Se crystal synthesized on mica. Reproduced with permission[11]. Copyright 2017, Nature Publishing Group. (c–f) Domain size and crystal phase transition of Bi2O2Se thin films. Reproduced with permission[31]. Copyright 2017, American Chemical Society. (g, h) SEM of both transverse Bi2O2Se and vertical triangular Bi2OxSe. Reproduced with permission41]. Copyright 2018, Wiley-VCH. (i) Improved preparation method. Reproduced with permission[32]. Copyright 2019, American Chemical Society. (j, k) Schematic of VS growth mechanism. Reproduced with permission[20]. Copyright 2019, Wiley-VCH. (l) SEM pictures of Bi2O2Se on STO. Reproduced with permission[19]. Copyright 2019, American Chemical Society. (m) Vertical growth of 2D Bi2O2Se films. Reproduced with permission[43]. Copyright 2019, Wiley-VCH.
Fig. 3. (Color online) (a) Crystal structure of Bi2O2Se. Reproduced with permission[46]. Copyright 2018, Wiley-VCH. (b, c) HRTEM of Bi2O2Se. Reproduced with permission[31]. Copyright 2017, American Chemical Society.
Fig. 4. (Color online) (a–c) ARPES of Bi2O2Se films. Reproduced with permission[11]. Copyright 2017, Nature Publishing Group. (d) Band structure of Bi2O2Se films and bulk. Reproduced with permission[16]. Copyright 2019, Nature Publishing Group.
Fig. 5. (Color online) (a) Raman spectra of Bi2O2Se films with different layers. Reproduced with permission[20]. Copyright 2019, Wiley-VCH. (b–d) Four Raman vibration modes of Bi2O2Se. Reproduced with permission[55]. Copyright 2018, American Chemical Society.
Fig. 6. (Color online) (a, b) Transmittance and band gap of 2D Bi2O2Se films and bulk. Reproduced with permission[31]. Copyright 2017, American Chemical Society. (c) Peak differential reflection (blue symbols) and PL (red curve) of the 13 nm nanoplate as a function of the probe wavelength (upper panel) and its transmittance spectrum (lower panel). Reproduced with permission[56]. Copyright 2020, Wiley-VCH. (d) Peak differential reflection (blue symbols) and PL (red curve) of the monolayer as a function of the probe wavelength. Reproduced with permission[56]. Copyright 2020, Wiley-VCH.
Fig. 7. (Color online) (a) TEM image of Bi2O2Se on a copper grid. Reproduced with permission[20]. Copyright 2019, Wiley-VCH. (b) Photograph of 2D Bi2O2Se photodetectors and arrays on mica. (c) Optical image of 3 × 5 multi-pixel array of 2D Bi2O2Se photodetectors. (d) Photocurrent of a 2D Bi2O2Se photodetector in air. Reproduced with permission[40]. Copyright 2018, Nature Publishing Group.
Fig. 8. (a) Electrical conductivityσ of the Bi2O2Se film as a function of temperature. (b) Thermal conductivityκ of the Bi2O2Se rectangular block as a function of temperature. (c) Seebeck coefficientS of the Bi2O2Se film as a function of temperature. Reproduced with permission[59]. Copyright 2013, Elsevier.
Fig. 9. (Color online) Schematic representation of device applications of Bi2O2Se.
Fig. 10. (Color online) (a) Output curves of a 6.2 nm-thick Bi2O2Se device at room temperature. (b)μapp andIon/Ioff of Bi2O2Se FETs as a function of channel thickness. Reproduced with permission[11]. Copyright 2017, Nature Publishing Group. (c) OM image of centimeter-scale 2D Bi2O2Se arrays. (d) LinearIds–Vds curves of Bi2O2Se device with/without the illumination of 532 nm incident laser. Reproduced with permission[67]. Copyright 2017, Wiley-VCH. (e–g) Photocurrent, dark current, responsivity, detectivity, and response time as a function of temperature. Reproduced with permission[41]. Copyright 2018, Wiley-VCH.
Fig. 11. (Color online) (a) Schematic illustration of the PbSe/Bi2O2Se photodetector. (b) The estimated Type II energy band alignment between PbSe and Bi2O2Se before and after contact based on the estimated valence band offset and work function difference in UPS. (c) Photoresponse spectra. (d) Response decay dynamics of the hybrid photodetector. Reproduced with permission[68]. Copyright 2019, American Chemical Society. (e) Schematic illustration of the Bi2O2Se/MoSe2 heterojunction photodetector. Reproduced with permission[70]. Copyright 2019, Springer Science Business Media, LLC, part of Springer Nature. (f) OM image of as-fabricated device with 3-layered Bi2O2Se IPJ. (g) Output characteristic curve of the device in panel (m). Reproduced with permission[71]. Copyright 2019, Chinese Physical Society. (h) Preparation of Au/Bi2O2Se/Au MSM structures on mica substrates with a probe tip. Reproduced with permission[72]. Copyright 2019, Royal Society of Chemistry.
Fig. 12. (Color online) (a) Structure of polymer solar cells. (b) Energy diagram of the device. Reproduced with permission[79]. Copyright 2020, American Chemical Society.
Fig. 13. (Color online) (a) Schematic diagram of the Bi2O2Se device structure. (b) Optical image of the Bi2O2Se memristor. Reproduced with permission[25]. Copyright 2018, Wiley-VCH.
Fig. 14. (Color online) Structure diagram of a Bi2O2Se optical switch. Reproduced with permission[46]. Copyright 2018, Wiley-VCH.
Fig. 15. (Color online) (a) TEM image, (b) AFM image, and (c) height analysis of Bi2O2Se QDs. Reproduced with permission[23]. Copyright 2019, Wiley-VCH.
|
Table 0. Summary of preparation methods, growth conditions, and basic characteristics for Bi2O2Se.
|
Table 0. Comparisons of device performance of Bi2O2Se and other 2D materials.
Set citation alerts for the article
Please enter your email address



