Yiyuan YAN, Jiangwei JU, Meiyan YU, Shougang CHEN, Guanglei CUI. In-situ Polymerization Integrating 3D Ceramic Framework in All Solid-state Lithium Battery [J]. Journal of Inorganic Materials, 2020, 35(12): 1357
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 35, Issue 12, 1357 (2020)
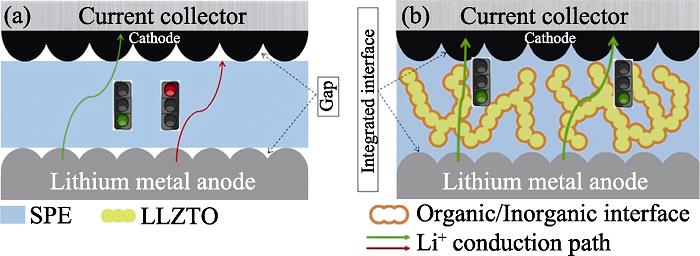
1. Schematic illustration of ASLB structure prepared via (a) ex-situ and (b) in-situ methods with p-LLZTO as ceramic fillers
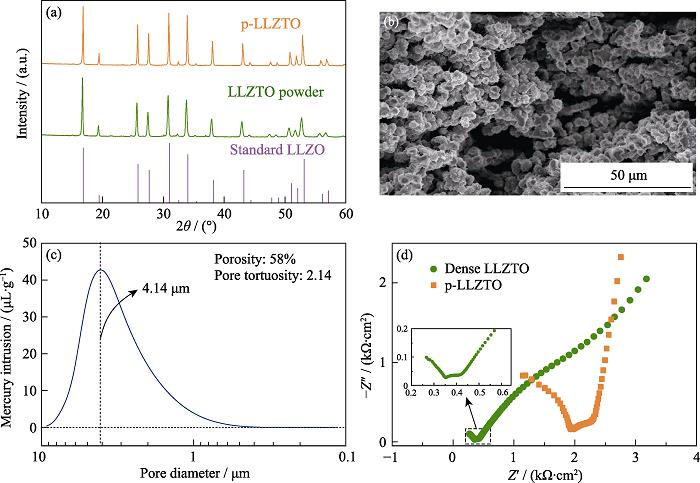
2. (a) XRD patterns of standard LLZO, the as-prepared LLZTO powders and p-LLZTO; (b) Cross sectional SEM image of p-LLZTO; (c) Pore size distribution of p-LLZTO; (d) EIS plots of dense LLZTO and p-LLZTO at room temperature with inset showing the partial magnified spectrum of the dense LLZTO
3. (a) FT-IR spectra of PEGMEA, P(PEGMEA), and P(PEGMEA) from the 3D composite; (b) 1H NMR spectra of PEGMEA and P(PEGMEA) from the 3D composite(the solvents are deuterated N ,N -dimethylformamide) with insets showing the corresponding structural formula of PEGMEA and P(PEGMEA); (c) Thermal evolution of ohmic resistance at 60 ℃ for steel|3D composite|steel symmetrical cell with inset showing the digital image of PEGMEA with/without p-LLZTO after heat-treatment at 60 ℃ for 24 h; (d) Relation between ionic conductivity of electrolyte and temperature for P(PEGMEA) and 3D composite; (e) Cross sectional SEM image and element mapping analysis of the 3D composite
4. EIS plots of (a-c) pre- and (d-f) post-treated Li-Li symmetrical batteries based on (a, d) PEGMEA, (b, e) LLZTO, (c, f) 3D composites; (g) Ohmic and (h) interfacial resistance comparison of pre- and post-treated Li-Li symmetrical cells; (i) DC galvanostatic cycle of Li-Li symmetrical batteries based on P(PEGMEA) and the 3D composite under room temperature at 0.1 mA·cm-2 with insets showing D.C. galvanostatic cycle of Li-Li symmetrical battery based on LLZTO(up) and the magnified profile of Li|3D composite|Li(down)
5. (a) Cycle performances of in-situ LiCoO2|3D composite|Li, in-situ LiCoO2|P(PEGMEA)|Li, ex-situ LiCoO2|3D composite|Li ASLBs; (b) Charge-discharge curves of in-situ LiCoO2|3D composite|Li, in-situ LiCoO2|P(PEGMEA)|Li, ex-situ LiCoO2|3D composite|Li ASLBs; Cross-sectional SEM images of the LiCoO2/3D composite interface from the disassembled (c) in-situ and (d) ex-situ LiCoO2|3D composite|Li ASLBs
S1. Linear sweep voltammetry measurement for 3D composite at room temperature
S2. Current variation with time during polarization of (a) Li|P(PEGMEA)|Li and (b) Li|3D composite|Li symmetrical cell at room temperature
|
Table 1. Conductivities $(\sigma_{Li^+})$ of different solid electrolytes at room temperature

Set citation alerts for the article
Please enter your email address



