Yao Tong, Qin Wang, Heng Yang, Xiaoting Liu, Enrou Mei, Xiaojuan Liang, Zhijun Zhang, Weidong Xiang. Enhanced multimodal luminescence and ultrahigh stability Eu3+-doped CsPbBr3 glasses for X-ray detection and imaging[J]. Photonics Research, 2021, 9(12): 2369
Search by keywords or author
- Photonics Research
- Vol. 9, Issue 12, 2369 (2021)
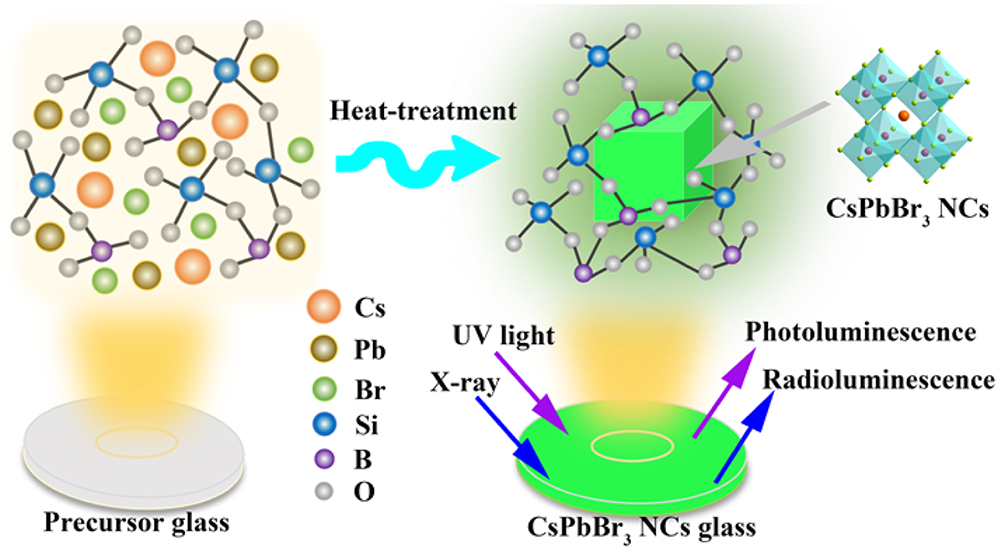
Fig. 1. Schematic illustration of the in situ crystallization process of CsPbBr 3
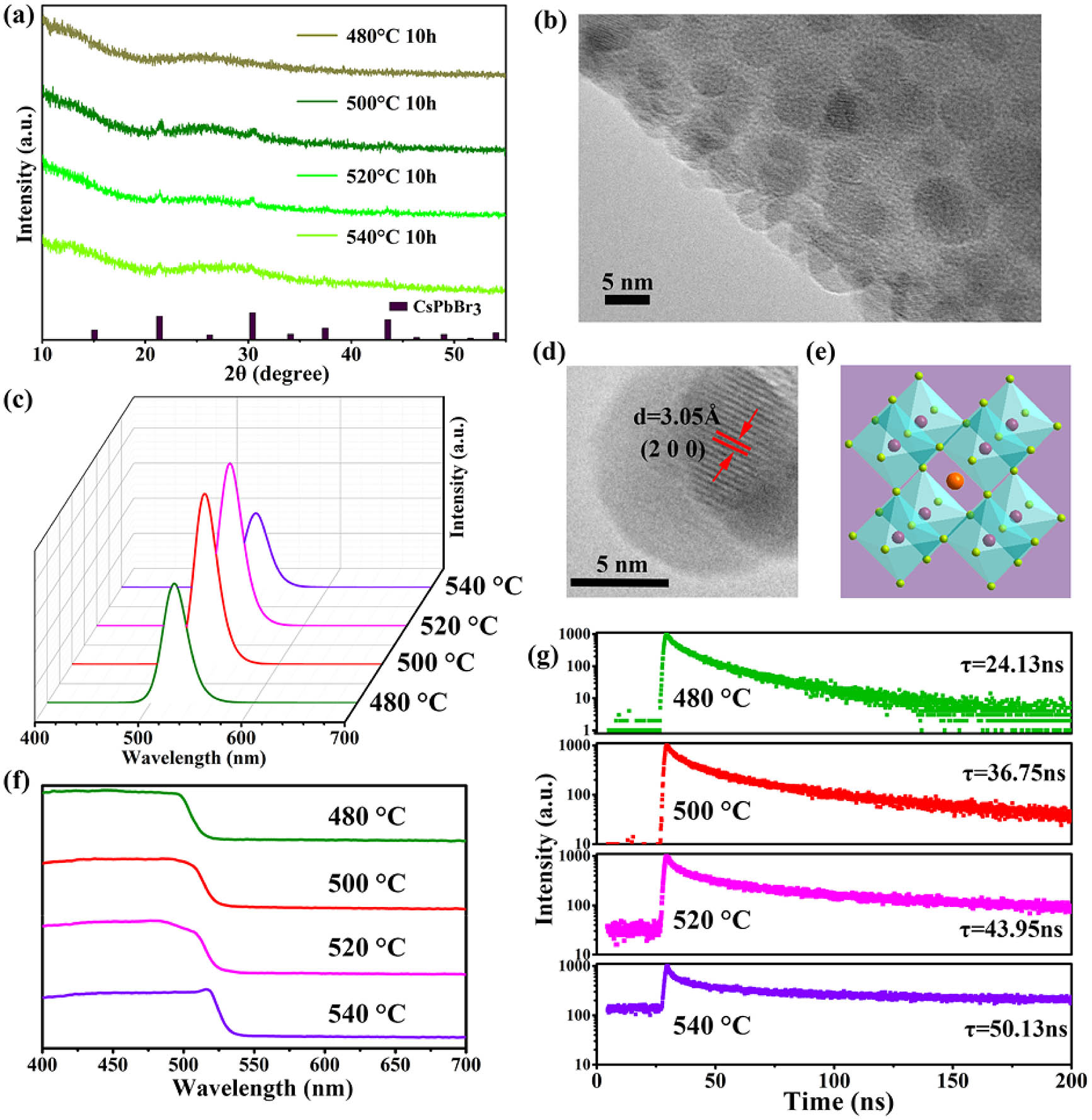
Fig. 2. (a) XRD patterns of CPB-0Eu at different heat-treatment temperatures. (b) TEM image of CPB-0Eu at 520°C for 10 h. (c) PL spectra under UV light excitation of CPB-0Eu at different heat-treatment temperatures. (d) HRTEM image of CPB-0Eu at 520°C for 10 h. (e) Standard structure model diagram of CsPbX 3
Fig. 3. (a) Images of CPB-x Eu glass samples under daylight (left) and in UV light (right). (b) XRD patterns of CPB-x Eu glasses. (c) Bandgap spectra of CPB-0Eu at 480°C–540°C. (d) PL spectra and (e) bandgap spectra of CPB-x Eu glasses.
Fig. 4. (a) TEM image of CPB-1.8Eu (illustration is the HRTEM image of CPB-1.8Eu at 520°C for 10 h). (b) XPS survey spectra of CPB-0Eu and CPB-1.8Eu. (c) EDX mapping images of CPB-1.8Eu. Corresponding high-resolution spectra of (d) Cs 3d for CPB-0Eu and CPB-1.8Eu, (e) Pb 4f for CPB-x Eu (x = 0
Fig. 5. TEM images and histograms of size distribution of (a) CPB-0Eu and (b) CPB-1.8Eu. (c) Transmittance spectra of CPB-x Eu (x = 0 x Eu (x = 0 x Eu (x = 0 ∼ 0.8 mm
Fig. 6. RL spectra of (a) CPB-0Eu, (b) CPB-0.6Eu, and (c) CPB-1.8Eu upon increasing current of X-ray cathode tube. (d), (e) Schematic illustration of RL mechanism about CPB-x Eu.
Fig. 7. (a) Schematic of the experimental setup used for X-ray imaging with sample (X-rays are excited directly at the side of the sample; voltage: 50 kV, dose rate: 69.5 μ Gy air / s
Fig. 8. Relative PL intensity at five different locations of the same sample.
Fig. 9. (a) Excitation spectra of CPB-x Eu glasses. (b)–(e) PL spectra of CPB-x Eu glasses excited at 365–395 nm.
Fig. 10. Characteristic PL spectra of Eu ions at 560–700 nm under different wavelength excitation of CPB-0.6Eu.
Fig. 11. PL decay of CPB-0Eu and CPB-0.6Eu glasses.
Fig. 12. RL spectra of CPB-x Eu (x = 0, 0.6, 1.8) under X-ray excitation at a radiation dose of 105 μGyair/s and a voltage of 50 kV, including the absorption spectrum of CPB-0.6Eu to reflect the self-absorption phenomenon.
Fig. 13. Linear relation based on RL intensity and X-ray dose rate.
Fig. 14. The relative RL intensity of CPB-0.6Eu and CPB-0Eu glass depending on time of X-ray radiation at a radiation dose of 105 μGyair/s and a voltage of 50 kV. The illustration shows a histogram of Pb2+ solubility after 5 days of water immersion before and after X-ray radiation.

Set citation alerts for the article
Please enter your email address



