Haipeng JI, Zongtao ZHANG, Jian XU, Setsuhisa TANABE, Deliang CHEN, Rongjun XIE. Advance in Red-emitting Mn4+-activated Oxyfluoride Phosphors [J]. Journal of Inorganic Materials, 2020, 35(8): 847
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 35, Issue 8, 847 (2020)
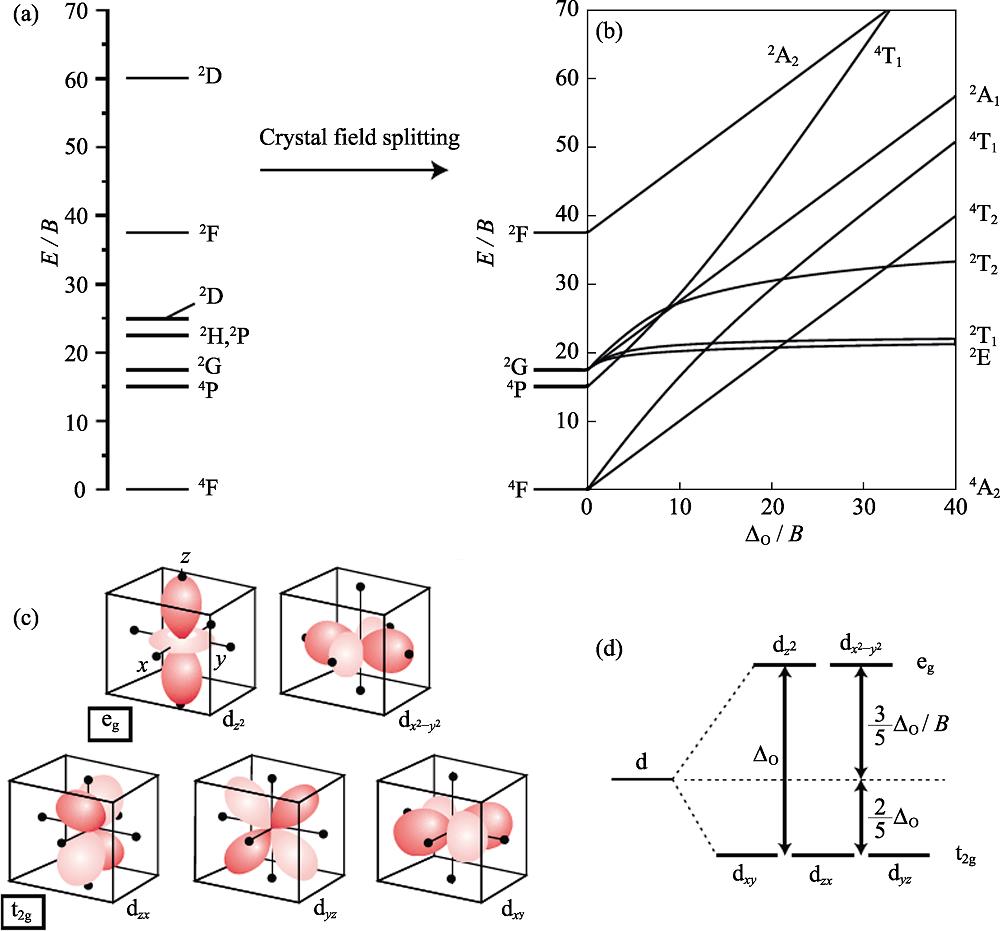
![Energy levels arising from a d3 configuration for a free transition metal ion (C=4.5B) (a), Tanabe-Sugano diagram for the d3 electron configuration in an octahedral crystal field (C=4.5B) (b), orientation of the five d-orbitals with respect to the ligands of an octahedral complex (black dots showing the ligands around the transition metal ion) (c), and crystal field splitting for the d-orbitals in an octahedral crystal field (d)[16]](/richHtml/jim/2020/35/8/847/img_1.png)
1. Energy levels arising from a d3 configuration for a free transition metal ion (C =4.5B ) (a), Tanabe-Sugano diagram for the d3 electron configuration in an octahedral crystal field (C =4.5B ) (b), orientation of the five d-orbitals with respect to the ligands of an octahedral complex (black dots showing the ligands around the transition metal ion) (c), and crystal field splitting for the d-orbitals in an octahedral crystal field (d)[16]
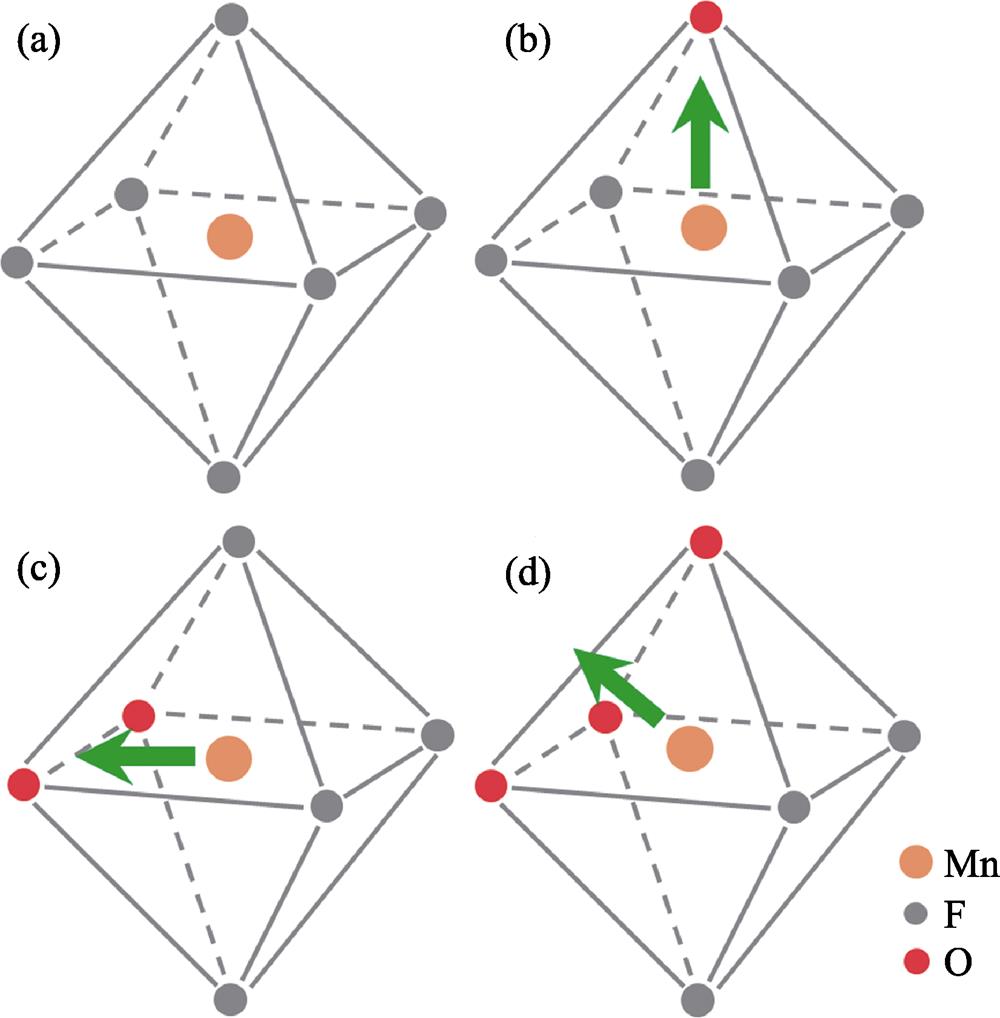
2. Regular octahedron coordination and distorted octahedra coordination
3. Unit cell of Na2WO2F4 (a), highly-distorted [WO2F4] octahedra (b), and emission spectrum of Na2WO2F4:Mn4+ (c) [21] with inset showing phosphor image under 460 nm light
4. (a) Unit cell of Cs2WO2F4 which contains slightly- distorted [W(O,F)6] octahedra, with the bottom-right showing the local coordination of Mn4+ in K2MnF6; (b) Excitation and emission spectra of Cs2WO2F4:Mn4+ with inset showing the phosphor image under 365 nm light[23]
5. PLE and DRS spectra of the Cs2NbOF5:Mn4+ phosphor (a) and temperature-dependent emission spectra of Cs2NbOF5:Mn4+ (b)[24] with the inset showing the intensity evolution of the integrated emission (I e), the stokes emission (I s) and the anti-stokes emmission (I a)
6. The PLE (a) and PL (b) spectra of the BaNbOF5:Mn4+ phosphor at temperature of 78 and 298 K with insets showing the phosphor images under natural or UV light[26]
7. Unit cell of Sr2ScO3F (a) and temperature-dependent emission spectra of Sr2ScO3F:Mn4+ (b)[27]Sr: yellow; Sc: blue; O: red; F: gray
8. Excitation and emission spectra of BaTiOF4:Mn4+ at room temperature (a), emission spectra of BaTiOF4:Mn4+ at 77 K and 293 K (b), unit cell of BaTiOF4 (c), and distorted octahedron coordination of [Ti2OF4] (d)[28]Ba: yellow; Ti: blue; O: red; F: gray
9. Comparison of the calculated Mn4+ energy levels in Mg28Ge7.55O32F15.04 for all possible Mn4+ positions in Ge/Mg sites with the measured spectrum[29]
10. Unit cell of LiAl4O6F and coordination of Al3+/Li+ (a) and emission spectra of LiAl4O6F:Mn4+ at temperature of 298-523 K (b) [30]
|
Table 1. The reported Mn4+ activated oxyfluoride phosphors
Set citation alerts for the article
Please enter your email address



