Wei ZHANG, Chen LIU, Yuantao CHEN, Wangsuo WU. Removal of Boron from Water by Mg-Al-Ce Hydrotalcite Adsorption [J]. Journal of Inorganic Materials, 2020, 35(3): 337
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 35, Issue 3, 337 (2020)
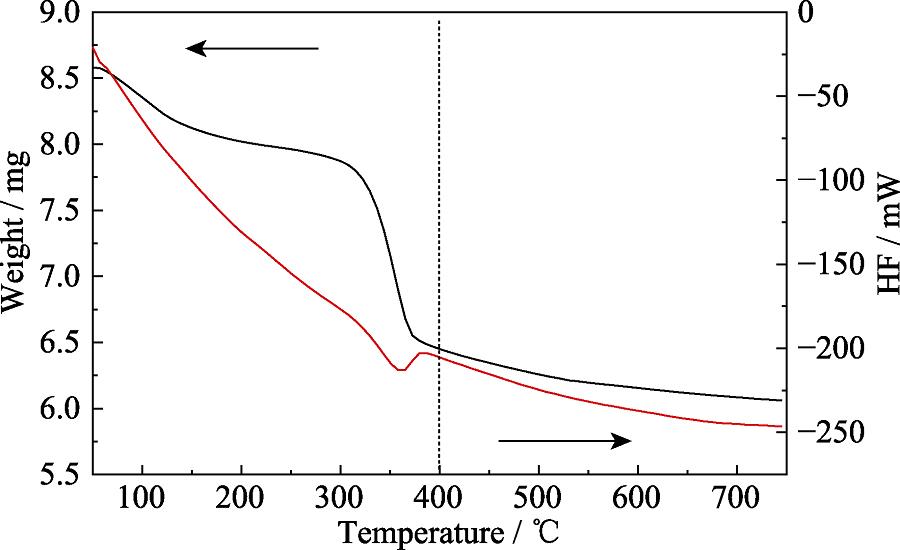
. TG-DSC curve of Mg-Al-Ce-HT precursor
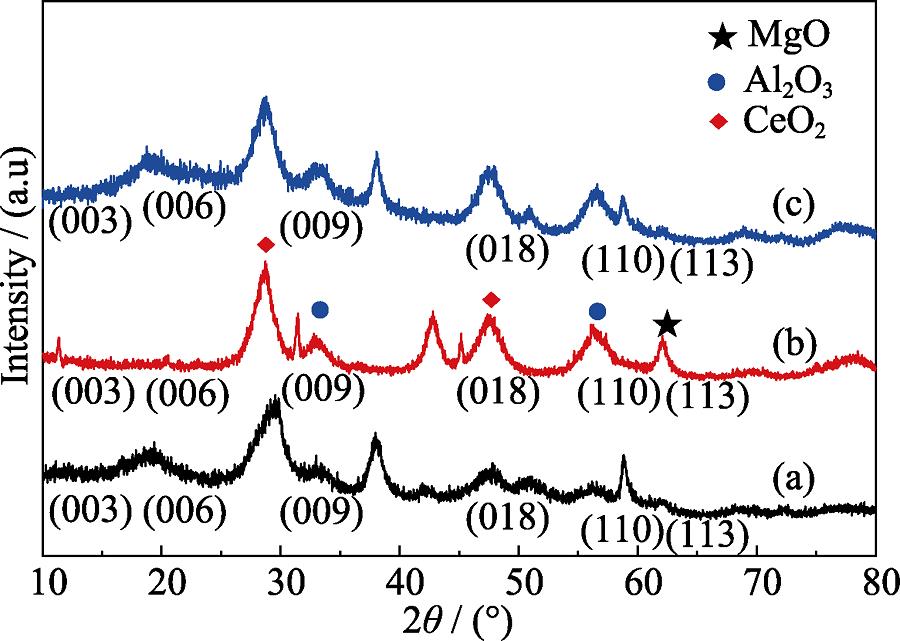
. XRD patterns of Mg-Al-Ce precursor (a), Mg-Al-Ce-HT before adsorption (b), and Mg-Al-Ce-HT after adsorption(c)
. FT-IR spectra of Mg-Al-Ce-HT precursor (a); Mg-Al-Ce-HT materials (b) and adsorbed materials (c)
. SEM images of Mg-Al-Ce-HT (a) and EDS analysis of Mg-Al-Ce-HT (b) and the corresponding EDS mappings of O (c), Mg (d), Ce (e), and Al (f) elements
. Schematic of the B(OH)3 adsorption process on the surface of Mg-Al-Ce-HT material(a), and XPS patterns of full elemental spectrogram (b)
. XPS spectra of Mg 2p, Al 2p, Ce 3d, C 1s, and O 1s in Mg-Al-Ce-HT before and after adsorption
. Effect of initial pH on adsorption efficiency (a) and the relationship between the conversion of H3BO3 and B(OH)4- (b)
. Effect of adsorbents, adsorption percentages, and adsorption capacity
. Effect of initial concentration of boric acid (a) and the Langmuir model (b)
. Effect of contact time on boron adsorption and kinetics model
. Van’s Hoff plot of boron adsorption on Mg-Al-Ce-HT
|
Table 1.
Thermodynamic parameters for boron adsorption on Mg-Al-Ce-HT

Set citation alerts for the article
Please enter your email address



