Jianan Yuan, Kang Xia, Chi Ding, Xiaomeng Wang, Qing Lu, Jian Sun. High-energy-density metal nitrides with armchair chains[J]. Matter and Radiation at Extremes, 2022, 7(3): 038402
Search by keywords or author
- Matter and Radiation at Extremes
- Vol. 7, Issue 3, 038402 (2022)
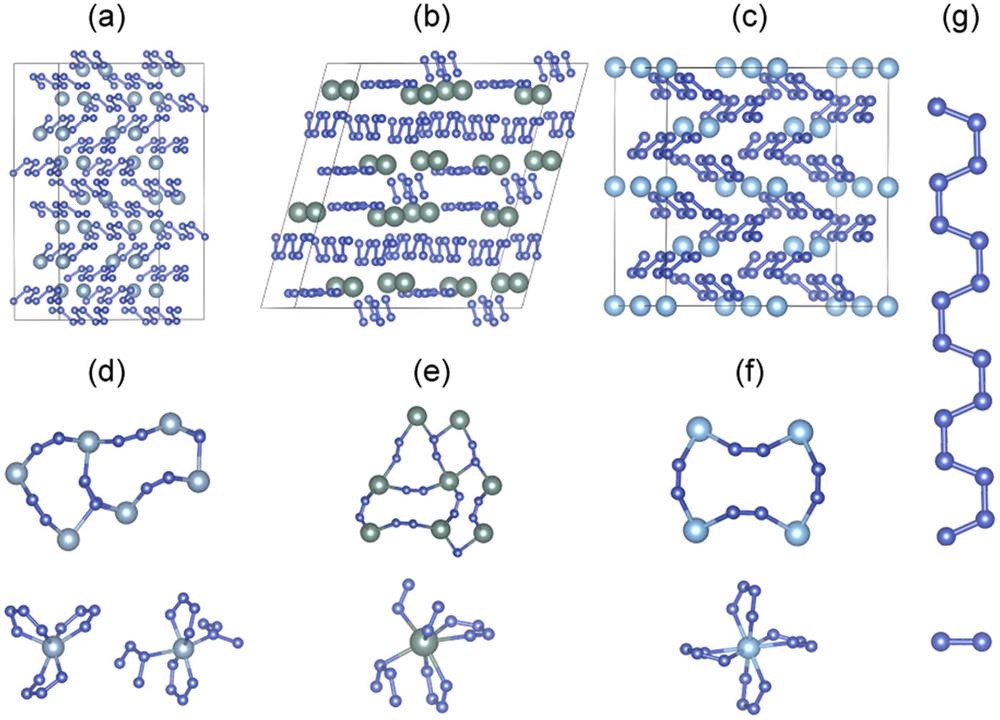
Fig. 1. (a) Isostructural AlN6 and GaN6 of the P 21 phase at 20 GPa. (b) and (c) Crystal structures of P -1-YN6 at 40 GPa and P 4/mnc -TiN8 at 30 GPa, respectively. (d)–(f) geometries of channels formed by metal atoms with different types of N4 coordination. (g) Typical poly-N 4 2 −
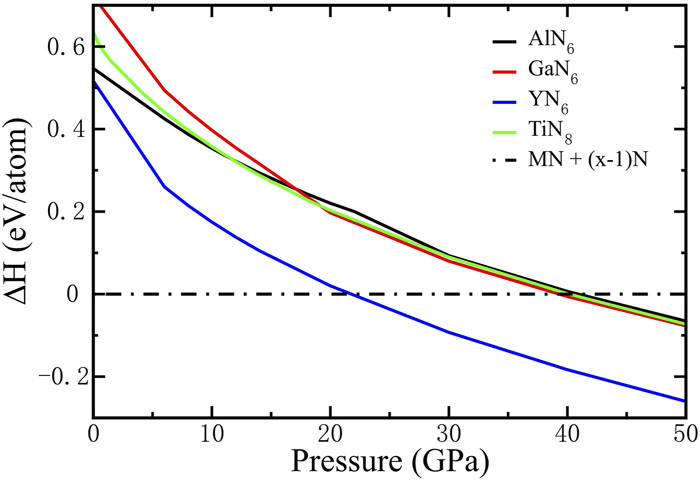
Fig. 2. Enthalpy difference of MNx (M = Al, Ga, Y, and Ti) relative to that of a mixture of ground-state MN (M = Al, Ga, Y, and Ti)24,62–64 structure and bulk nitrogen phase.65
Fig. 3. (a)–(d) Radial distribution functions (RDFs) g (r ) for MNx structures from MD simulations. The nitrogen-to-nitrogen pair (N–N) RDFs at different temperatures are shown as solid lines for (a) P 21-AlN6, (b) P 21-GaN6, (c) P -1-YN6, and (d) P 4/mnc -TiN8. Vertical dashed lines represent the averaged distance between nitrogen atoms in the structures relaxed at 0 K. The inset graphics show the corresponding statistically averaged structures.
Fig. 4. (a)–(d) Density of states (DOS) and (e)–(h) minus projected crystal orbital Hamilton population (−pCOHP) for P 21-AlN6 [(a) and (e)], P 21-GaN6 [(b) and (f)], P -1-YN6 [(c) and (g)], and P 4/mnc -TiN8 [(d) and (h)]. The dashed line indicates the Fermi energy level.
Fig. 5. Sketches of (a) type A and (b) type B chains for N4–metal coordination, and fragment structures of (c) type A and (d) type B poly-N 4 2 −
Fig. 6. Plots of the RDG s vs electron density multiplied by the sign of the second Hessian eigenvalue for (a) P 21-AlN6, (b) P 21-GaN6, (c) P -1-YN6, and (d) P 4/mnc -TiN8.
| ||||||||||||||||||
Table 1. Average −IpCOHP values for MNx.
|
Table 2. Comparison of the detonation properties of MNx structures estimated using the Kamlet–Jacobs empirical equations22,69 with the corresponding experimental values for TNT and HMX.70

Set citation alerts for the article
Please enter your email address



