Meixia XIAO, Miaomiao LI, Erhong SONG, Haiyang SONG, Zhao LI, Jiaying BI. Halogenated Ti3C2 MXene as High Capacity Electrode Material for Li-ion Batteries [J]. Journal of Inorganic Materials, 2022, 37(6): 660
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 37, Issue 6, 660 (2022)
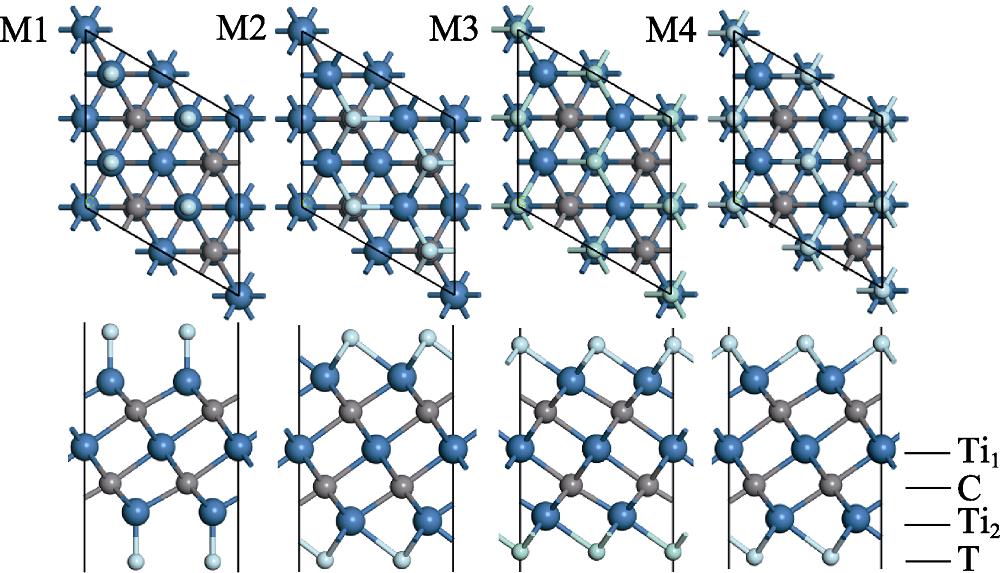
1. Top and side views of M1, M2, M3, and M4 Ti3C2T2 configurations
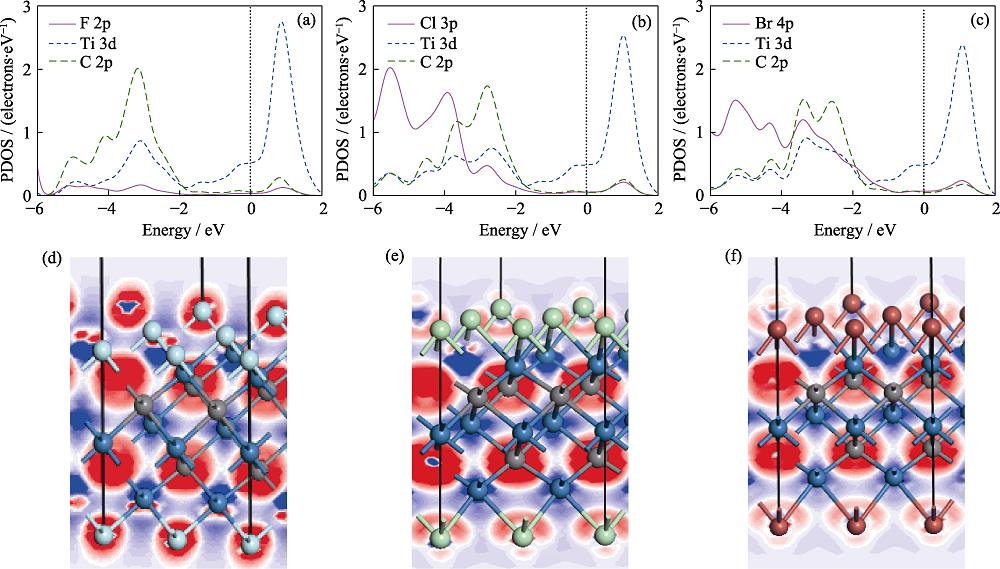
2. Partial density of states (PDOS) and electron density difference of M3-Ti3C2T2
3. Strain energy to strain curves of M3-Ti3C2T2 along (a) x - and (b) y -direction, respectively, and (c) elasticmodulus values in x and y directions
4. Schematic diagram of Li-ion migration path on (a) Ti3C2F2, (b) Ti3C2Cl2 and (c) Ti3C2Br2, and corresponding energy profiles and transition states of Li-ion migration on (d) Ti3C2F2, (e) Ti3C2Cl2 and (f) Ti3C2Br2 surfaces
5. Top and side views of (a) Ti3C2F2, (b) Ti3C2Cl2 and (c) Ti3C2Br2 with the adsorption of multi-layer Liions
6. Partial density of states (PDOS) of M3-Ti3C2T2 electrode with the maximum adsorption of Liions
7. Top and side views of three feasible stacking configurations of double Ti3C2Cl2.
8. (a) Schematic illustration of the diffusion path of Liion, (b) corresponding energy profiles and configurations of transition states, (c) atomic structures and interlayer distances at initial and transition states of D33-Ti3C2Cl2 during interlayer Li-ion migration.
|
Table 1. Formation energy Ef of (eV) Ti3C2T2 with four configurations
|
Table 2. Lattice parameters a of M3-Ti3C2T2 and corresponding bond lengths of lTi2-T and lTi2-C
|
Table 3. Adsorption energy (Eab), charge transfer amount (Δq) and adsorption height (Δh), of Liion for stable M3-Ti3C2T2-Li with Liion adsorbed on Ti3C2T2 surface
|
Table 4. Average open circuit voltage (OCV), adsorption energies of the Liions on the double (E2nd) and triple (E3rd) layers, and the maximum theoretical capacity (CM) of the multi-layer Li-ion adsorption of M3-Ti3C2T2

Set citation alerts for the article
Please enter your email address



