Zhenping Liu, Rong Jiang, Kejing Pang. Highly Sensitive Detection of Copper Ions by Reduced Glutathione Modified Gold Nanocluster Fluorescence Sensor[J]. Laser & Optoelectronics Progress, 2021, 58(14): 1404002
Search by keywords or author
- Laser & Optoelectronics Progress
- Vol. 58, Issue 14, 1404002 (2021)
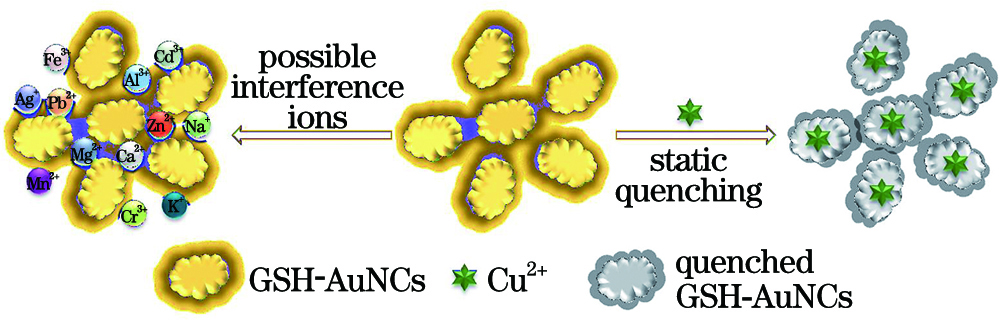
Fig. 1. Principle of Cu2+ detection by GSH-AuNCs fluorescence sensor
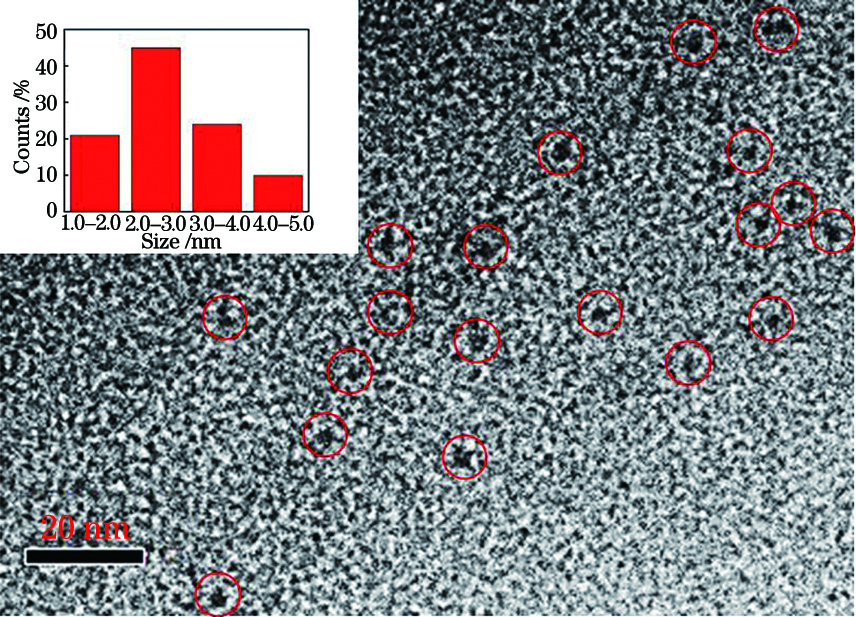
Fig. 2. GSH-AuNCs transmission electron microscope image, illustrated with particle size distribution
Fig. 3. Infrared spectra of GSH-AuNCs
Fig. 4. Fluorescence spectra of GSH-AuNCs
Fig. 5. Relationship between GSH-AuNCs fluorescence intensity and Cu2+ concentration
Fig. 6. Fluorescence attenuation curves of GSH-AuNCs and GSH-AuNCs /Cu2+
Fig. 7. Fluorescence emission spectra of GSH-AuNCs under different wavelengths of excitation light
Fig. 8. Effect of pH on fluorescence intensity of GSH-AuNCs
Fig. 9. Effect of different Cu2+ concentrations on fluorescence intensity of GSH-AuNCs, embedded images a and b are the photos of GSH-AuNCs before and after fluorescence is quenched by Cu2+ under ultraviolet light
Fig. 10. Relationship between Cu2+ concentration and natural logarithm of GSH-AuNCs fluorescence intensity ratios before and after quenching
Fig. 11. Specificity analysis of fluorescence sensor
|
Table 1. Comparison with other detection methods
|
Table 2. Test results of Cu2+ in water samples

Set citation alerts for the article
Please enter your email address



