- Journal of Semiconductors
- Vol. 40, Issue 2, 022101 (2019)
Abstract
1. Introduction
Since the gas sensitivity of the material was discovered, it has been widely concerned in the field of gas detection. The gas sensors have been widely used, including the detection of industrial toxic or explosive gases[
Herein, we synthesized three-dimensional (3D) CuO nanomaterials assembled from nanosheets. Meanwhile, Au nanoparticles were synthesized and the method was similar to the preparation of Ag nanoparticles[
2. Experimental section
2.1. Synthesis process
2.1.1. Preparation of CuO nanomaterial
All the chemical regents (analytical grade) were directly used without further purification. CuO nanomaterials are synthesized by the hydrothermal method, which is similar to the method of Yang and Liu[
2.1.2. Preparation of Au-loaded CuO nanomaterial
For the synthesis of Au nanoparticles, 4.0 g glucose and 0.9 g PVP (K30) were dissolved into 30 mL deionized water, and 4.1 mL HAuCl4solution (0.02 M) was added dropwise into the above solution under vigorous stirring. Then the above solution was transferred into a 50 mL of Teflon-lined stainless steel autoclave, and continuously heated at 180 ºC for 4 h. After the autoclave was quickly cooled by cold water, nano-gold nanoparticles were collected and washed twice with deionized water and ethanol alternately by centrifugation at 9000 rpm. Finally, the Au nanoparticles were dispersed in ethanol with the assistance of ultrasonication.
The CuO powder and Au nanoparticles were dispersed into Ethanol solvent, and the samples were prepared through adjusting the molar ratio of Au to CuO. The obtained product was named pure CuO: 0.5 Au–CuO, 1.0 Au–CuO, and 2.0 Au–CuO (corresponding to the ratio 0, 0.5‰, 1.0‰, and 2.0‰, respectively.)
2.2. Characterization
The as-synthesized CuO nanomaterial and Au nanoparticles were characterized by X-ray diffraction (XRD, Rigaku D/max-2400) with copper Kα radiation. The microscopic images of the products were obtained by scanning electron microscopy (SEM, Hitachi S-4800).
2.3. Fabrication and measurement of gas sensors
The gas sensor was fabricated using ceramic tube adjacent thermal structure in accordance with literature[
The response time (Tres) and recovery time (Trec) were defined as the time required for the resistance to reach 90% of the final equilibrium value in the case of adsorption and desorption, respectively. The concentration of the target gas in the chamber was calculated by Eq. (2):
where C (ppm) is the target gas concentration in the chamber, 24.5 is the gas molar volume at 25 ºC 101 kPa, ρ (g/mL) is the density of volatile liquid, V1 (μL) is the volume of volatile liquid, φ is the actual content of the target substance in the liquid, M (g/mol) is the molar mass of the target substance, and V2 (L) is the volume of the chamber.
3. Results and discussion
3.1. X-ray diffraction and morphology analysis
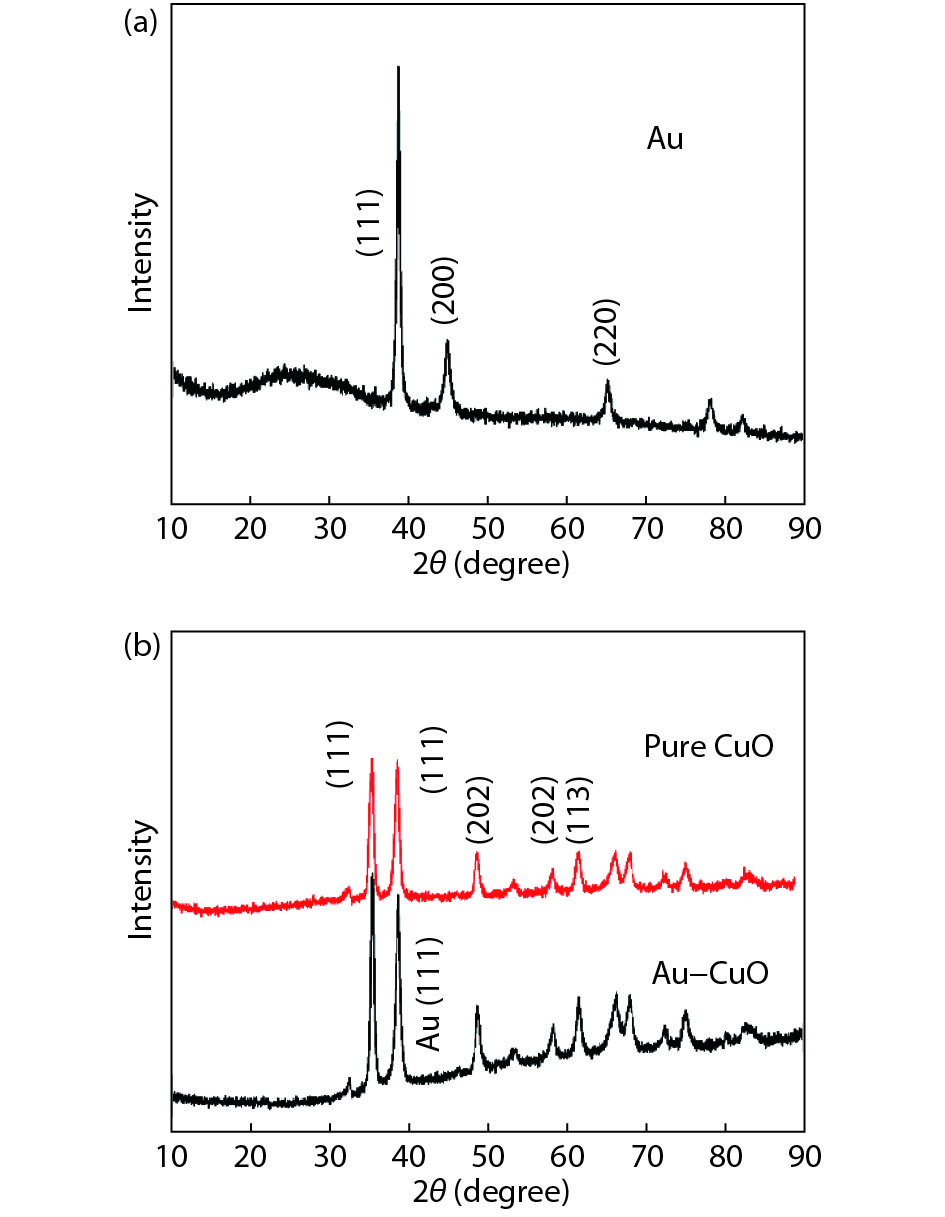
The material composition and phase structure of the samples were determined by XRD. Fig. 1(a) shows the XRD pattern of the as-synthesized Au nanoparticles, all diffraction peaks were matched with JCPDS 04-0784 of Au. The peak of angle 20º to 30º was the amorphous diffraction peak of glass substrate. The peaks centered at 38.18º, 44.39º, and 64.58º were indexed to the (111), (200), and (220) planes of Au, respectively. The sample was identified as the Au. Fig. 1(b) shows the XRD pattern of the as-synthesized Au–CuO and pure CuO nanomaterials, all diffraction peaks were matched with JCPDS 48-1548 of CuO for the pure CuO samples. Such as the diffraction peaks of the as-synthesized CuO nanomaterial at 2θ 35.54º, 38.71º, 48.72º, and 61.52º were indexed to the (11
![]()
Figure 1.(Color online) XRD patterns of (a) the Au samples, and (b) the Au–CuO and pure CuO samples.
The morphologies of the as-synthesized CuO nanomaterial and Au nanoparticles were characterized by SEM, as shown in Fig. 2. The panoramic morphology of CuO is shown in Fig. 2(a). The prepared CuO was assembled regularly from many nanosheets. The size of the entire structure is about 5 µm. Some unassembled nanosheets can be found as well. Fig. 2(b) is a high magnification of the local structure, the assembly of nanosheets can be clearly discovered. The thickness of nanosheets is about 100 nm. The panoramic morphology of Au nanoparticles is shown in Fig. 2(c). Au nanoparticles were well dispersed on the substrate with the size ranging from 50 to 200 nm. The Au nanoparticles were coated with a layer of polymer. Fig. 2(d) is a partial photomicrograph of the Au-loaded CuO. The Au nanoparticles coated by polymer attached to the CuO nanoplatelets.
![]()
Figure 2.SEM micrographs of (a,b) the CuO nanomaterial, (c) the Au nanoparticles, and (d) the Au-loaded CuO nanomaterial.
3.2. Gas sensing properties
In order to research the potential applications of the Au-loaded CuO nanomaterials, pure CuO gas sensors and different concentrations of Au-loaded CuO gas sensors were fabricated. The gas sensing properties were investigated. The gas sensitivity of metal oxide semiconductors is mainly reflected by the change of material resistance[
![]()
Figure 3.(Color online) (a) Relation of the gas response with operating temperature for the pure CuO, and 0.5‰, 1.0‰, and 2.0‰ Au-loaded CuO gas sensors to 500 ppm of ethanol. (b) The response of the sensors based on pure CuO and Au-loaded CuO to different concentrations of ethanol at 160 ºC.
Figs. 4(a) and 4(b) exhibit the response and recovery behavior of the gas sensors based on pure CuO and 1.0‰ Au-loaded CuO to different concentrations of ethanol at 160 ºC, respectively. Corresponding to Fig. 3(b), the concentrations of ethanol were regulated from 10 to 800 ppm. It can be found that the higher the ethanol concentration, the larger the gas sensors resistance in the gas. Figs. 4(c) and 4(d) exhibit the response time (Tres) and the recovery time (Trec) of the gas sensors based on pure CuO and 1.0‰ Au-loaded CuO to 400 ppm of ethanol at 160 ºC, respectively. For the pure CuO gas sensor, the response time and the recovery time was 18 and 12 s, respectively. For the 1‰ Au-loaded CuO gas sensor, the response time and the recovery time were 31 s and 32 s, respectively. Compared with the pure CuO gas sensor, the response time and the recovery time of the 1‰ Au-loaded CuO gas sensor became longer.
![]()
Figure 4.(Color online) (a, b) Response and recovery behavior of the gas sensors based on pure CuO and 1.0‰ Au-loaded CuO to different concentrations of ethanol at 160 ºC. (c, d) The response time (
Note the response behavior of the pure CuO gas sensor to different concentrations of ethanol in Fig. 4. When the sensor was exposed to ethanol, the resistance increased rapidly and then decreased slightly. Subsequently, the resistance increased again. The Au-loaded CuO gas sensor also had similar behavior to a low concentration of ethanol. The prepared CuO was assembled from nanosheets. So, the nanosheets can be regarded as the basic unit, and the whole gas sensitive materials were made up by these basic units in series and parallel (ignoring the effects of grain boundary barriers). The basic unit can reflect the overall situation of gas sensitive materials. Fig. 5 shows the equivalent diagram of the resistance of Au-loaded CuO nanosheet. The equivalent resistance of the Au-loaded CuO nanosheet is R, which can also be seen as the parallel of the resistance R1 and the resistance R2. The R1 is the surface resistance that mainly participated in adsorption and desorption of gases, while the R2 is the bulk resistance away from the surface. The process of gas response could be divided into three stages. (i) Initial adsorption and reaction. When the CuO was exposed to ethanol gas, the ethanol molecules were adsorbed on the surface of the material, and a large number of holes recombined with electrons on the surface of material. It gave rise to the increase of the R1. At this time, the concentration of the holes inside the material did not change significantly. It can be considered that the R2 was unchanged. Therefore, the total resistance R increased. (ii) The diffusion to the surface of the holes. The rate of holes diffusion was higher than that of the electrons produced by the adsorption of ethanol. It caused the increase of the surface holes concentration and the obvious reduction of R1. Since the change of the concentration of holes inside the CuO was very small, the R2 had no significant increase. It eventually led to the reduction of the total resistance R. (iii) Dynamic balance of the holes. The final result was the reduction of the concentration of the total holes of CuO and the increase of the R. However, for the Au-loaded CuO gas sensors, the catalysis of gold could accelerate the rate of release of electrons in the adsorption process of ethanol. In the second stage of the response process of the Au-loaded CuO gas sensors toward the high concentration of ethanol gas, the electrons release rate was higher than the diffusion efficiency of the holes to the material surface. So, the R1 would keep rising. As a result, it led to the continuous increase in total resistance. However, for low concentration of ethanol gas, even if there was a catalytic effect, the rate of generation of electrons would be lower than the rate of transport of the holes to the surface layer of the CuO. As a result, the total resistance R decreased in the second stage of the response process.
![]()
Figure 5.(Color online) Equivalent diagram of the resistance of the Au-loaded CuO nanosheet.
Selectivity is an important performance parameter of the gas sensors. Therefore, the response of gas sensors to five kinds of common organic gases with 400 ppm was researched. Fig. 6 shows the response of gas sensors (based on the pure CuO, 0.5‰ Au-loaded CuO, 1.0‰ Au-loaded CuO, and the 2.0‰ Au-loaded CuO) to 400 ppm of ethanol, methanol, acetone, formaldehyde, and xylene (C2H5OH, CH3OH, CH3COCH3, HCHO, and C8H10) at 160 ºC. The results showed that the pure CuO gas sensor had less disparity for the gas response to ethanol, methanol, acetone, and formaldehyde. Meanwhile, the response to xylene was significantly lower than the response to other gases. On the other hand, all the Au-loaded CuO gas sensors had a higher response to ethanol and xylene than the pure CuO gas sensor. It is noticed that the response of all the Au-loaded CuO gas sensors to methanol, acetone, and formaldehyde decreased compared with the pure CuO gas sensor. With the increasing of the concentration of Au nanoparticles, the decrease was more pronounced. As a result, the Au-loaded CuO gas sensors had obvious disparity for gas response to ethanol compared with other gases, especially the gas sensors based on 1.0‰ Au-loaded CuO and 2.0‰ Au-loaded CuO. It was mainly ascribed to the following two aspects. (i) Nano-gold has been widely used in physical and chemical processes as a kind of efficient catalyst. Some previous works have reported that Au can improve the gas sensing properties of metal oxide semiconductor[
![]()
Figure 6.(Color online) Response of the gas sensors based on the pure CuO and 0.5‰, 1.0‰, and 2.0‰ Au-loaded CuO to 400 ppm of various test gases at 160 ºC.
Fig. 7 shows the response of the sensors based on the pure CuO and 0.5‰, 1.0‰, and 2.0‰ Au-loaded CuO to different concentrations of formaldehyde at 160 ºC. The concentrations of formaldehyde were regulated from 10 to 800 ppm. The response of the Au-loaded CuO gas sensors was significantly decreased compared with the pure CuO gas sensor at the same concentration. For 400 ppm of formaldehyde, the response of the 0.5‰, 1.0‰, and 2.0‰ Au-loaded CuO gas sensors decreased to 86%, 72%, and 58% of the pure CuO gas sensor, respectively.
![]()
Figure 7.(Color online) Response of the gas sensors based on the pure CuO and 0.5‰, 1.0‰, and 2.0‰ Au-loaded CuO to different concentrations of formaldehyde at 160 ºC.
Stability (or repeatability) is one of the important parameters of gas sensors. Sometimes the gas sensor may have poor stability due to incomplete desorption of gas molecules and poor thermal stability of materials. So, the stability of the gas sensor was investigated by testing 300 ppm of ethanol seven times at the same conditions. Fig. 8 shows the characteristic curve of the response and recovery. There was no obvious change in response time and recovery time. It indicated that the rate of adsorption and desorption had not changed obviously. The average response was 6.4, and the variance of the response was 0.008. It shows that the Au-loaded CuO gas sensors have good stability.
![]()
Figure 8.(Color online) Seven cycles of response-recovery to 300 ppm of ethanol of the 2.0‰ Au-loaded CuO gas sensor at 160 ºC.
4. Conclusions
In summary, the 3D nano-CuO and the Au nanoparticles were synthesized by the hydrothermal method respectively. The Au-loaded CuO gas sensors were fabricated. It is found that the Au-loaded CuO gas sensors enhanced the selectivity to ethanol by increasing the response to ethanol while reducing the response to methanol, acetone, and formaldehyde. At the optimum operating temperature of 160 ºC, the response of 400 ppm of ethanol increased by 1.8 times through the load of 1.0‰ gold. It is noticed that the response of 400 ppm of methanol, acetone, and formaldehyde decreased to 61%, 66%, and 58% respectively through the load of 2.0‰ gold. Meanwhile, it is not hard to find that the Au-loaded CuO gas sensors had obvious disparity for the gas response to ethanol compared with other test gases. Furthermore, the Au-loaded CuO gas sensors had good stability. The Au nanoparticles prepared with PVP as stabilizer have great potential and promising applications in improving the selectivity of gas sensors for ethanol.
Acknowledgements
Project supported by the Provincial Natural Science Foundation of Gansu (No. 1606RJZA026), the Scientific and Technological Project of Chengguan District of Lanzhou (No. 2016-2-4), and Institute of Sensor Technology, Gansu Academy of Science.
References
[1] H Gong, J Q Hu, J H Wang et al. Nano-crystalline Cu-doped ZnO thin film gas sensor for CO. Sensor Actuat B, 115, 247(2006).
[2] J Zhou, P Li, S Zhang et al. Zeolite-modified microcantilever gas sensor for indoor air quality control. Sensor Actuat B, 94, 337(2003).
[3] K Kawamura, K Kerman, M Fujihara et al. Development of a novel hand-held formaldehyde gas sensor for the rapid detection of sick building syndrome. Sensor Actuat B, 105, 495(2005).
[4] W Tsujita, A Yoshino, H Ishida et al. Gas sensor network for air-pollution monitoring. Sensor Actuat B, 110, 304(2005).
[5] H Huang, J Zhou, S Chen et al. A highly sensitive QCM sensor coated with Ag+-ZSM-5 film for medical diagnosis. Sensor Actuat B, 101, 316(2004).
[6] E Abad, S Zampolli, S Marco et al. Flexible tag microlab development: gas sensors integration in RFID flexible tags for food logistic. Sensor Actuat B, 127, 2(2007).
[7] T Ozawa, Y Ishiguro, K Toyoda et al. Detection of decomposed compounds from an early stage fire by an adsorption/combustion-type sensor. Sensor Actuat B, 108, 473(2005).
[8] N Dossi, R Toniolo, A Pizzariello et al. An electrochemical gas sensor based on paper supported room temperature ionic liquids. Lab Chip, 12, 153(2012).
[9] B Liu, H Yang, H Zhao et al. Synthesis and enhanced gas-sensing properties of ultralong NiO nanowires assembled with NiO nanocrystals. Sensor Actuat B, 156, 251(2011).
[10] J Y Shen, M D Wang, Y F Wang et al. Iron and carbon codoped WO3 with hierarchical walnut-like microstructure for highly sensitive and selective acetone sensor. Sensor Actuat B, 256, 27(2018).
[11] X Li, Y Zhao, X Wang et al. Reduced graphene oxide (rGO) decorated TiO2 microspheres for selective room-temperature gas sensors. Sensor Actuat B, 230, 330(2016).
[12] W Tan, J Tan, L Li et al. Nanosheets-assembled hollowed-out hierarchical Co3O4 microrods for fast response/recovery gas sensor. Sensor Actuat B, 249, 66(2017).
[13] C Liu, H Gao, L Wang et al. Facile synthesis and the enhanced sensing properties of Pt-loaded α-Fe2O3 porous nanospheres. Sensor Actuat B, 252, 1153(2017).
[14] S Wang, J Cao, W Cui et al. Oxygen vacancies and grain boundaries potential barriers modulation facilitated formaldehyde gas sensing performances for In2O3 hierarchical architectures. Sensor Actuat B, 255, 159(2018).
[15] N D Khoang, D D Trung, N Van Duy et al. Design of SnO2/ZnO hierarchical nanostructures for enhanced ethanol gas-sensing performance. Sensor Actuat B, 174, 594(2012).
[16] H J Park, N J Choi, H Kang et al. A ppb-level formaldehyde gas sensor based on CuO nanocubes prepared using a polyol process. Sensor Actuat B, 203, 282(2014).
[17] C Dong, X Liu, X Xiao et al. Combustion synthesis of porous Pt-functionalized SnO2 sheets for isopropanol gas detection with a significant enhancement in response. J Mater Chem A, 2, 20089(2014).
[18] F Wang, H Li, Z Yuan et al. A highly sensitive gas sensor based on CuO nanoparticles synthetized via a sol-gel method. RSC Adv, 6, 79343(2016).
[19] H Gao, H Jia, B Bierer et al. Scalable gas sensors fabrication to integrate metal oxide nanoparticles with well-defined shape and size. Sensor Actuat B, 249, 639(2017).
[20] C Yang, F Xiao, J Wang et al. 3D flower- and 2D sheet-like CuO nanostructures: microwave-assisted synthesis and application in gas sensors. Sensor Actuat B, 207, 177(2015).
[21] D R Miller, S A Akbar, P A Morris. Nanoscale metal oxide-based heterojunctions for gas sensing: a review. Sensor Actuat B, 204, 250(2014).
[22] C Feng, X Kou, B Chen et al. One-pot synthesis of In doped NiO nanofibers and their gas sensing properties. Sensor Actuat B, 253, 584(2017).
[23] C Wang, J Liu, Q Yang et al. Ultrasensitive and low detection limit of acetone gas sensor based on W-doped NiO hierarchical nanostructure. Sensor Actuat B, 220, 59(2015).
[24] X Hu, Z Zhu, C Chen et al. Highly sensitive H2S gas sensors based on Pd-doped CuO nanoflowers with low operating temperature. Sensor Actuat B, 253, 809(2017).
[25] S Park, Z Cai, J Lee et al. Fabrication of a low-concentration H2S gas sensor using CuO nanorods decorated with Fe2O3 nanoparticles. Mater Lett, 181, 231(2016).
[26] L Wang, B Han, Z Wang et al. Effective improvement of sensing performance of amperometric NO2 sensor by Ag-modified nano-structured CuO sensing electrode. Sensor Actuat B, 207, 791(2015).
[27] J S Lee, A Katoch, J H Kim et al. Effect of Au nanoparticle size on the gas-sensing performance of p-CuO nanowires. Sensor Actuat B, 222, 307(2016).
[28] S Kim, S Park, S Park et al. Acetone sensing of Au and Pd-decorated WO3 nanorod sensors. Sensor Actuat B, 209, 180(2015).
[29] S Zhang, P Song, J Zhang et al. Highly sensitive detection of acetone using mesoporous In2O3 nanospheres decorated with Au nanoparticles. Sensor Actuat B, 242, 983(2017).
[30] J Zhang, P Song, Z Li et al. Enhanced trimethylamine sensing performance of single-crystal MoO3 nanobelts decorated with Au nanoparticles. J Alloy Compd, 685, 1024(2016).
[31] Z Li, N Wang, Z Lin et al. Room-temperature high-performance H2S sensor based on porous CuO nanosheets prepared by hydrothermal method. ACS Appl Mater Inter, 8, 20962(2016).
[32] A Umar, A A Alshahrani, H Algarni et al. CuO nanosheets as potential scaffolds for gas sensing applications. Sensor Actuat B, 250, 24(2017).
[33] X Liu, Y Sun, M Yu et al. Enhanced ethanol sensing properties of ultrathin ZnO nanosheets decorated with CuO nanoparticles. Sensor Actuat B, 255, 3384(2018).
[34] X M Sun, Y D Li. Ag@C core/shell structured nanoparticles: controlled synthesis, characterization, and assembly. Langmuir, 21, 6019(2005).
[35] H Yang, Z H Liu. Preparation and properties of flower-like CuO nanostructures. J Shaanxi Normal University, 37, 60(2009).
[36] X Liu, N Chen, B Han et al. Nanoparticle cluster gas sensor: Pt activated SnO2 nanoparticles for NH3 detection with ultrahigh sensitivity. Nanoscale, 7, 14872(2015).
[37] S Bai, X Liu, D Li et al. Synthesis of ZnO nanorods and its application in NO2 sensors. Sensor Actuat B, 153, 110(2011).
[38] G S Guo, K W Lin, D M Han et al. Functionalization of flower-like ZnO nanostructures with Au@CuO nanoparticles for detection of ethanol. IEEE Sens J, 14, 1797(2014).
[39] X Li, W Feng, Y Xiao et al. Hollow zinc oxide microspheres functionalized by Au nanoparticles for gas sensors. RSC Adv, 4, 28005(2014).
[40] X Li, X Zhou, H Guo et al. Design of Au@ZnO yolk-shell nanospheres with enhanced gas sensing properties. ACS Appl Mater Inter, 6, 18661(2014).
[41] L Wang, S Wang, M Xu et al. A Au-functionalized ZnO nanowire gas sensor for detection of benzene and toluene. Phys Chem Chem Phys, 15, 17179(2013).
[42] J Guo, J Zhang, M Zhu et al. High-performance gas sensor based on ZnO nanowires functionalized by Au nanoparticles. Sensor Actuat B, 199, 339(2014).
[43] C Dong, Q Li, G Chen et al. Enhanced formaldehyde sensing performance of 3D hierarchical porous structure Pt-functionalized NiO via a facile solution combustion synthesis. Sensor Actuat B, 220, 171(2015).
[44] J Park, X Shen, G Wang. Solvothermal synthesis and gas-sensing performance of Co3O4 hollow nanospheres. Sensor Actuat B, 136, 494(2009).
[45] C Liu, ST Navale, ZB Yang et al. Ethanol gas sensing properties of hydrothermally grown α-MnO2 nanorods. J Alloy Compd, 727, 362(2017).
[46] L Guan, H Pang, J Wang et al. Fabrication of novel comb-like Cu2O nanorod-based structures through an interface etching method and their application as ethanol sensors. Chem Commun, 46, 7022(2010).
[47] M Parmar, R Bhatia, V Prasad et al. Ethanol sensing using CuO/MWNT thin film. Sensor Actuat B, 158, 229(2011).
Set citation alerts for the article
Please enter your email address



