- Journal of Semiconductors
- Vol. 41, Issue 5, 051202 (2020)
Abstract
1. Introduction
Since the first organic–inorganic hybrid perovskite solar cell (PSCs) was proposed by Miyasaka’s group[
CsPbX3 is a promising candidate to conquer these problems because Cs+ is the most feasible inorganic cation to replay volatility and hygroscopic A-site with suitable tolerance factor[
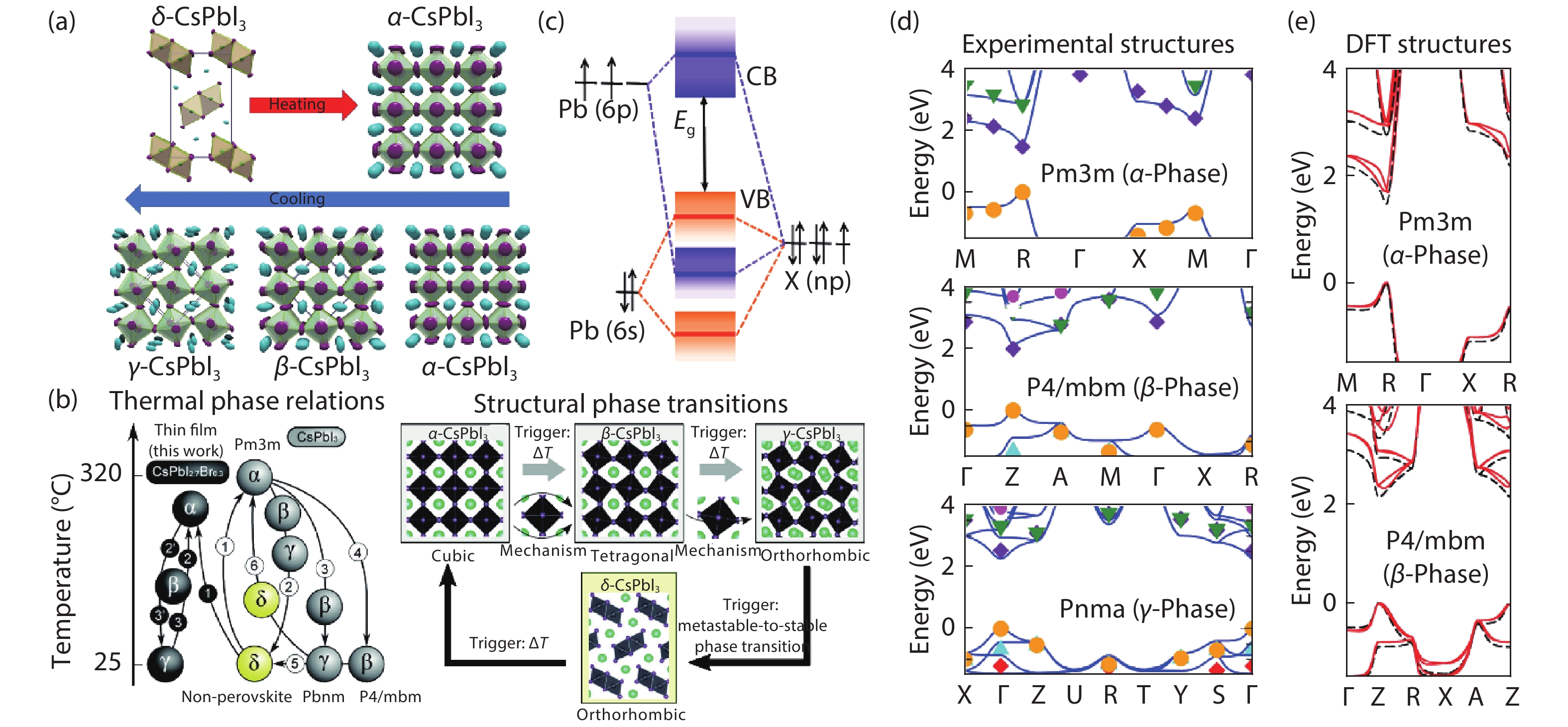
CsPbI3 has four different phases (cubic (α), tetragonal (β), orthorhombic (γ) and non-perovskite yellow (δ) phase) and each phase transforms under different temperatures[
In this review, we aim to summarize the latest works about CsPbI3 PSCs based on HI hydrolysis-derived intermediate. First, we briefly review the different crystal and electronic structures of CsPbI3. We then trace the history and disputes of HI hydrolysis-derived intermediate to make this review more logical. Afterward, we highlight the functions of HI hydrolysis-derived intermediate, and systematically summarize some advanced works about HI hydrolysis-derived intermediate on CsPbI3 PSCs. Finally, present issues and outlines are discussed to further increase the CsPbI3 PSCs performance.
2. Crystal/electronic structure
Photo-electric properties (e.g., optical transitions, charger transfer) are greatly related to crystal and electronic properties (e.g., phase transition, energy band)[
2.1. Crystal structure
The CsPbI3 perovskite structure can be described as: Pb-site and I-site ion form a corner sharing [PbI6]4– octahedron, while the Cs cation resides in the cuboctahedral cavities[
![]()
Figure 1.(Color online) (a) The structure and transition of CsPbI3 phases versus temperature. Reproduced with permission[
The detailed transition temperature between each phase was researched by Even et al., based on density functional theory (DFT) analyzation. The increasing thermal parameters of I– tended to strength the dynamic motion of the corner-connected [PbI6/2]– octahedral, which further induced a change of the unit cell volume and made δ-phase transformed to α-phase at 595 K. Then, with dynamic states gradually relaxing, α-phase transitions to β-phase at 539 K; β-phase transitions to γ-phase at 425 K; finally γ-phase turns to yellow non-perovskite phase (δ-phase) at RT[
The different stability of each phases can be ascribed to the different dissociated energies. The dissociation energy from CsPbI3 to CsI and PbI2 for α-, γ-, and δ-phase are 0.04, –0.09 and –0.16 eV, respectively. δ-phase CsPbI3 shows a small Pb–I–Pb bond angel (95.09° and 91.40°) than α-phase CsPbI3 (180°) and γ-phase CsPbI3 (154.74°), which reduces the orbital overlap between Pb and I atoms and also makes δ-phase CsPbI3 with a deeper defect transition energy level than α-, γ-phase. This indicates that δ-phase is the most stable phase because of its lowest dissociation energy[
2.2. Electronic structure
The valence band maximum (VBM) of CsPbI3 perovskite is constituted of antibonding hybridized Pb 6s and X np orbitals, among which X np takes the lead. However, Pb 6p is in the dominant place of conduction band minimum (CBM), as shown in Fig. 1(c)[
The calculated electronic of α-, β- and γ-CsPbI3 are depicted in Figs. 1(d) and 1(e)[
3. The functions of HI hydrolysis-derived intermediate
We summarize the performance of CsPbI3 PSCs after introducing HI hydrolysis-derived intermediate in Table 1 (sPCE is the stable PCE). Its main functions can be summarized as following:
1) Reducing crystallization energy barrier in low temperature fabrication;
2) Increasing iodide coordination numbers to decrease structural disorder, modifying structure and forming higher-order iodoplumbate complexes;
3) Slowing down the rapid crystalline process and obtaining high-quality CsPbI3 film;
4) Inducing strain to generate distorted metastable phase (β- and γ- CsPbI3);
5) Modifying the band gap of perovskites films.
4. History and disputes of HI hydrolysis-derived intermediate
The solution one-step method has advantages of simple, convenience and facile process, and can also be compatible roll-to-roll fabrication technology[
4.1. HI
In the early stage, researchers focused on using HI additive in CsPbI3 PSCs fabrication to cause a microstrain and induce a low temperature phase transition process. Meanwhile, extra halides in HI precursor solution tended to fill the vacancies of perovskites, resulting in change of metal–halogen–metal bond connectivity, and consequently cell volumes and optical bandgap[
In 2015, HI was first used as an additive in CsPbI3 PSCs. Snaith et al. introduced a small amount of HI in the precursor solution before spin-coating. They found that HI additive could change the solubility of precursor materials and induce a strain to lower the temperature phase transition. Then, strain triggered small crystals appearing and significantly stabilized its structure in RT, as shown in Fig. 2(a)[
![]()
Figure 2.(Color online) (a) The diagrammatic of HI fabricated CsPbI3. Reproduced with permission[
4.2. PbI2·xHI or HPbI3
In addition to HI additive, HI hydrolysis-derived intermediate is more effective because it eliminates water in the HI solution and is an intermediate compound to increase perovskite crystallinity[
Zhu et al. introduced HPbI3 into the CsPbI3 PSCs and assisted with a triple cation NH3+C2H4NH2+C2H4NH3+ (named as DETA3+) to further stabilize the α-CsPbI3 perovskite phases[
4.3. PbI2·xDMAI or DMAPbI3
Kanatzidis et al. recently claimed that HPbI3 did not exist and was replaced by a compound of DMAPbI3, which generated through DMF hydrolysis in HI solution. Importantly, they pointed out that some early reports of inorganic perovskite are actually the hybrid perovskite. They found that DMAPbI3 possessed a larger tolerance factor and mixing with Cs+ could adjust tolerance factor (t) of the compounds (Cs1−xDMAxPbI3) toward an ideal factor (t, 0.9–1). Finally, they achieved a champion PCE of 12.62% in Cs1−xDMAxPbI3-based PSCs, as shown in Fig. 2(d)[
![]()
Figure 3.(Color online) (a) Schematic illustration the fabrication process of Cs
Zhao et al. used PbI2·xDMAI to fabricate CsPbI3 PSCs recently, and they concluded that the fabricated perovskites are actually all inorganic composition because the organic ion DMA+ are easily lost during the high-temperature (210 °C) annealing process[
Our groups also confirmed this conclusion. We synthesized a series of intermediate compounds (DMAI and DMAPbI3) by different ratio of HI/DMF, and used them to fabricate CsPbI3 PSCs. After detailed analysis, we found that the major component of CsPbI3 was still inorganic in this reaction route. Most of DMA+ organic molecules lost during the annealing process, and only a small amount of DMA+ remained to stabilize perovskite structure. Excessive DMA+ interacted with Pb2+ to further passivate CsPbI3 surface, as shown in Fig. 3(d)[
In conclusion, the organic molecule DMAI mainly influence the crystallization kinetics and perovskite phase. During the annealing process, DMAI will sublimate quickly, change the rate of crystallization and form metastable (β- and γ-) phase based CsPbI3. The controllable crystallization kinetics and stable (β- and γ-) phase are beneficial to morphology and stability of perovskite, respectively. Besides, DMA+ (2.72 Å) possesses a larger ionic radius than Cs+ (1.88 Å)[
5. Applying HI hydrolysis-derived intermediate in CsPbI3 PSCs
5.1. α-phase CsPbI3 based PSCs
As we discussed earlier, HI hydrolysis-derived intermediate showed a lot of advantages in high-quality film fabrication and device performance. Importantly, perovskite films with better crystallinity, morphology, and higher range of absorption are the foundation of efficiency.
The first working α-CsPbI3 PSCs with a PCE of 2.9% was fabricated in low temperature (100 °C) by Snaith and his cooperators via a small amount HI additive adding[
![]()
Figure 4.(Color online) (a) The detail information of PbI2.HI and PbI2 fabricated perovskite, inserted pictures are their digital photos. Reproduced with permission[
Compared with HI, the absence of H2O molecules in HI hydrolysis-derived intermediate can optimize the perovskite crystallinity and morphology. Chen and his cooperators replaced PbI2 with HPbI3 in fabricating stable α-CsPbI3 film. They found that the bandgap was shifted from 1.72 to 1.68 eV owing to formation of tensile lattice strain. Finally, a HTL free α-CsPbI3 was obtained with a higher PCE of 9.5%. Besides, the optimal device showed enhanced stability, which maintained 90% of its initial PCE under illumination for more than 3000 h in dry environment, as shown in Fig. 4(b)[
5.2. Metastable (β- and γ-) phase CsPbI3 based PSCs
Recently, the CsPbI3 films fabricated by HI hydrolysis-derived intermediate were proved metastable phases (combined β-phase CsPbI3 with γ-phase CsPbI3).
The β-phase CsPbI3 can also be formed at low temperature and show more stable perovskite structure than α-phase one. However, it is difficult to deposit and stabilize its perovskite structure[
![]()
Figure 5.(Color online) (a) Schematic illustration of CHI crack-filling interface engineering. Reproduced with permission[
The γ-phase CsPbI3 is the most stable black phase because of its lowest dissociation energy[
In our recent research, we reported the synergistic effect of HI and PEAI additives, where HI transferred to an intermediate (HPbI3+x) to fabricate distorted black phase-based CsPbI3 thin films and PEAI induced a steric effects to avoid phase transition. It is noteworthy that the best device maintained 92% of its initial PCE for 60 days storage in ambient (RH ~ 20%–30%, 25 °C), while the reference one degraded to 0.65% in the same condition for 8 days, as shown in Fig. 5(d)[
One of the notorious problems to limit CsPbI3 performance is the lower JSC compared with hybrid one. Thus, we have also developed several strategies to increase its JSC, such as harvesting short wavelength ultraviolet light (UV-light) or near-infrared (NIR) light, and designing device structure to capture light. First, we developed a downconversion nanoparticles (DCNPs) nitrogen-doped graphene quantum dots (N-GQDs) as an energy-down-shift to harvest the short wavelength (< 350 nm) UV-light. After combining it with HPbI3-formed γ-CsPbI3, the optimal device showed an improved short circuit current density (JSC) from 18.67 to 19.15 mA/cm2, with an increase of 2.57%. Meanwhile, its performance was greatly increased 3.15%, from 15.53% to 16.02%[
5.3. Low dimension CsPbI3 based PSCs
Reducing dimension can further increase the stability of CsPbI3 PSCs because reducing materials dimension can lead to more symmetric crystal structure and show a smaller surface energy[
However, the poor solubility of CsX in the precursor solution would severely limited the thickness of CsPbI3 film and influence the light absorption. Chen et al. used HPbX3 and CsAc as new precursor to overcome the poor solubility of Cs+ precursor and fabricate α-CsPbX3 with optimal thickness. They introduced phenylethylammonium iodide (PEAI) to HPbX3 and CsAc system and further controlled the dimension of CsPbX3 from three dimension (3D) to two dimension (2D). Finally, a champion PCE of 12.4% in 2D CsPbI3 perovskite was obtained, and maintained 93% of its initial PCE in ambient for 40 days, as shown in Fig. 6(a)[
![]()
Figure 6.(Color online) (a) The structure and decomposition energies of different n values PEA2Cs
Pradhan et al. fabricated stable CsPbI3 nanocrystals (NCs) with superior stability by using a higher temperature (260 °C) than usual (160 °C), and adding olelyamine (OLA) and HI respectively in the reaction process (noted that only OLA or HI are less efficient). Taking the NMR analyzation into account, they found that higher temperature helped the OLA+ ligands to occupy the Cs+ position on the surface and further stabilized its structure[
6. Prospects and outlook
Although lots of advanced works about HI hydrolysis-derived intermediate have been done to boost PCE of CsPbI3 PSCs, its PCE still far behind the hybrid ones. Therefore, we need to analyze the urgent problems that remain and develop corresponding strategies to improve the performance of CsPbI3 perovskites.
In conclusion, CsPbI3 perovskite, particularly the metastable phases (β- and γ-Phase CsPbI3), is a promising material to replace the unstable hybrid perovskite. Besides, CsPbI3 PSCs with suitable bandgap make it more suitable to apply in tandem solar cells and commercialization. Based on these advantages, we conclude that CsPbI3 PSCs maybe the mainstream research direction in the near future, and we should adopt a positive attitude to it.
Acknowledgements
This work was funded by the National Natural Science Foundation of China (51902148, 61704099 and 51801088), the Fundamental Research Funds for the Central Universities (lzujbky-2020-61, lzujbky-2019-88 and lzujbky-2020-kb06), and the Special Funding for Open and Shared Large-Scale Instruments and Equipments of Lanzhou University (LZU-GXJJ-2019C023 and LZU-GXJJ-2019C019).
References
[1] A Kojima, K Teshima, Y Shirai et al. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J Am Chem Soc, 131, 6050(2009).
[2]
[3] J Jiang, Q Wang, Z Jin et al. Polymer doping for high-efficiency perovskite solar cells with improved moisture stability. Adv Energy Mater, 8, 1701757(2018).
[4] J Jiang, Z Jin, F Gao et al. CsPbCl3-driven low-trap-density perovskite grain growth for > 20% solar cell efficiency. Adv Sci, 5, 1800474(2018).
[5] C Wehrenfennig, G E Eperon, M B Johnston et al. High charge carrier mobilities and lifetimes in organolead trihalide perovskites. Adv Mater, 26, 1584(2014).
[6] W Hu, H Cong, W Huang et al. Germanium/perovskite heterostructure for high-performance and broadband photodetector from visible to infrared telecommunication band. Light: Sci Appl, 8, 106(2019).
[7] V D'Innocenzo, G Grancini, M J P Alcocer et al. Excitons versus free charges in organo-lead tri-halide perovskites. Nat Commun, 5, 3586(2014).
[8] Q Lin, A Armin, R C R Nagiri et al. Electro-optics of perovskite solar cells. Nat Photon, 9, 106(2014).
[9] H H Fang, F Wang, S Adjokatse et al. Photoexcitation dynamics in solution-processed formamidinium lead iodide perovskite thin films for solar cell applications. Light: Sci Appl, 5, e16056(2016).
[10] J H Noh, S H Im, J H Heo et al. Chemical management for colorful, efficient, and stable inorganic–organic hybrid nanostructured solar cells. Nano Lett, 13, 1764(2013).
[11] H Bian, D Bai, Z Jin et al. Graded bandgap CsPbI2+
[12] S D Stranks, G E Eperon, G Grancini et al. Electron-hole diffusion lengths exceeding 1 micrometer in an organometal trihalide perovskite absorber. Science, 342, 341(2013).
[13] H Wang, H Bian, Z Jin et al. Synergy of hydrophobic surface capping and lattice contraction for stable and high-efficiency inorganic CsPbI2Br perovskite solar cells. Solar RRL, 2, 1800216(2018).
[14] C C Stoumpos, C D Malliakas, M G Kanatzidis. Semiconducting tin and lead iodide perovskites with organic cations: phase transitions, high mobilities, and near-infrared photoluminescent properties. Inorg Chem, 52, 9019(2013).
[15] Y C Zhao, W K Zhou, X Zhou et al. Quantification of light-enhanced ionic transport in lead iodide perovskite thin films and its solar cell applications. Light: Sci Appl, 6, e16243(2017).
[16] C Xiao, Z Li, H Guthrey et al. Mechanisms of electron-beam-induced damage in perovskite thin films revealed by cathodoluminescence spectroscopy. J Phys Chem C, 119, 26904(2015).
[17] A F Akbulatov, S Y Luchkin, L A Frolova et al. Probing the intrinsic thermal and photochemical stability of hybrid and inorganic lead halide perovskites. J Phys Chem Lett, 8, 1211(2017).
[18] W Zhou, Y Zhao, X Zhou et al. Light-independent ionic transport in inorganic perovskite and ultrastable cs-based perovskite solar cells. J Phys Chem Lett, 8, 4122(2017).
[19] Q Wang, X Zhang, Z Jin et al. Energy-down-shift CsPbCl3:Mn quantum dots for boosting the efficiency and stability of perovskite solar cells. ACS Energy Lett, 2, 1479(2017).
[20] Z Jin, J Yan, X Huang et al. Solution-processed transparent coordination polymer electrode for photovoltaic solar cells. Nano Energy, 40, 376(2017).
[21] J Jiang, Z Jin, J Lei et al. ITIC surface modification to achieve synergistic electron transport layer enhancement for planar-type perovskite solar cells with efficiency exceeding 20%. J Mater Chem A, 5, 9514(2017).
[22] R E Beal, D J Slotcavage, T Leijtens et al. Cesium lead halide perovskites with improved stability for tandem solar cells. J Phys Chem Lett, 7, 746(2016).
[23] X Jia, C Zuo, S Tao et al. CsPb(I
[24] X Zhang, Z Jin, J Zhang et al. All-ambient processed binary CsPbBr3-CsPb2Br5 perovskites with synergistic enhancement for high-efficiency Cs-Pb-Br-based solar cells. ACS Appl Mater Interfaces, 10, 7145(2018).
[25] J Zhang, D Bai, Z Jin et al. 3D–2D–0D interface profiling for record efficiency all-inorganic CsPbBrI2 perovskite solar cells with superior stability. Adv Energy Mater, 8, 1703246(2018).
[26] D Bai, J Zhang, Z Jin et al. Interstitial Mn2+-driven high-aspect-ratio grain growth for low-trap-density microcrystalline films for record efficiency CsPbI2Br solar cells. ACS Energy Lett, 3, 970(2018).
[27] Y Y Zhang, S Chen, P Xu et al. Intrinsic instability of the hybrid halide perovskite semiconductor CH3NH3PbI3. Chin Phys Lett, 35, 036104(2018).
[28] C H Kang, I Dursun, G Liu et al. High-speed colour-converting photodetector with all-inorganic CsPbBr3 perovskite nanocrystals for ultraviolet light communication. Light: Sci Appl, 8, 94(2019).
[29] G Liu, C Zhou, F Wan et al. Dependence of power conversion properties of perovskite solar cells on operating temperature. Appl Phys Lett, 113, 3501(2018).
[30] G Liu, B Yang, B Liu et al. Irreversible light-soaking effect of perovskite solar cells caused by light-induced oxygen vacancies in titanium oxide. Appl Phys Lett, 111, 3501(2017).
[31] J F Wang, D X Lin, Y B Yuan. Recent progress of ion migration in organometal halide perovskite. Acta Phys Sin, 68, 158801(2019).
[32] W Ahmad, J Khan, G Niu et al. Inorganic CsPbI3 perovskite-based solar cells: a choice for a tandem device. Solar RRL, 1, 1700048(2017).
[33] P Wang, X Zhang, Y Zhou et al. Solvent-controlled growth of inorganic perovskite films in dry environment for efficient and stable solar cells. Nat Commun, 9, 2225(2018).
[34] X Zhang, Q Wang, Z Jin et al. Stable ultra-fast broad-bandwidth photodetectors based on
[35] J A Steele, H D Jin, I Iurii et al. Thermal unequilibrium of strained black CsPbI3 thin films. Science, 365, 679(2019).
[36] J B Hoffman, A L Schleper, P V Kamat. Transformation of sintered CsPbBr3 nanocrystals to cubic CsPbI3 and gradient CsPbBr
[37] Q Wang, Z Jin, D Chen et al.
[38] H Zhao, J Xu, S Zhou et al. Preparation of tortuous 3D
[39] A S Dayan, B E Cohen, S Aharon et al. Enhancing stability and photostability of CsPbI3 by reducing its dimensionality. Chem Mater, 30, 8017(2018).
[40] T Ye, B Zhou, F Zhan et al. Below 200 °C fabrication strategy of black phase CsPbI3 film for ambient-air-stable solar cells. Solar RRL, 10(2019).
[41] S Xiang, W Li, Y Wei et al. Natrium doping pushes the efficiency of carbon-based CsPbI3 perovskite solar cells to 10.7%. iScience, 15, 156(2019).
[42] G E Eperon, G M Paternò, R J Sutton et al. Inorganic caesium lead iodide perovskite solar cells. J Mater Chem A, 3, 19688(2015).
[43] K Wang, Z Jin, L Liang et al. All-inorganic cesium lead iodide perovskite solar cells with stabilized efficiency beyond 15%. Nat Commun, 9, 4544(2018).
[44] Y Wang, T Zhang, M Kan et al. Bifunctional stabilization of all-inorganic
[45] A Swarnkar, V K Ravi, A Nag. Beyond colloidal cesium lead halide perovskite nanocrystals: analogous metal halides and doping. ACS Energy Lett, 2, 1089(2017).
[46] A Marronnier, G Roma, S Boyer-Richard et al. Anharmonicity and disorder in the black phases of cesium lead iodide used for stable inorganic perovskite solar cells. ACS Nano, 12, 3477(2018).
[47] D Bai, H Bian, Z Jin et al. Temperature-assisted crystallization for inorganic CsPbI2Br perovskite solar cells to attain high stabilized efficiency 14.81%. Nano Energy, 52, 408(2018).
[48] M A Green, A Ho-Baillie, H J Snaith. The emergence of perovskite solar cells. Nat Photon, 8, 506(2014).
[49] G E Eperon, S D Stranks, C Menelaou et al. Formamidinium lead trihalide: a broadly tunable perovskite for efficient planar heterojunction solar cells. Energy Environ Sci, 7, 982(2014).
[50] J Zhang, G Hodes, Z Jin et al. All-inorganic CsPbX3 perovskite solar cells: progress and prospects. Angew Chem Int Ed, 58, 15596(2019).
[51] Y Huang, W J Yin, Y He. Intrinsic point defects in inorganic cesium lead iodide perovskite CsPbI3. J Phys Chem C, 122, 1345(2018).
[52] R J Sutton, M R Filip, A A Haghighirad et al. Cubic or orthorhombic? revealing the crystal structure of metastable black-phase CsPbI3 by theory and experiment. ACS Energy Lett, 3, 1787(2018).
[53] J K Sun, S Huang, X Z Liu et al. Polar solvent induced lattice distortion of cubic CsPbI3 nanocubes and hierarchical self-assembly into orthorhombic single-crystalline nanowires. J Am Chem Soc, 140, 11705(2018).
[54] V K Ravi, G B Markad, A Nag. Band edge energies and excitonic transition probabilities of colloidal CsPbX3 (X = Cl, Br, I) perovskite nanocrystals. ACS Energy Lett, 1, 665(2016).
[55] C C Stoumpos, M G Kanatzidis. The renaissance of halide perovskites and their evolution as emerging semiconductors. Acc Chem Res, 48, 2791(2015).
[56] C Katan, L Pedesseau, M Kepenekian et al. Interplay of spin–orbit coupling and lattice distortion in metal substituted 3D tri-chloride hybrid perovskites. J Mater Chem A, 3, 9232(2015).
[57] L Zheng, D Zhang, Y Ma et al. Morphology control of the perovskite films for efficient solar cells. Dalton Trans, 44, 10582(2015).
[58] C M M Soe, C C Stoumpos, B Harutyunyan et al. Room temperature phase transition in methylammonium lead iodide perovskite thin films induced by hydrohalic acid additives. ChemSusChem, 9, 2656(2016).
[59] A Sharenko, C Mackeen, L Jewell et al. Evolution of iodoplumbate complexes in methylammonium lead iodide perovskite precursor solutions. Chem Mater, 29, 1315(2017).
[60] D K Mohamad, B G Freestone, R Masters et al. Optimized organometal halide perovskite solar cell fabrication through control of nanoparticle crystal patterning. J Mater Chem C, 5, 2352(2017).
[61] F Haque, M Wright, M A Mahmud et al. Effects of hydroiodic acid concentration on the properties of CsPbI3 perovskite solar cells. ACS Omega, 3, 11937(2018).
[62] Y G Kim, T Y Kim, J H Oh et al. cesium lead iodide solar cells controlled by annealing temperature. Phys Chem Chem Phys, 19, 6257(2017).
[63] D Y Heo, S M Han, N S Woo et al. Role of additives on the performance of CsPbI3 solar cells. J Phys Chem C, 122, 15903(2018).
[64] Y Wei, W Li, S Xiang et al. Precursor effects on methylamine gas-induced CH3NH3PbI3 films for stable carbon-based perovskite solar cells. Solar Energy, 174, 139(2018).
[65] F Wang, H Yu, H Xu et al. HPbI3: a new precursor compound for highly efficient solution-processed perovskite solar cells. Adv Funct Mater, 25, 1120(2015).
[66] S Pang, Y Zhou, Z Wang et al. Transformative evolution of organolead triiodide perovskite thin films from strong room-temperature solid-gas interaction between HPbI3–CH3NH2 precursor pair. J Am Chem Soc, 138, 750(2016).
[67] X Ding, H Chen, Y Wu et al. Triple cation additive NH3+C2H4NH2+C2H4NH3+-induced phase-stable inorganic
[68] S Xiang, Z Fu, W Li et al. Highly air-stable carbon-based
[69] Y Wang, T Zhang, F Xu et al. A Facile low temperature fabrication of high performance CsPbI2Br all-inorganic perovskite solar cells. Solar RRL, 2, 1700180(2018).
[70] Q Ye, Y Zhao, S Mu et al. Cesium lead inorganic solar cell with efficiency beyond 18% via reduced charge recombination. Adv Mater, 31, e1905143(2019).
[71] W Ke, I Spanopoulos, C C Stoumpos et al. Myths and reality of HPbI3 in halide perovskite solar cells. Nat Commun, 9, 4785(2018).
[72] Y Pei, Y Liu, F Li et al. Unveiling property of hydrolysis-derived DMAPbI3 for perovskite devices: composition engineering, defect mitigation, and stability optimization. iScience, 15, 165(2019).
[73] Y Wang, M I Dar, L K Ono et al. Thermodynamically stabilized
[74] Y Wang, X Liu, T Zhang et al. the role of dimethylammonium iodide in CsPbI3 perovskite fabrication: additive or dopant. Angew Chem Int Ed, 58, 16691(2019).
[75] H Meng, Z Shao, L Wang et al. Chemical composition and phase evolution in DMAI-derived inorganic perovskite solar cells. ACS Energy Lett, 5, 263(2020).
[76] H Bian, H Wang, Z Li et al. Unveiling the effects of hydrolysis-derived DMAI/DMAPbI
[77] A Dutta, N Pradhan. Phase-stable red-emitting CsPbI3 nanocrystals: successes and challenges. ACS Energy Lett, 4, 709(2019).
[78] T Zhang, M I Dar, G Li et al. Bication lead iodide 2D perovskite component to stabilize inorganic
[79] Y Wang, T Zhang, M Kan et al. Efficient
[80] T Wu, Y Wang, Z Dai et al. Efficient and stable CsPbI3 solar cells via regulating lattice distortion with surface organic terminal groups. Adv Mater, 31, e1900605(2019).
[81] Y Fu, M T Rea, J Chen et al. Selective stabilization and photophysical properties of metastable perovskite polymorphs of CsPbI3 in thin films. Chem Mater, 29, 8385(2017).
[82] P Becker, J A Márquez, J Just et al. Low temperature synthesis of stable
[83] B Zhao, S Jin, S Huang et al. Thermodynamically stable orthorhombic
[84] C Liu, Y Yang, X Xia et al. Soft Template-controlled growth of high-quality CsPbI3 films for efficient and stable solar cells. Adv Energy Mater, 10, 1903751(2020).
[85] L Liang, L Zhizai, F Zhou et al. Humidity-insensitive fabrication of efficient CsPbI3 solar cells in ambient air. J Mater Chem A, 7, 26776(2019).
[86] H Wang, H Bian, Z Jin et al. Cesium lead mixed-halide perovskites for low-energy loss solar cells with efficiency beyond 17%. Chem Mater, 31, 6231(2019).
[87] K Wang, Z Jin, L Liang et al. Chlorine doping for black
[88] Z Yao, Z Jin, X Zhang et al. Pseudohalide (SCN-)-doped CsPbI3 for high performance solar cells. J Mater Chem C, 7, 13736(2019).
[89] H Bian, Q Wang, S Yang et al. Nitrogen-doped graphene quantum dots for 80% photoluminescence quantum yield for inorganic
[90] L Liang, M Liu, Z Jin et al. Optical Management with nanoparticles for a light conversion efficiency enhancement in inorganic
[91] H Bian, Q Wang, L Ding et al. Light management via tuning the fluorine-doped tin oxide glass haze-drives high-efficiency CsPbI3 solar cells. Phys Status Solidi A, 216, 1900602(2019).
[92] Q Wang, X Zheng, Y Deng et al. Stabilizing the
[93] Z Jin, M Yuan, H Li et al. Graphdiyne: an efficient hole transporter for stable high-performance colloidal quantum dot solar cells. Adv Funct Mater, 26, 5284(2016).
[94] Z Jin, A Wang, Q Zhou et al. Detecting trap states in planar PbS colloidal quantum dot solar cells. Sci Rep, 6, 37106(2016).
[95] H Yao, F Zhou, Z Li et al. Strategies for improving the stability of tin-based perovskite (ASnX3) solar cells. Adv Sci, 10, 1903540(2020).
[96] Z Jin, Q Zhou, Y Chen et al. Graphdiyne:ZnO nanocomposites for high-performance UV photodetectors. Adv Mater, 28, 3697(2016).
[97] Y Jiang, J Yuan, Y Ni et al. Reduced-dimensional
[98] K Wang, Z Li, F Zhou et al. Ruddlesden–popper 2D component to stabilize
[99] H Wang, S Xiang, W Li et al. Skillfully deflecting the question: a small amount of piperazine-1,4-diium iodide radically enhances the thermal stability of CsPbI3 perovskite. J Mater Chem C, 7, 11757(2019).
[100] A Dutta, S K Dutta, S Das Adhikari et al. Phase-stable CsPbI3 nanocrystals: the reaction temperature matters. Angew Chem Int Ed, 57, 9083(2018).
[101] J Xi, C Piao, J Byeon et al. Rational core-shell design of open air low temperature in situ processable CsPbI3 quasi-nanocrystals for stabilized p –i –n solar cells. Adv Energy Mater, 9, 1901787(2019).
[102] B Li, Y Zhang, L Fu et al. Surface passivation engineering strategy to fully-inorganic cubic CsPbI3 perovskites for high-performance solar cells. Nat Commun, 9, 1076(2018).
[103] J Gan, J He, R L Z Hoye et al.
[104] X Zhang, R Munir, Z Xu et al. Phase transition control for high performance ruddlesden-popper perovskite solar cells. Adv Mater, 30, 1707166(2018).
Set citation alerts for the article
Please enter your email address



