Shuangyi Zhao, Qionghua Mo, Baiqian Wang, Zhigang Zang. Research progress in inorganic perovskites white LEDs and visible light communication (Invited)[J]. Infrared and Laser Engineering, 2022, 51(1): 20210772
Search by keywords or author
- Infrared and Laser Engineering
- Vol. 51, Issue 1, 20210772 (2022)
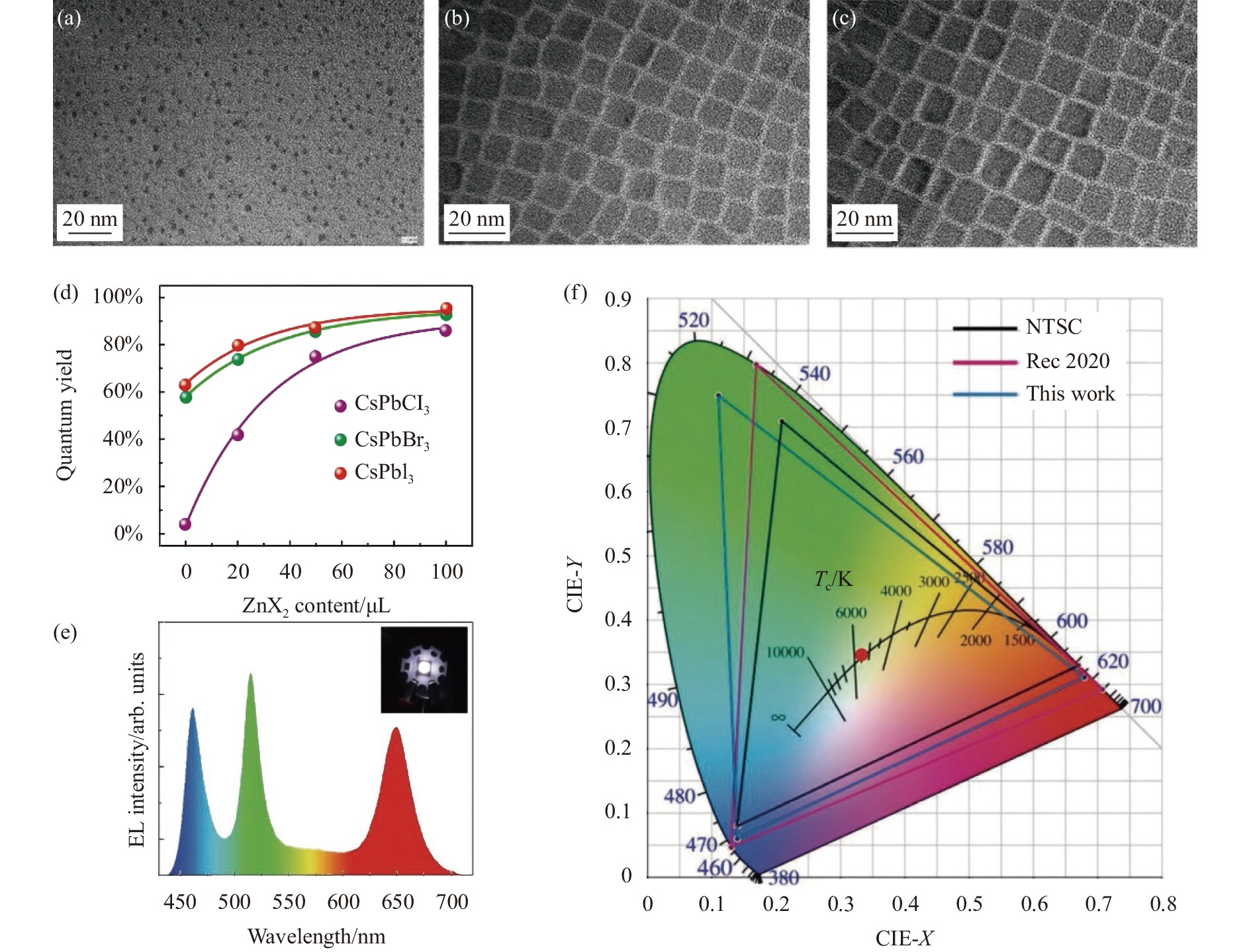
![Morphologies of CsPbBr3 NCs synthesized at (a) 80 ℃, (b) 120 ℃ and (c) 140 ℃[22]; (d) Variations of the PLQYs depending on the added amounts of ZnX2 of CsPbBr3 NCs[24]; (e) Spectra of white LED and (f) CIE color coordinates and color triangle based on CsPbBr3 and CsPbBr1.5I1.5[25]](/richHtml/irla/2022/51/1/20210772/img_1.jpg)
Fig. 1. Morphologies of CsPbBr3 NCs synthesized at (a) 80 ℃, (b) 120 ℃ and (c) 140 ℃[22]; (d) Variations of the PLQYs depending on the added amounts of ZnX2 of CsPbBr3 NCs[24]; (e) Spectra of white LED and (f) CIE color coordinates and color triangle based on CsPbBr3 and CsPbBr1.5I1.5[25]
![(a) TEM image of SiO2-coating CsPbBr3 NCs[27]; (b) TEM image of SiO2/CsPbBr3 NCs with Janus structure; (c) Schematic of the whole formation process of CsPbBr3/SiO2 NCs with Janus structure[29]; (d) Schematic of the two-step synthesis of CsPbX3-zeolite-Y composites; Spectra change of white LEDs composed of (e) CsPb(Br,I)3 perovskite QDs and (f) CsPb(Br,I)3-zeolite-Y composites with increase of currents[31]; (g) CIE color coordinate and color triangle of white LEDs based on mesoporous silica CsPbBr3 NCs[32]](/richHtml/irla/2022/51/1/20210772/img_2.jpg)
Fig. 2. (a) TEM image of SiO2-coating CsPbBr3 NCs[27]; (b) TEM image of SiO2/CsPbBr3 NCs with Janus structure; (c) Schematic of the whole formation process of CsPbBr3/SiO2 NCs with Janus structure[29]; (d) Schematic of the two-step synthesis of CsPbX3-zeolite-Y composites; Spectra change of white LEDs composed of (e) CsPb(Br,I)3 perovskite QDs and (f) CsPb(Br,I)3-zeolite-Y composites with increase of currents[31]; (g) CIE color coordinate and color triangle of white LEDs based on mesoporous silica CsPbBr3 NCs[32]
Fig. 3. (a) Schematics showing the Al3+ doping in dimer form[34]; Variety of (b) PL spectra and (c) PLQY of NCs with increase of doping ratio of Sn2+[36]; Evolution of (d) EL spectra and (e) CIE color coordinates of white LEDs based on Mn2+-doping CsPb(Br/Cl)3[37]
Fig. 4. (a) Crystal structure, (b) PL and PLE spectra, (c) schematic configuration coordinate for the excited-state reorganization of Cs3Cu2I5[46]; (d) PL spectra of Cs3Cu2Cl5 and CsCu2Cl3 synthesized at different temperatures[39]; (e) Power-dependent PL spectra of Cs4MnBi2Cl12; (f) Spectra of white LEDs based on Cs4MnBi2Cl12[40]
Fig. 5. (a) Crystal structure and (b) PL spectra excited under 310 nm of Pb2+-doped Cs3Cu2Br5[41]; (c) White-light PL spectra in Sb3+/Bi3+-codoped Cs2NaInCl6[42]; (d) Optical absorption and PL spectra of Cs2AgInCl6 and Cs2Ag0.6Na0.4InCl6[43]; (e) PL spectra of Cs2AgIn0.6B0.15La0.25Cl6 and Cs2AgIn0.8Bi0.2Cl6[47]; (f) CIE color coordinates of Li+/K+ alloyed Cs2AgBi0.01In0.99Cl6[48]
Fig. 6. (a) Schematic of band structure and (b) variety of EL spectra with the changes of blend ratio of white LED in CsPbBrx Cl3−x /MEH:PPV as luminous layer[50]; (c) Device structure, (d) EL spectrum, (e) current density-voltage and luminance-voltage curves, (f) EQE-current density and current efficiency-current density curves of white LEDs mixed with α /δ -CsPbI3 emitting layers[53]
Fig. 7. (a) Schematic diagram of the preparation process of CsCu2I3@Cs3Cu2I5 composites; (b) Absorption and PL spectra of the CsCu2I3@Cs3Cu2I5 composites with varied CsI/CuI molar ratios precursor[54]; Time-resolved GIWAXS profiles of CsCu2I3/Cs3Cu2I5 composites (c) without Tween and (d) with Tween; (e) EL spectra of the device under different voltages and (f) curve of EQE versus current density[55] of white LED after CsCu2I3/Cs3Cu2I5processed by Tween
Fig. 8. (a) Bit-error rates(BERs) at different data rates[56]; (b) Typical schematic diagrams of VLC test system[58]; (c) Electrical-optical-electrical frequency response and (d) received signal-to-noise ratio of white LEDs signal in VLC system based on ZrO2/CsPbBr3[28]; (e) Frequency response of white LEDs at different current densities[59]; (f) Constellation diagrams for white LEDs in VCL based on Cs3Cu2Cl5 NCs[60]
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 1. Optimized technologies and device parameters of typical inorganic perovskite white LEDs
Set citation alerts for the article
Please enter your email address



