- Journal of Semiconductors
- Vol. 41, Issue 9, 091702 (2020)
Abstract
1. Introduction
Under the demand of global sustainable development, solar energy and hydrogen energy are two clean and environmentally friendly new energy sources that attract full attention. With the help of photocatalysts, artificial simulation of photosynthesis in green plants can simultaneously achieve the absorption and utilization of sunlight and the production of hydrogen and oxygen by solar water splitting.
In 1972, Fujishima et al. from Japan discovered that under the illumination of near-ultraviolet light (380 nm), the rutile TiO2 single crystal electrode could split water into H2 and O2 at normal temperature[
Considering the extensive application of ZnO in PEC water splitting and the lack of relevant reviews, this paper will systematically describe the research of ZnO as a photoanode for solar water splitting. Firstly, the basic principle of PEC water splitting and the essential properties of ZnO, as well as related applications, are indispensably introduced. Secondly, several methods for enhancing the absorption efficiency of visible light, improving the efficiency of electron and hole separation, and improving the effectiveness of the surface catalytic reaction of ZnO-based photoanodes are described in this review, respectively, including morphology regulation, element doping, construction of the heterogeneous structure, and ferroelectric enhancement. This review aims to summarize recent findings and developments of ZnO-based photoanodes and to provide some useful guidance for the design and preparation of high-efficiency ZnO-based photoanodes for solar water splitting.
1.1. The fundamental of photoelectrochemical water splitting
With a semiconductor material fixed on a conductive substrate and working as an electrode, photogenerated electrons or holes are forced to move towards the opposite electrodes under an applied bias voltage, and thus the photogenerated carriers are separated. It is called the PEC method. Compared with photocatalytic water splitting using a powder-photocatalyst, PEC water splitting can not only compensate the valence band (VB) or conduction band (CB) potentials and overpotential of the semiconductor but also promote the separation of electrons and holes. Attributed to the existence of applied bias, PEC water splitting is expected to solve the problems of the high probability of electron–hole pair recombination, low apparent quantum yield, and low solar energy conversion efficiency of photocatalyst[
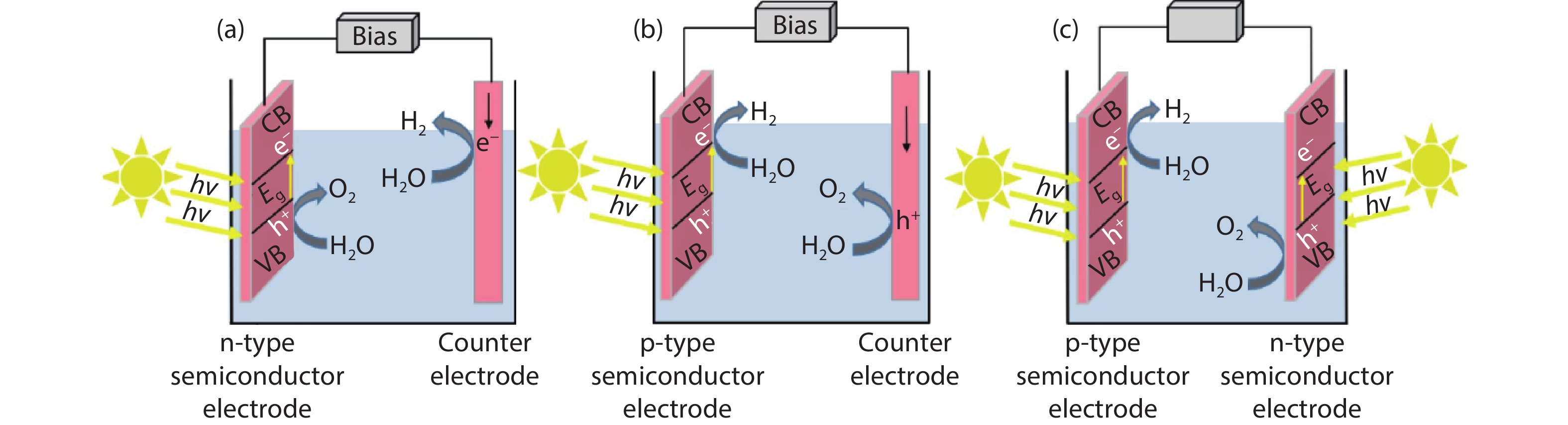
In general, as depicted in Fig. 1, there are three types of PEC water splitting system: (a) the PEC system with n-type semiconductor materials as the photoanode and hydrogen evolution at the counter electrode; (b) the PEC system with a p-type semiconductor as the photocathode and oxygen evolution at the counter electrode; (c) the PEC system with n-type and p-type semiconductor as the photoanode and photocathode respectively, and hydrogen and oxygen evolution simultaneously.
![]()
Figure 1.(Color online) PEC water splitting in (a) the n-type semiconductor-based PEC system, (b) p-type semiconductor-based PEC system, and (c) tandem system[
For a photoelectrochemistry device with an n-type semiconductor worked as the photoanode and hydrogen evolution at the counter electrode, the whole process at the photoanode can be divided into the following four steps (Fig. 2): (a) the absorption of light by the photoanode and the generation of photogenerated carriers; (b) the separation of carriers within the photoanode and at the solid–liquid interface; (c) carriers transport inside the photoanode; and (d) surface chemical redox reactions at the solid–liquid interface. In each of the above steps, there are energy losses, such as recombination and the capture of charge carriers, slow kinetics due to overpotential, and interruption of carrier transport. Therefore, the key to improving the conversion efficiency of PEC cells is to improve the photoanode performance of materials and minimize unnecessary energy losses.
![]()
Figure 2.(Color online) Main processes of PEC water splitting for n-type semiconductors.
The selection of photoanode semiconductor nanomaterials for PEC cells is the top priority in the construction of an efficient PEC system. Four core problems corresponding to the above four steps must be considered: (1) the light absorption capacity of the photoanode and the generation efficiency of photogenerated carriers; (2) separation and transport efficiency of photogenerated electron–hole pairs; (3) carriers extraction at the photoanode/electrolyte interface and generation of electrochemical products; (4) stability of the photoanode in the electrolyte solution. Only by taking the above four factors into full consideration can a photoanode with high photoelectric conversion efficiency be obtained, so as to further improve the performance of the PEC water splitting system.
1.2. Characteristics and related applications of ZnO
Among copious semiconductor materials, the third-generation semiconductor ZnO nanomaterials are one of the most importantly exploited objects. ZnO is an n-type direct wide bandgap (Eg = 3.37 eV)[
Generally, there are three types of crystal phases for ZnO, including the hexagonal wurtzite structure, zinc blende, and rare rock salt structure. Among them, the wurtzite structure with the highest stability[
![]()
Figure 3.(Color online) (a) ZnO model of the hexagonal wurtzite structure, (b) schematic illustrations of atoms and charges distribution in the unit cell of Wurlitzer-structure ZnO, where
2. Application of ZnO in PEC water splitting
In the past few decades, many metal oxide semiconductor materials, including TiO2, ZnO, α-Fe2O3, BiVO4, WO3, and perovskite materials, have been widely studied and employed as photoanodes in PEC water splitting due to their excellent optical-electrical characteristic, suitable band structure (the VB position should be more positive than the redox potential of O2/H2O to ensure the oxidation characteristics of photogenerated holes, which is more conducive to the oxygen evolution reaction), the appropriate onset potential, high catalytic activity, and low cost in the process of the water oxidation reaction[
As shown in Fig. 4, the bottom of the CB for ZnO is slightly more negative than the reduction potential of hydrogen, while the top of VB is far more positive than that of water oxidation. Thus, as a photoanode, ZnO has excellent advantages in conducting holes to water, which can enhance the separation of photogenerated electron–hole pairs at the photoanode and promote the PEC conversion efficiency of the whole system effectively.
![]()
Figure 4.(Color online) Energy potentials of ZnO and redox potentials for PEC water splitting at pH = 7, relative to NHE (normal hydrogen electrode).
The advantages of ZnO nanomaterials in PEC water splitting are as follows: (1) ZnO nanomaterials usually have low reflectivity and strong light capture ability due to the fine nanostructure on the surface, which can endow ZnO with a high light absorption performance; (2) the surface of ZnO usually adsorbs abundant O2 molecules due to the existence of a large number of surface states. It then tends to capture photogenerated holes, which ultimately prolongs the lifetime of photogenerated electrons and improves the separation efficiency of photogenerated electron-hole pairs; (3) ZnO single crystal nanostructures with excellent crystallization performance prepared by various methods have high electron mobility[
However, ZnO has a wide bandgap and a band absorption edge at about 368 nm. It can only utilize the ultraviolet light, which accounts for only 4% of the solar spectrum[
![]()
Figure 5.(Color online) Carrier transport mechanism of the ZnO photoanode.
2.1. The morphology control of ZnO
At present, various morphologies of ZnO nanostructures can be prepared by a variety of deposition methods, physical and chemical synthesis methods, including nanoparticles of various shapes, nanowires, nanorods, and nanosheets, as well as relatively uncommon nanopencil and nanotetrapod structures.
In the photocatalytic reaction, the low dimension catalytic nanomaterials have short photogenerated carrier migration paths, fast carriers migration efficiency, and a large surface area[
ZnO's one-dimensional nanostructure significantly improves its photoelectric and power transmission performance and is widely used in solar cells, PEC cells and other photoelectric conversion devices[
Zhang et al.[
Li et al.[
Yang et al.[
Gong et al.[
Li et al. firstly fabricated open bird-nest (BN) structured ZnO/TiO2 and sealed core-shell (CS) structured ZnO/TiO2 (Fig. 6)[
![]()
Figure 6.(Color online) Schematic illustration of the preparation processes of CS ZnO/TiO2 and BN ZnO/TiO2[
The purpose of controlling the morphology of ZnO nanomaterials is mainly to increase the contact area between the semiconductor materials of photoanode and electrolyte or to change the surface roughness of the photoanode, so as to expose more reaction sites and reduce the carrier migration distance. Control of morphology is a fundamental method in the study of the material system, and it is generally combined with other means to improve PEC water splitting efficiency.
2.2. The element doping of ZnO
Through physical or chemical methods, the extra ions are injected into the semiconductor materials, and this method is called element doping. Additional doping can increase the donor concentration based on a certain number of electron donors[
![]()
Figure 7.(Color online) Effect of element doping on band structure[
In order to improve the electronic and optical properties, and to inhibit inherent photocorrosion defects of ZnO as much as possible, several impurity atoms can be permeated into the lattice of ZnO. Currently, the common doped elements include Fe, Al, Cu, Co, Cr, Ni, C, Cl, and N elements[
Theoretically, N-doping to replace significant amounts of the lattice oxygen with nitrogen (N2p orbitals are energetically more negative than O2p orbital) is a strategy designed to shift the VB higher[
![]()
Figure 8.(Color online) (a) Schematic diagram of N gradient doped ZnO nanorods and stepped band structure to promote carrier separation[
Commandeur et al. first synthesized Y-doped ZnO vertically aligned nanorods using a unique rapid microwave-assisted method[
2.3. The heterostructure construction of ZnO
The construction of heterostructure with rational band alignment seems to be a very effective method to improve the performance of photoanode semiconductors in PEC water splitting. In the existing research, the heterostructure of ZnO is mainly constructed by utilizing the carrier separation effect of type-II, Z-scheme heterojunction or p–n junction, the quantum dot sensitization, the surface plasmon resonance of noble nanometals, the passivation layer of the heterojunction and the modified OER co-catalyst for the application of PEC water splitting. These are described in the categories as below.
2.3.1. Construction of the ZnO-based heterojunction structure
When other semiconductor materials are compounded with ZnO nanomaterials, three possible kinds of heterojunction will be formed, including a type-II junction, p–n junction, and Z-scheme system junction. The electron transport of these three heterojunctions is shown in Figs. 9(a)–9(c). The type-II heterojunction is defined as the staggered alignment of the energy bands at the interface[
![]()
Figure 9.(Color online) Schematic diagrams of the forms of (a) type-II junction, (b) p–n junction, (c) Z-scheme system, and (d) hot-electron injection[
Wannapop and Somdee[
![]()
Figure 10.(Color online) Schematic illustration of the proposed mechanism for the charge transfer (a) in ZnWO4/ZnO photoanode[
Amorphous MoSx was decorated on the surface of vertically grown ZnO thin sheets via the electrodeposition technique in Sharma’s work[
The p–n heterojunction of CuO/ZnO in nanorod array photoanodes was reported[
Zhang et al.[
Hsu et al. firstly synthesized the ZnO–Au–SnO2 Z-scheme system photoanode for PEC water splitting, and the sample was prepared by the deposition of a thin layer of SnO2 on ZnO nanorods modified with Au nanoparticles[
As mentioned earlier, due to the Z-scheme heterostructure formed between ZnO and TiO2, the PEC performance of the open BN ZnO/TiO2 fabricated by Li et al.[
2.3.2. The construction of ZnO heterogeneous structure based on quantum dot sensitization
By modifying the quantum dot semiconductor materials on the ZnO photoanode system and combining it with the light absorption performance of the quantum dot, it is expected to solve the problem that the absorption region of ZnO is concentrated in the ultraviolet region. The modified ZnO-based semiconductor composite system with various band gaps has a broader spectral response range, resulting to the improved light absorption efficiency of ZnO. Semiconductor materials CdS and CdSe are widely used as modified quantum dots due to their narrow bandgap of 2.42 and 0.52 eV, respectively, and the corresponding light absorption cut-off wavelengths are 513 and 2387 nm, respectively. In addition to narrow-band gap semiconductors, low-dimensional carbon materials can also be used as sensitizers to promote the light absorption of ZnO.
Liu et al. prepared single-layer CdTe quantum dot sensitized ZnO nanowire array photoelectrodes and realized the sensitization of CdX quantum dots to ZnO nanowires under the non-sacrificial agent electrolyte for the first time. The maximum PCE of the system was up to 1.83%, which was 200% higher than that of the original ZnO nanowires[
Li et al. prepared a co-sensitized ZnO nanowire array of double-sided CdS and CdSe quantum dots for PEC hydrogen evolution[
![]()
Figure 11.(Color online) (a) Bilateral CdS–ZnO–ZnO–CdSe nanowire array photoanode structure and corresponding energy level diagram[
Tian et al.[
Han et al. explored the application of a unique combination of metal–organic framework (MOF)-decorated ZnO 1D nanostructures as the host and nitrogen-doped carbon dots (N-CDs) as the guest species in PEC water splitting[
2.3.3. The construction of heterostructure of ZnO based on the surface plasmon resonance
When the frequency of the light is the same as the frequency of the electrons themselves on the surface of the metal, the valence electrons of some nano-sized metal particles (mainly precious metals, e.g., Au, Ag and Pt) oscillate collectively. In other words, resonance happens when the electron oscillation and the oscillating electric field have the same frequency; at this point, the metal nanoparticles show the light absorption ability. This phenomenon is the surface plasmon resonance effect of precious metals. Based on this, when noble metal nanoparticles are loaded on the ZnO matrix composite surface, the excited energy transferred from the nanometals could improve the absorption and scattering efficiency of the adjacent semiconductors. The hot electron injection to the semiconductor (as shown in Fig. 9(d)) and the enhanced local electric field are responsible for the improvement of PEC performance[
Kang et al. prepared a 3D branched ZnO nanowire array modified with nano Au for PEC water splitting. This composite electrode exhibits excellent PEC activity in both the ultraviolet and visible region. The enhancement of optical activity in the visible region was due to the surface plasmon resonance effect of Au nanoparticles. The metal/semiconductor interface was conducive to the extraction of hot electrons from nanometer Au, so the PEC efficiency was improved (Fig. 12(a))[
![]()
Figure 12.(Color online) (a) The main mechanism of Au/3D ZnO nanowire photoelectrode[
2.3.4. Construction of ZnO heterogeneous structure based on the passivation layer and co-catalyst
The oxidation potential of ZnO lies above the oxidation potential of water[
Zhang et al. constructed an ultra-thin TiO2 shell structure to improve the photocatalytic activity and chemical stability of the nanowires photoanode. The coating TiO2 could passivate the surface capture state of ZnO effectively, and compared with pure ZnO nanowires, the composite electrode had a 25% PEC efficiency improvement[
Bamboo shoot-shaped FeVO4 (FVO, a novel and seldom studied n-type indirect bandgap semiconductor) passivated ZnO heterojunction nanorods photoanode was prepared for solar water splitting only by the spin-coating and calcination method[
![]()
Figure 13.(Color online) Schematic band alignment of charge transport and recombination models in (a) ZnO and (b) FVO/ZnO photoanodes[
Metal–organic framework (MOF)-based transparent surface passivation layer modified ZnO nanorod arrays with Ni(OH)2 nanosheets grafted on as the co-catalyst were introduced for PEC water splitting[
2.4. The piezo-photoelectric enhancement of ZnO
Wurtzite ZnO exhibits an inherent piezotronic effect and piezo-phototronic effect[
In order to further solve the limited light utilization and fast charge carrier recombination of ZnO, the piezoelectric effect of ZnO also plays a particular role in PEC water splitting, realizing the synergy of piezocatalysis and photoelectrocatalysis. Under light and mechanical stress, the piezoelectric ZnO can generate charge carriers directly[
Initially, the electrons in the conduction band of an n-type semiconductor in a loop current tend to drift toward the semiconductor side due to the bent banding upward in the interface between the semiconductor and electrolyte, and then transport to the counter electrode through the external circuit. At the same time, the holes in the valance band can move toward the electrolyte directly. When a tensile strain is applied, the side directly interfacing with the electrolyte will have a negative piezopotential, which can lead to an upward bending of the band edge, thus efficiently reducing the local resistance or threshold voltage for the surface oxidation reaction. In comparison, if a compressive strain is applied, a positive piezopotential will be generated, which will lower and flatten the band edges at the semiconductor/electrolyte interface, thus reducing the driving force of electron–hole separation and reducing the redox efficiency (Fig. 14)[
![]()
Figure 14.(Color online) Piezo-phototronic effect on the photoelectrocatalytic process (photoanode). Illustration of the photoelectrocatalytic process (a) without strain, (b) under tensile strain, and (c) under compressive strain[
In order to generate high piezopotential along with c-axis under axial stress, Wang et al. fabricated a vertically aligned ZnO nanorod array with a high aspect ratio, and they further deposited Au NPs only at the tip of the ZnO nanorods via selective ZnO tip exposure (Fig. 15). It showed that asymmetric gold–zinc oxide (Asy–Au–ZnO) could facilitate the separation and migration of the electron–hole pairs to the material surface to enhance the PEC performance. However, high piezo-photocatalytic performance with a catalytic efficiency of 95% in 75 min for dye degradation rather than water splitting was gained in this work[
![]()
Figure 15.(Color online) Schematic illustration of the enhanced catalytic performance induced by piezotronic effect and unique asymmetric nanostructure under light irradiation and ultrasonic actuation (
3. Summary and prospects
The third-generation direct bandgap semiconductor of ZnO possesses various advantages in the application in PEC water splitting as the photoanode, such as suitable band structure, high electron mobility, environmental friendliness, fine surface structure and comparable efficiency for various photocatalytic reactions compared with TiO2; nevertheless, due to its wide bandgap, ZnO has a relatively low visible light response. Besides, it has fast recombination of photogenerated charge carriers and poor chemical stability resulting from intrinsic photocorrosion. As a result of the above drawbacks, the application of ZnO in PEC water splitting is limited to the experimental stage and cannot be carried out on a large scale in practice. To this end, various photoanodes based on ZnO nanomaterials for efficient PEC water splitting have been constructed through many regulations and much engineering, including morphology control, element doping, heterostructure construction, and piezo-photoelectric enhancement. In this paper, we systematically reviewed the relevant research on optical model optimization, the energy band structure regulation, heterojunction band matching design, the effects of surface plasmon resonance of noble nanometals, and surface passivation. Based on the above strategies, the behaviors of the photogenerated charge carriers, including generation, separation, and recombination within the semiconductor photoanode, are effectively controlled, and excellent PEC properties are obtained.
The engineering of a nanostructured ZnO photoanode is not a single project, but a combination of various means to improve PEC water splitting efficiency. At present, a one-dimensional ZnO nanoarray structure, combined with element doping or the construction of a surfaced heterogeneous structure, modified with a quantum dot sensitizer or noble metal, is a more concerned and efficient PEC water splitting system. The piezoelectric effect in wurtzite ZnO is beneficial for modulating the migration and separation of internal charge carriers, resulting in the improved performance of the PEC water splitting system. Unfortunately, there are few pieces of research on the piezoelectric effect of ZnO, specifically for PEC water splitting.
Even though many strategies have been developed to modulate ZnO nanomaterials for PEC water splitting, the low solar-to-hydrogen conversion efficiency of the ZnO-based PEC water splitting systems hinders it for large-scale applications. Therefore, further studies and new strategies are demanded. The future research of ZnO as an efficient PEC water splitting photoanode can be mainly carried out from the following aspects: (1) design and construction of multi-heterojunction to enhance visible light absorption and electron–hole pair separation; (2) preparation and application of various one-dimensional ZnO nanoarray structures; (3) application of piezoelectric effect to enhance the separation of photogenerated carriers; and (4) radial-gradient element doping of ZnO one-dimensional structures to change the band structure of ZnO.
Acknowledgements
This work was mostly supported by the National Natural Science Foundation of China (Grant No. 21975245, 51972300 and 61674141), the Key Research Program of Frontier Science, CAS (Grant No. QYZDB-SSW-SLH006), the National Key Research and Development Program of China (Grant No. 2017YFA0206600, 2018YFE0204000), the Strategic Priority Research Program of Chinese Academy of Sciences (Grant No. XDB43000000), K. L. also acknowledges the support from the Youth Innovation Promotion Association, Chinese Academy of Sciences (No. 2020114).
References
[1] A Fujishima, K Honda. Electrochemical photolysis of water at a semiconductor electrode. Nature, 238, 37(1972).
[2] M D Bhatt, J S Lee. Recent theoretical progress in the development of photoanode materials for solar water splitting photoelectrochemical cells. J Mater Chem, 3, 10632(2015).
[3] Y C Qiu, Z H Pan, H N Chen et al. Current progress in developing metal oxide nanoarrays-based photoanodes for photoelectrochemical water splitting. Sci Bull, 64, 1348(2019).
[4] L Wang, S Liu, Z Wang et al. Piezotronic effect enhanced photocatalysis in strained anisotropic ZnO/TiO2 nanoplatelets via thermal stress. ACS Nano, 10, 2636(2016).
[5] B D Liu, J Li, W J Yang et al. Semiconductor solid-solution nanostructures: Synthesis, property tailoring, and applications. Small, 13, 1701998(2017).
[6] H C He, A Z Liao, W L Guo et al. State-of-the-art progress in the use of ternary metal oxides as photoelectrode materials for water splitting and organic synthesis. Nano Today, 28, 100763(2019).
[7] Y Y Zhang, M K Ram, E K Stefanakos et al. Synthesis, characterization, and applications of ZnO nanowires. J Nanomater, 2012, 1(2012).
[8] P S Xu, Y M Sun, C S Shi et al. The electronic structure and spectral properties of ZnO and its defects. Nucl Instrum Methods Phys Res B, 199, 286(2003).
[9] S Xu, Z L Wang. One-dimensional ZnO nanostructures: Solution growth and functional properties. Nano Res, 4, 1013(2011).
[10] Y I Özgür, C Alivov et al. A comprehensive review of ZnO materials and devices. J Appl Phys, 98, 041301(2005).
[11] Z L Wang. Progress in piezotronics and piezo-phototronics. Adv Mater, 24, 4632(2012).
[12] Y Yuan, A W Tang. Progress on the controllable synthesis of all-inorganic halide perovskite nanocrystals and their optoelectronic applications. J Semicond, 41, 011201(2020).
[13] Y B Huang, J Liu, Y C Deng et al. The application of perovskite materials in solar water splitting. J Semicond, 41, 011701(2020).
[14] A J E Rettie, H C Lee, L G Marshall et al. Combined charge carrier transport and photoelectrochemical characterization of BiVO4 single crystals: Intrinsic behavior of a complex metal oxide. J Am Chem Soc, 135, 11389(2013).
[15] M Wang, F Ren, J G Zhou et al. N doping to ZnO nanorods for photoelectrochemical water splitting under visible light: Engineered impurity distribution and terraced band structure. Sci Rep, 5, 12925(2015).
[16] A M Pourrahimi, K Villa, C L Manzanares Palenzuela et al. Catalytic and light-driven ZnO/Pt Janus nano/micromotors: Switching of motion mechanism via interface roughness and defect tailoring at the nanoscale. Adv Funct Mater, 29, 1808678(2019).
[17] F E Osterloh. Inorganic nanostructures for photoelectrochemical and photocatalytic water splitting. Chem Soc Rev, 42, 2294(2013).
[18] C Zhao, B Xu, Z J Wang et al. Boron-doped III–V semiconductors for Si-based optoelectronic devices. J Semicond, 41, 011301(2020).
[19] R N Wang, S B Chen, Y H Ng et al. ZnO/CdS/PbS nanotube arrays with multi-heterojunctions for efficient visible-light-driven photoelectrochemical hydrogen evolution. Chem Eng J, 362, 658(2019).
[20] A Wolcott, W A Smith, T R Kuykendall et al. Photoelectrochemical study of nanostructured ZnO thin films for hydrogen generation from water splitting. Adv Funct Mater, 19, 1849(2009).
[21] X Y Yang, A Wolcott, G M Wang et al. Nitrogen-doped ZnO nanowire arrays for photoelectrochemical water splitting. Nano Lett, 9, 2331(2009).
[22] Y C Qiu, K Y Yan, H Deng et al. Secondary branching and nitrogen doping of ZnO nanotetrapods: Building a highly active network for photoelectrochemical water splitting. Nano Lett, 12, 407(2012).
[23] T Wang, R Lv, P Zhang et al. Au nanoparticle sensitized ZnO nanopencil arrays for photoelectrochemical water splitting. Nanoscale, 7, 77(2015).
[24] T S Zhou, J C Wang, S Chen et al. Bird-nest structured ZnO/TiO2 as a direct Z-scheme photoanode with enhanced light harvesting and carriers kinetics for highly efficient and stable photoelectrochemical water splitting. Appl Catal B, 267, 118599(2020).
[25] T Tynell, M Karppinen. Atomic layer deposition of ZnO: A review. Semicond Sci Technol, 29, 043001(2014).
[26] J Z Zhang. Metal oxide nanomaterials for solar hydrogen generation from photoelectrochemical water splitting. MRS Bull, 36, 48(2011).
[27] Z Kang, H N Si, S C Zhang et al. Interface engineering for modulation of charge carrier behavior in ZnO photoelectrochemical water splitting. Adv Funct Mater, 29, 1808032(2019).
[28] S S K Ma, T Hisatomi, K Domen. Hydrogen production by photocatalytic water splitting. J Jpn Petrol Inst, 56, 280(2013).
[29] S L Xie, X H Lu, T Zhai et al. Enhanced photoactivity and stability of carbon and nitrogen co-treated ZnO nanorod arrays for photoelectrochemical water splitting. J Mater Chem, 22, 14272(2012).
[30] D Commandeur, G Brown, P McNulty et al. Yttrium-doped ZnO nanorod arrays for increased charge mobility and carrier density for enhanced solar water splitting. J Phys Chem C, 123, 18187(2019).
[31] S Wannapop, A Somdee. Effect of citric acid on the synthesis of ZnWO4/ZnO nanorods for photoelectrochemical water splitting. Inorg Chem Commun, 115, 107857(2020).
[32] M D Sharma, C Mahala, M Basu. Sensitization of vertically grown ZnO 2D thin sheets by MoS
[33] C Liu, F L Meng, L Zhang et al. CuO/ZnO heterojunction nanoarrays for enhanced photoelectrochemical water oxidation. Appl Surf Sci, 469, 276(2019).
[34] J M Li, H Y Cheng, Y H Chiu et al. ZnO–Au–SnO2 Z-scheme photoanodes for remarkable photoelectrochemical water splitting. Nanoscale, 8, 15720(2016).
[35] H M Chen, C Chen, Y C Chang et al. Quantum dot monolayer sensitized ZnO nanowire-array photoelectrodes: True efficiency for water splitting. Angew Chem Int Ed, 49, 5966(2010).
[36] C X Guo, Y Q Dong, H B Yang et al. Graphene quantum dots as a green sensitizer to functionalize ZnO nanowire arrays on F-doped SnO2 Glass for enhanced photoelectrochemical water splitting. Adv Energy Mater, 3, 997(2013).
[37] G M Wang, X Y Yang, F Qian et al. Double-sided CdS and CdSe quantum dot Co-sensitized ZnO nanowire arrays for photoelectrochemical hydrogen generation. Nano Lett, 10, 1088(2010).
[38] H Y Jiang, Y J Chen, L Li et al. Hierarchical ZnO nanorod/ZnFe2O4 nanosheet core/shell nanoarray decorated with PbS quantum dots for efficient photoelectrochemical water splitting. J Alloy Compd, 828, 154449(2020).
[39] H Han, F Karlicky, S Pitchaimuthu et al. Highly ordered N-doped carbon dots photosensitizer on metal–organic framework-decorated ZnO nanotubes for improved photoelectrochemical water splitting. Small, 15, 1902771(2019).
[40] X Zhang, Y Liu, Z H Kang. 3D branched ZnO nanowire arrays decorated with plasmonic au nanoparticles for high-performance photoelectrochemical water splitting. ACS Appl Mater Interfaces, 6, 4480(2014).
[41] Y F Wei, L Ke, J H Kong et al. Enhanced photoelectrochemical water-splitting effect with a bent ZnO nanorod photoanode decorated with Ag nanoparticles. Nanotechnology, 23, 235401(2012).
[42] R B Wei, P Y Kuang, H Cheng et al. Plasmon-enhanced photoelectrochemical water splitting on gold nanoparticle decorated ZnO/CdS nanotube arrays. ACS Sustain Chem Eng, 5, 4249(2017).
[43] S Y Chen, L W Wang. Thermodynamic oxidation and reduction potentials of photocatalytic semiconductors in aqueous solution. Chem Mater, 24, 3659(2012).
[44] C F Liu, Y J Lu, C C Hu. Effects of anions and pH on the stability of ZnO nanorods for photoelectrochemical water splitting. ACS Omega, 3, 3429(2018).
[45] J Kegel, I M Povey, M E Pemble. Zinc oxide for solar water splitting: A brief review of the material's challenges and associated opportunities. Nano Energy, 54, 409(2018).
[46] M Z Liu, C Y Nam, C T Black et al. Enhancing water splitting activity and chemical stability of zinc oxide nanowire photoanodes with ultrathin titania shells. J Phys Chem C, 117, 13396(2013).
[47] M F Shao, F Y Ning, M Wei et al. Nanowire arrays: Hierarchical nanowire arrays based on ZnO core-layered double hydroxide shell for largely enhanced photoelectrochemical water. Adv Funct Mater, 24, 565(2014).
[48] X F Long, L L Gao, F Li et al. Bamboo shoots shaped FeVO4 passivated ZnO nanorods photoanode for improved charge separation/transfer process towards efficient solar water splitting. Appl Catal B, 257, 117813(2019).
[49] X Li, S W Liu, K Fan et al. MOF-based transparent passivation layer modified ZnO nanorod arrays for enhanced photo-electrochemical water splitting. Adv Energy Mater, 8, 1800101(2018).
[50] H X He, Y M Fu, W L Zang et al. A flexible self-powered T-ZnO/PVDF/fabric electronic-skin with multi-functions of tactile-perception, atmosphere-detection and self-clean. Nano Energy, 31, 37(2017).
[51] C F Pan, J Y Zhai, Z L Wang. Piezotronics and piezo-phototronics of third generation semiconductor nanowires. Chem Rev, 119, 9303(2019).
[52] X Y Chen, L F Liu, Y W Feng et al. Fluid eddy induced piezo-promoted photodegradation of organic dye pollutants in wastewater on ZnO nanorod arrays/3D Ni foam. Mater Today, 20, 501(2017).
[53] D L Xiang, Z R Liu, M Q Wu et al. Enhanced piezo-photoelectric catalysis with oriented carrier migration in asymmetric Au–ZnO nanorod array. Small, 16, 1907603(2020).
[54] L Pan, S C Sun, Y Chen et al. Advances in piezo-phototronic effect enhanced photocatalysis and photoelectrocatalysis. Adv Energy Mater, 10, 2000214(2020).
Set citation alerts for the article
Please enter your email address



