- High Power Laser Science and Engineering
- Vol. 7, Issue 3, 03000e46 (2019)
Abstract
1 Introduction
Deuterated potassium dihydrogen phosphate (DKDP) crystals are optical materials with excellent properties, such as high transmittance, high electro-optic coefficient and high laser damage threshold, which are widely used for frequency conversion and as electro-optical
The traditional temperature-reduction method for producing crystals has been widely used to grow large-size, highly deuterated DKDP crystals, which requires a long period of more than one year[
2 Experiment
2.1 Synthesis
Synthesis of the growth solution is the most important factor for the growth of highly deuterated DKDP crystals. The usual procedure was adopted to synthesize the high-deuterium concentration growth solution, which was divided into two steps. First, a phosphorus pentoxide (
Sign up for High Power Laser Science and Engineering TOC. Get the latest issue of High Power Laser Science and Engineering delivered right to you!Sign up now
| Element | Concentration (ppb) | Element | Concentration (ppb) |
|---|---|---|---|
| Al | 46.81 | Pb | 9.02 |
| As | 32.74 | Li | 10.11 |
| Ba | 13.62 | Mg | 99.08 |
| Co | 2.87 | Mn | 8.32 |
| Cr | 10.93 | Ni | 7.51 |
| Cu | 13.06 | Sr | 3.84 |
| Fe | 100.82 | Zn | 25.19 |
Table 1. Concentration of the main metallic ionic impurities in
| Element | Concentration (ppb) | Element | Concentration (ppb) |
|---|---|---|---|
| Al | 123.52 | Pb | 18.83 |
| As | 34.98 | Li | 39.56 |
| Ba | 38.12 | Mg | 172.84 |
| Co | 10.36 | Mn | 99.75 |
| Cr | 55.46 | Ni | 40.88 |
| Cu | 39.65 | Sr | 20.71 |
| Fe | 546.12 | Zn | 118.23 |
Table 2. Concentration of the main metallic ionic impurities in
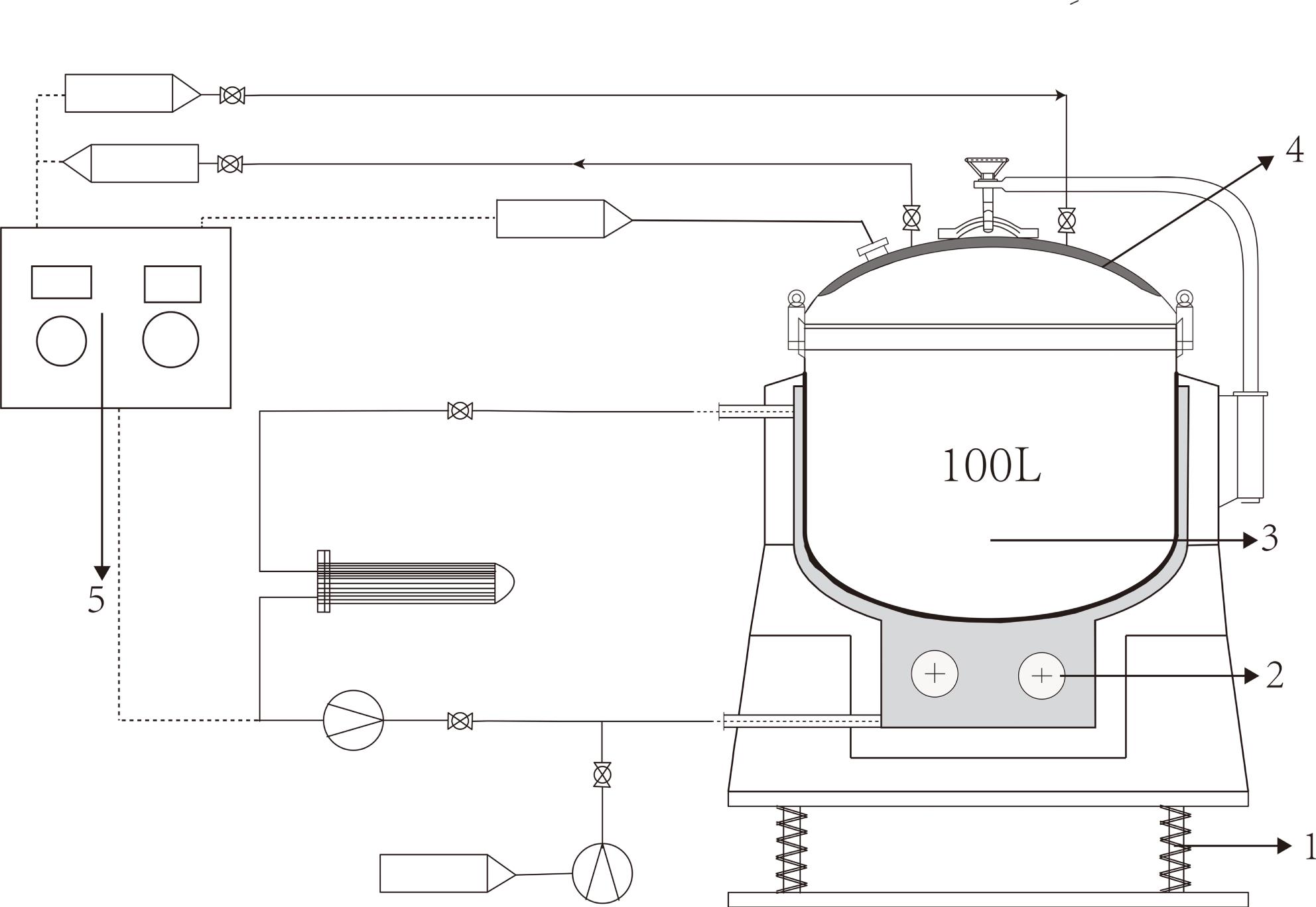
To avoid hydrogen–deuterium exchange, a synthesis tank device was designed to provide a closed environment for these two reactions. The schematic of the synthesis tank device is shown in Figure
Before the reactions,
2.2 Crystal growth
The solution was overheated for more than 72 h in the 500-L standard hold-type crystallizer to avoid spontaneous nucleation. The highly deuterated DKDP crystal was grown rapidly from a
The as-grown crystal was
2.3 Measurements
Transmittance spectra of the crystal were recorded by a Lambda 950 ultraviolet–visible–near-infrared (UV–vis–NIR) spectrophotometer in the range 200–2000 nm. The spectral resolution of the spectrophotometer was 1 nm. The transmittance value was converted into the absorption coefficient according to the Lambert–Beer law.
The laser-induced damage threshold (LIDT) of DKDP crystals was measured by the Shanghai Institute of Optics and Fine Mechanics. The experimental setup for assessing the damage threshold is shown in Figure
3 Results and discussion
3.1 Deuterium content of the grown crystal
In DKDP crystals, especially high-deuterium crystal, an accurate measurement of the deuterium concentration is very important. The chemical and physical properties of the hydrogen and deuterium atoms in the DKDP crystal, such as the structural dimensions, bond lengths and bond angles, are closely related to the deuterium content of DKDP crystals[
Liu
The as-grown DKDP crystal was
| No. | 1 | 2 | 3 | 4 | Average |
|---|---|---|---|---|---|
| Deuterium level (%) | 97.7 | 98.0 | 97.9 | 98.0 | 97.9 |
Table 3. Results for the deuterium level of DKDP crystal as assessed from IR spectra.
To accurately measure the degree of deuteration of crystals, we also used the TGA method to determine the deuterium concentration of the as-grown DKDP crystal. Samples of approximately 80 mg from different parts of the as-grown DKDP crystal were selected for measurement of the thermo-gravimetric curve. Samples 1 and 2 were taken from the pyramid and prismatic regions of the crystal, respectively, with the location of each sample being shown in Figure
The TG curves of the DKDP crystals are shown in Figure
The effective distribution coefficient of deuterium is described by the following exponential equation[
| Samples | Residual mass (%) | Deuterium level (%) |
|---|---|---|
| DKDP-1# | 85.53191 | 97.22 |
| DKDP-2# | 85.53906 | 96.65 |
| Average | 85.53549 | 96.94 |
Table 4. Results of the residual mass and deuterium level of DKDP.
3.2 Optical properties of DKDP crystal
Highly deuterated DKDP crystals with excellent optical properties are widely used for optical applications. The absorption coefficient of DKDP crystals is a significant parameter used to assess the optical properties and quality of crystals, which was measured from the transmission spectra in this paper. Samples of
The highly deuterated DKDP crystal was used in electro-optical
| Samples | KDP | 70% DKDP | 97.9% DKDP | |||
|---|---|---|---|---|---|---|
| (%) | (%) | (%) | ||||
| 1# | 85.645 | 0.082 | 89.410 | 0.031 | 93.407 | 0.0052 |
| 2# | 86.122 | 0.071 | 89.707 | 0.028 | 93.407 | 0.0052 |
| Average | 85.884 | 0.077 | 89.559 | 0.029 | 93.407 | 0.0052 |
Table 5. Results for the transmittance and absorption coefficient with different deuterium levels in DKDP and KDP crystals.
In accordance with the Lambert–Beer law, the transmittance result is determined by the interface and internal transmittance in the crystal. The relationship can be expressed by the following formula[
3.3 LIDT of DKDP crystals
The LIDT is one of the most important properties of DKDP crystals and is a measure of the resistance to laser damage. As-grown DKDP crystal wafers with dimensions of
| Samples | LIDT | LIDT | LIDT |
|---|---|---|---|
| at 8.8 ns | at 3 ns | at 10 ns | |
| 43.9 | 25.6 | 46.8 | |
| 35.6 | 20.8 | 38.0 | |
| Average | 39.7 | 23.2 | 42.4 |
Table 6. LIDT results at 8.8 ns, 3 ns and 10 ns.
4 Conclusions
The properties of a highly deuterated DKDP crystal with sizes of
References
[1] N. P. Zaitseva, J. J. De Yoreo, M. R. Dehaven, R. L. Vital, K. E. Montgomery, M. Richardson, L. J. Atherton. J. Cryst. Growth, 180, 255(1997).
[2] N. Zaitseva, L. Carman. Prog. Cryst. Growth Charact. Mater., 43, 1(2001).
[3] J. J. De Yoreo, A. K. Burnham, P. K. Whitman. Int. Mater. Rev., 47, 113(2002).
[4] W. Han, F. Wang, L.-D. Zhou, F.-Q. Li, B. Feng, H.-B. Cao, J.-P. Zhao, S. Li, K.-X. Zheng, X.-F. Wei, M.-L. Gong, W.-G. Zheng. Opt. Express, 21, 30481(2013).
[5] S. G. Demos, R. N. Raman, S. T. Yang, R. A. Negres, K. I. Schaffers, M. A. Henesian. Opt. Express, 19, 21050(2011).
[6] W. Han, Y. Xiang, F.-Q. Li, F. Wang, L.-D. Zhou, J.-P. Zhao, B. Feng, K.-X. Zheng, Q.-H. Zhu, X.-F. Wei, W.-G. Zheng, M.-L. Gong. Appl. Opt., 54, 4167(2015).
[7] A. M. Vakulenko, N. G. Kryukov, Yu A. Matveets, V. I. Panteleev, Yu V. Senatskii, A. I. Fedosimov, V. T. Yurov. Soviet J. Quantum Electronics, 4, 76(1974).
[8] L.-S. Zhang, G.-W. Yu, H.-L. Zhou, L. Li, M.-X. Xu, B.-A. Liu, S.-H. Ji, L.-L. Zhu, F.-F. Liu, X. Sun. J. Cryst. Growth, 401, 190(2014).
[9] P. A. Baisden, L. J. Atherton, R. A. Hawley, T. A. Land, J. A. Menapace, P. E. Miller, M. J. Runkel, M. L. Spaeth, C. J. Stolz, T. I. Suratwala, P. J. Wegner, L. L. Wong. Fusion Sci. Technol., 69, 295(2017).
[10] S.-L. Wang, Z.-S. Gao, Y.-J. Fu, A.-D. Duan, X. Sun, C.-S. Fang, X.-Q. Wang. Cryst. Res. Technol., 38, 941(2003).
[11] M.-H. Jiang, C.-S. Fang, X.-L. Yu, M. Wang, T.-H. Zheng, Z.-S. Gao. J. Cryst. Growth, 53, 283(1981).
[12] N. P. Zaitseva, L. N. Rashkovich, S. V. Bogatyreva. J. Cryst. Growth, 148, 276(1995).
[13] L.-S. Zhang, F. Zhang, M.-X. Xu, Z.-P. Wang, X. Sun. RSC Adv., 5, 74858(2015).
[14] D.-W. Fu, W. Zhang, R.-G. Xiong. Cryst. Growth Des., 8, 3461(2008).
[15] S. G. Demos, P. DeMange, R. A. Negres, M. D. Feit. Opt. Express, 18, 13788(2010).
[16] M. A. Yakshin, D. W. Kim, Y. S. Kim, Y. Y. Broslavets, O. E. Sidoryuk, S. Goldstein. Laser Phys., 7, 941(1997).
[17] G. M. Loiacono, J. F. Balascio, W. Osborne. Appl. Phys. Lett., 24, 455(1974).
[18] T. Huser, C. W. Hollars, W. J. Siekhaus, J. J. De Yoreo, T. I. Suratwala, T. A. Land. Appl. Spectrosc., 58, 349(2004).
[19] J. Leroudier, J. Zaccaro, J. Debray, P. Segonds, A. Ibanez. Cryst. Growth Des., 13, 3613(2013).
[20] G.-H. Li, G.-B. Su, X.-X. Zhuang, Z.-D. Li, Y.-P. He. Opt. Mater., 29, 220(2006).
[21] F.-F. Liu, M.-X. Xu, B.-A. Liu, X.-P. Chen, L. Xie, Y.-H. Xia, C.-W. Wen, L.-S. Zhang, X. Ju, G.-G. Sun, X. Sun. Opt. Mater. Express, 6, 2221(2016).
[22] X.-Y. Xie, H.-J. Qi, B. Wang, H. Wang, D.-Y. Chen, J.-D. Shao. J. Cryst. Growth, 487, 45(2018).
[23] D.-T. Cai, Y.-F. Lian, X.-X. Chai, L.-S. Zhang, L.-M. Yang, M.-X. Xu. Cryst. Eng. Comm., 20, 7357(2018).
[24] M.-X. Xu, X. Sun, Z.-P. Wang, X.-F. Cheng, S.-T. Sun, L.-L. Ji, Y.-A. Zhao, B.-A. Liu, H. Gao, X.-G. Xu. Cryst. Res. Technol., 45, 763(2010).
[25] X.-D. Wang, B.-T. Tian, Y.-Y. Niu, G.-M. Wu, B. Zhou, J. Shen. Rare Metal Mat. Eng., 45, 370(2016).
Set citation alerts for the article
Please enter your email address



