Jian Zheng, Dajun Shu. Regulation of surface properties of photocatalysis material TiO2 by strain engineering[J]. Journal of Semiconductors, 2020, 41(9): 091703
Search by keywords or author
- Journal of Semiconductors
- Vol. 41, Issue 9, 091703 (2020)
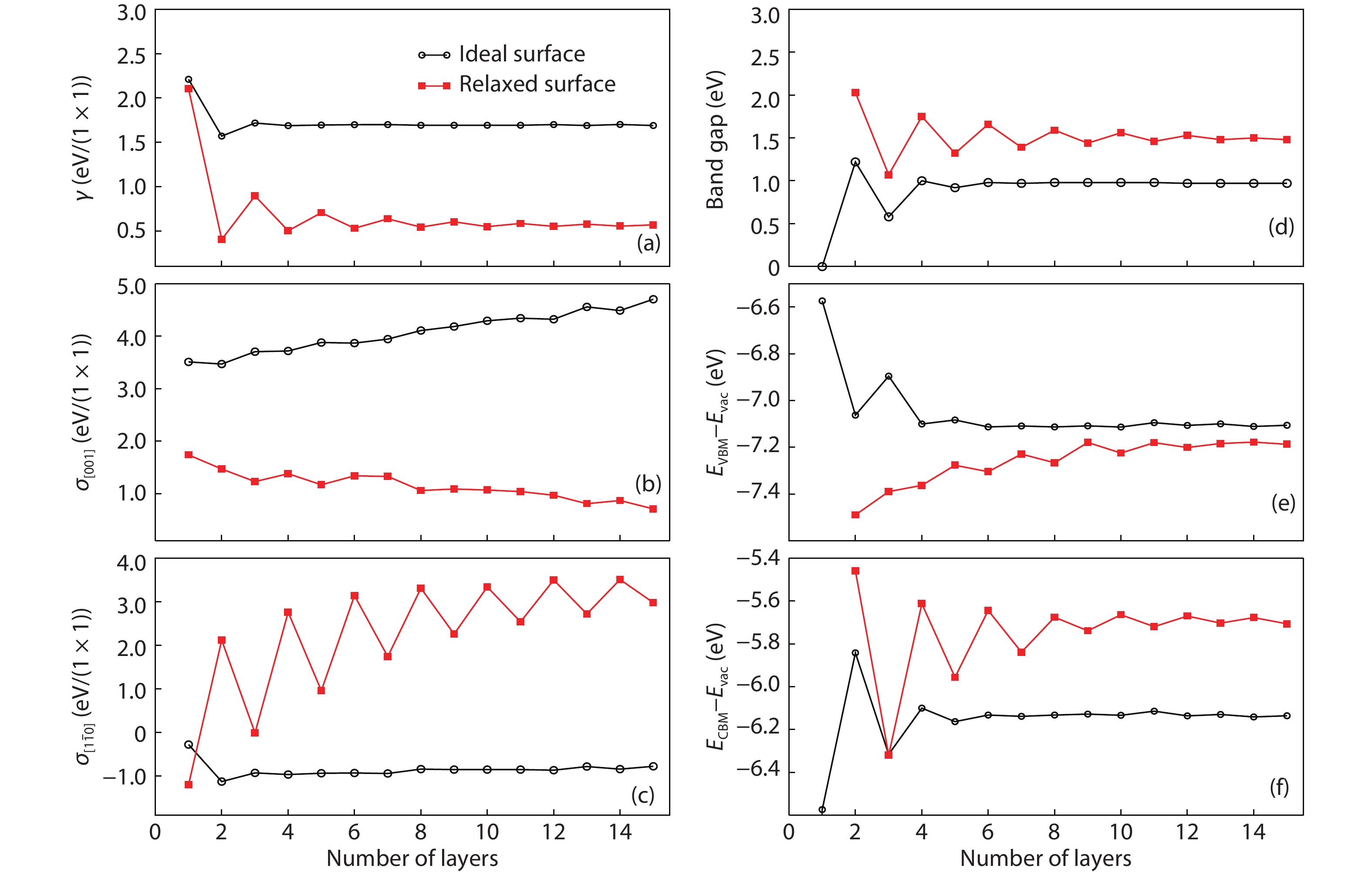
![(Color online) (a–c) Variations of surface energies and surface stresses, as well as (d–f) the electronic structure properties with the slab thickness. Black solid curves and red dashed curves are for the unrelaxed and relaxed surfaces, respectively. The valence band maximum (VBM,(e)) and the conduction band minimum (CBM,(f)) are referenced to the vacuum level. These figures are adapted from Ref. [14].](/richHtml/jos/2020/41/9/091703/img_1.jpg)
Fig. 1. (Color online) (a–c) Variations of surface energies and surface stresses, as well as (d–f) the electronic structure properties with the slab thickness. Black solid curves and red dashed curves are for the unrelaxed and relaxed surfaces, respectively. The valence band maximum (VBM,(e)) and the conduction band minimum (CBM,(f)) are referenced to the vacuum level. These figures are adapted from Ref. [14 ].
![(Color online) (a) The area percentage of anatase (001) facets with variation of the applied strain. The inset displays the equilibrium shape of the anatase crystallite. (b –d) The surface energy ratio and the ratio of with the variation of the applied strain. The anatase (001) surface appears only when the surface energy ratio is smaller than . These figures are adapted from Ref. [13].](/richHtml/jos/2020/41/9/091703/img_2.jpg)
Fig. 2. (Color online) (a) The area percentage of anatase (001) facets with variation of the applied strain. The inset displays the equilibrium shape of the anatase crystallite. (b –d) The surface energy ratio
13 ].
Fig. 3. (Color online) Schematic of the rutile TiO2(110) surface. The bridging oxygen (BO), the subbridging oxygen (SBO) and the in-plane oxygen (IPO) atoms are indicated by the arrows.
12 ].
Fig. 4. (Color online) (a) The formation energy of OV and (b) the changes of the surface stresses and surface elastic constants induced by the OV as a function of the OV depth. The depth is defined as the distance below the unrelaxed 1-bridging O atoms. The dashed horizontal line in (a) denotes the bulk vacancy formation energy. Solid or dashed curves in (b) are to guide the eyes.
10 ].
Fig. 5. Phase diagram of the type of the energetically most favorable OV as a function of the external strain
Table 3 . It indicates that BOV, IPOV, or SBOV can be energetically most favorable within different range of the external strain. This figure is adapted from Ref. [11 ].
Fig. 6. (Color online) Energy profiles of OV pathways within the primitive cell of rutile TiO
12 ].
Fig. 7. (Color online) (a) Schematic of bulk OV diffusion along the [110] and
12 ].
Fig. 8. (Color online) The strain-dependent diffusion barrier of the surface OV along [001] (Path I) and along
11 ].
Fig. 9. (Color online) Adsorption energies per water molecule in (a) molecular state and (b) dissociative state on the surface with different supercell size and different thickness
x and y direction of the surface is along [001] and
14 ] by the permission of PCCP.
Fig. 10. (Color online) The change of the total energy of the s-TiO2(110) surface induced by the in-plane polarization under strain along
15 ] by the permission of PCCP.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 1. The elastic constant
B ), Young’s modulus along the
r direction (
13 ].
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. The calculated surface energy and surface mechanical properties of rutile and anatase phases.
13 ].
|
Table 3. Changes of surface energy, surface stress and surface elasticity induced by different types of oxygen vacancies. The units are eV/
10 , 11 ].
Set citation alerts for the article
Please enter your email address



