- Journal of Semiconductors
- Vol. 41, Issue 9, 091703 (2020)
Abstract
1. Introduction
Titanium dioxide is a promising photocatalysis material, which also has wide applications in heterogeneous catalysis, solar cells, gas sensor, etc[
Strain field is unavoidable in fabrication of nanostructures and thin film[
In this paper, we review our work on the strain engineering of the surface properties of TiO2. The following section is arranged as follows. In the second section, we will first introduce the simple theoretical background on the strain engineering. In the third section, we will discuss how the strain can be used to improve the relative percentage of the reactive surface. In the fourth and fifth sections, the thermodynamic and kinetic properties of oxygen vacancies under strain are reviewed, respectively. In the sixth and seventh sections, the effect of strain on the surface adsorption on stoichiometric and reduced TiO2(110) are reviewed, respectively.
2. Elastic properties of TiO2 surface
The structure and thus the properties of a bulk material respond to the external strain differently according to its bulk elastic properties. The bulk elastic properties of TiO2 are described by six independent elastic constants. The bulk modulus and Young’s modulus can be further calculated by these independent elastic constants. The results are presented in Table 1, including our own calculations based on the density functional theory (DFT)[
Similar to the case of a bulk material, the response of a surface to the external strain depends on the surface elastic properties. Generally speaking, different surface structures also have different surface elasticity. Therefore, the thermodynamic equilibrium surface structures would adjust themselves according to the external strain, and the surface properties change with the strain correspondingly.
The surface elastic properties can be described mainly by the surface stresses and surface elastic constants, which are defined as follows[
Here
Under a certain strain, by comparing the surface energies of different surface structures, one can expect to investigate the effect of strain on the surface thermodynamic or kinetic properties.
The surface elastic properties of TiO2 are shown in Table 2, the calculation details of which can be found in Ref. [13]. The surface elastic constants
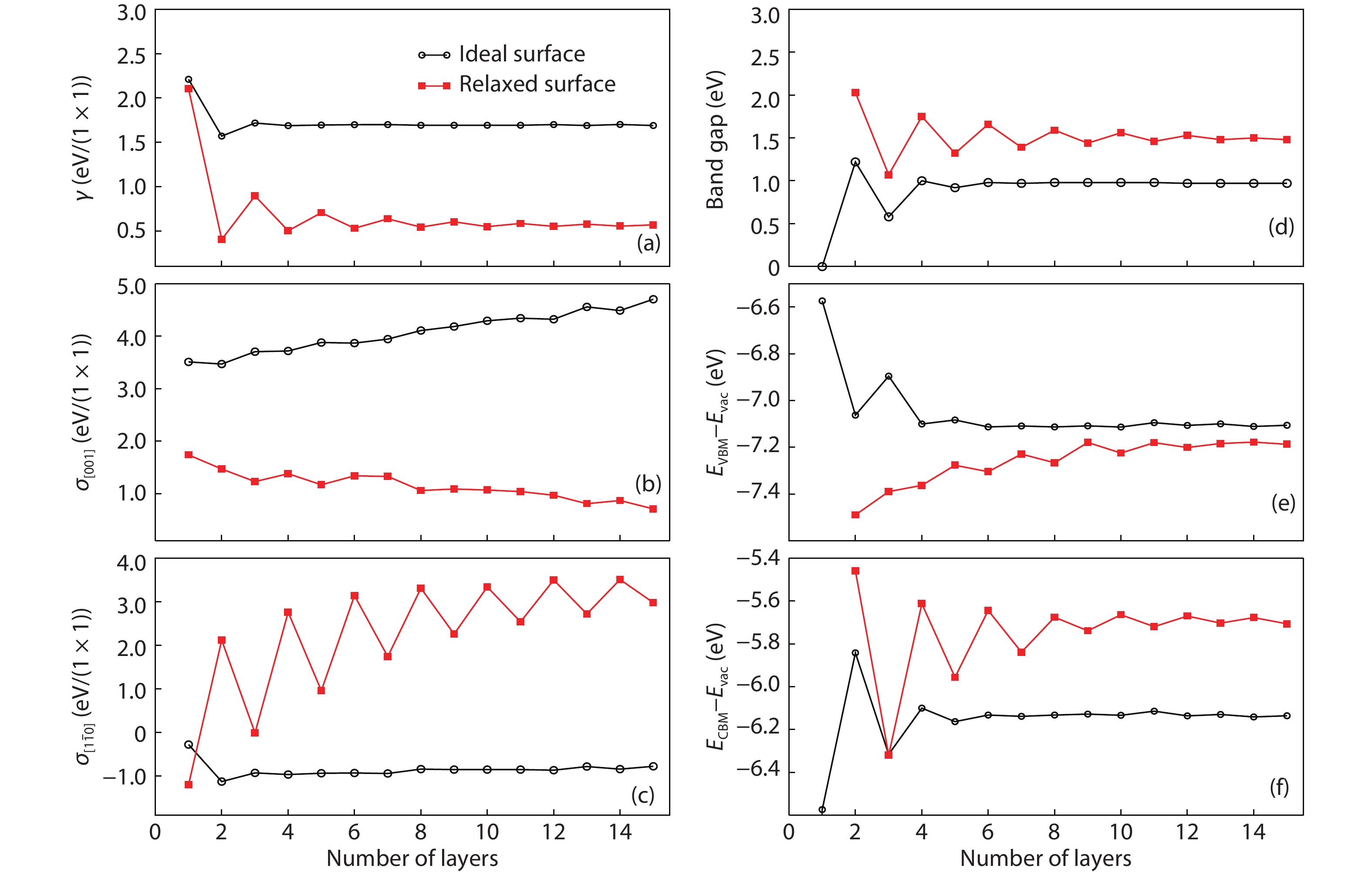
It is worthy to note that there is an odd–even oscillation of the surface properties of the relaxed TiO2 in the thin film slab. For instance, for rutile (110) surface, although the surface energy and the band gap converge in a slab within about ten trilayers, the surface stresses in the
![]()
Figure 1.(Color online) (a–c) Variations of surface energies and surface stresses, as well as (d–f) the electronic structure properties with the slab thickness. Black solid curves and red dashed curves are for the unrelaxed and relaxed surfaces, respectively. The valence band maximum (VBM,(e)) and the conduction band minimum (CBM,(f)) are referenced to the vacuum level. These figures are adapted from Ref. [
3. Improving the percentage of reactive surfaces
Generally speaking, there is an intrinsic controversy between stability and reactivity in nature. The more reactive a surface is, the less stable it is, or the higher surface energy it is. According to the Wulff's theorem, it means that the percentage of a reactive surface is usually low. For example, in equilibrium anatase TiO2, the most reactive (001) surface has a surface energy much larger than the (101) surface, as shown in Table 2. As a result, the area proportion of anatase (001) surface is no more than 3%, which strongly limits the reactive efficiency of TiO2 in application as a photocatalyst[
Fortunately, the anatase (001) surface possesses the largest surface stress among all the surfaces, which is positive and occurs along the [100] direction. We therefore expect that the relative stability of (001) surface can be significantly improved by applying an compressive strain along the [100] direction, and thus its area percentage of anatase (001) according to Wulff construction. By first principle calculations, we have verified this[
![]()
Figure 2.(Color online) (a) The area percentage of anatase (001) facets with variation of the applied strain. The inset displays the equilibrium shape of the anatase crystallite. (b –d) The surface energy ratio
4. Formation of surface oxygen vacancies under strain
Oxygen vacancies are the most common defects in transition metal oxide. The reduced materials usually show different physical properties compared to the stoichiometric one. In TiO2, the oxygen vacancy (OV) has a low formation energy, and it has been experimentally found reactive in the surface reaction[
There are three types of oxygen atoms, bridging, in-plane and subbridging oxygen atoms, as shown in Fig. 3. Correspondingly, three different types of OV can be introduced by removing corresponding different types of oxygen atoms, namely BOV, IPOV and SBOV. We named different types of OVs to include the site of the defect. For example, 2-in-plane OV represents the OV formed by removing an in-plane oxygen atom in the second trilayer.
![]()
Figure 3.(Color online) Schematic of the rutile TiO2(110) surface. The bridging oxygen (BO), the subbridging oxygen (SBO) and the in-plane oxygen (IPO) atoms are indicated by the arrows.
The OV formation energy can be calculated as follows,
where
The OV formation energy as a function of OV depth is shown in Fig. 4(a). Obviously the 1-bridging OV is the most easily formed OV defect because of the lowest formation energy. In addition, the in-plane OV formation energy remains almost constant at different depth, while the bridging and subbridging OVs formation energies appear a kind of odd-even oscillation behavior. With the position of OV going deeper, both of them tend to approach the formation energy of bulk OV[
![]()
Figure 4.(Color online) (a) The formation energy of OV and (b) the changes of the surface stresses and surface elastic constants induced by the OV as a function of the OV depth. The depth is defined as the distance below the unrelaxed 1-bridging O atoms. The dashed horizontal line in (a) denotes the bulk vacancy formation energy. Solid or dashed curves in (b) are to guide the eyes.
To get the influence of oxygen vacancies on the surface elasticity, we also calculated the vacancy-induced change of surface energy,
where
The value of
By substituting the data in Table 3 into Eq. (2), a phase diagram can be obtained for the most stable oxygen vacancy in the first trilayer[
![]()
Figure 5.Phase diagram of the type of the energetically most favorable OV as a function of the external strain
It is worthy to mention that we did not realize until recently that the incipient ferroelectricity of TiO2 is also quite important for its surface properties. Under tensile strain, it tends to be more stable in the ferroelectric polarization state with lower symmetry[
5. Diffusion kinetics of oxygen vacancies
The distribution of defects can also be influenced by the diffusion kinetics. We have extensively studied the diffusion properties of OVs in TiO2 and the effects of strain on the kinetics[
![]()
Figure 6.(Color online) Energy profiles of OV pathways within the primitive cell of rutile TiO
When a strain is applied along [001] direction or biaxially along [100] and [010], it would not break the symmetry. In this case, we found that the diffusion barrier is reduced by tensile strain and enhanced by compressive strain. If, however, the strain is just applied in [110] and
![]()
Figure 7.(Color online) (a) Schematic of bulk OV diffusion along the [110] and
The diffusion barrier of OV on the surface is much lower than that of bulk OV. However, a highest diffusion barrier of OV occurs between the second trilayer and the third one, which hinders the OVs in bulk from diffusing to the surface[
The diffusion of OVs within the (110) surface is also anisotropic. As shown in Fig. 8, the surface bridging OV diffuses more rapidly along
![]()
Figure 8.(Color online) The strain-dependent diffusion barrier of the surface OV along [001] (Path I) and along
6. Water adsorption and dissociation energy on s-TiO2(110)
Since the hydroxyl ions and protons introduced by water dissociation play key roles in most relevant surface reactions, the adsorption and dissociation of water on TiO2 surfaces have been extensively studied in the past years. We have found that the adsorption and dissociation of water on stoichiometric rutile (110) surface depend on both the coverage and the thin film thickness[
where
As shown in Fig. 9, both the adsorption and dissociation of water are energetically more favorable when the thin film has an odd O–Ti–O trilayers. Especially, when the adsorbed water has no neighboring one along the [001] direction, the dissociative water is more favorable on the TiO2(110) surface of a thin film with three trilayer. This can be easily understood by referring to Fig. 1(a), since the slab containing an odd number of layers is less stable and thus more reactive than the one of even number of layers. With increasing thickness of the thin film or increasing coverage of the water, both the adsorption and the dissociation of water become less favorable.
![]()
Figure 9.(Color online) Adsorption energies per water molecule in (a) molecular state and (b) dissociative state on the surface with different supercell size and different thickness
The adsorption of water is more energetically favorable on thin films under tensile strain. Besides, the dissociation barrier of the water also decreases with increasing tensile strain[
7. Adsorption and dissociation on reduced TiO2(110): Effect of strain via polarization
As mentioned above, the rutile TiO2 is a well-known incipient ferroelectric material. Previous theoretical studies suggested that the transition can occur under negative pressure or uniaxial strain. For the most stable TiO2(110) surface, spontaneous in-plane polarization is predicted to occur under tensile strain. This strain-induced in-plane polarization is expected to play roles in the surface reactivity of the TiO2(110) surface, which we have just explored[
By studying the adsorption of a series of molecules on the reduced TiO2(110) surface under tensile strain, we found that the stabilities of the polarized structure relative to the non-polarized one are determined by the surface doping caused by the charge transfer between the adsorbates and the surface. The polarized structure favors the surface adsorption, and vice versa, if the adsorbate captures extra electrons of the reduced TiO2 surface and introduces no additional holes. It is because the free carriers would screen the long-range Coulomb interactions and suppress the spontaneous polarization. The electrostatic interaction between the polar molecule and the in-plane polarization is of less importance relative to the effect of the surface doping.
The adsorbates we calculated were a series of chalcogen-containing molecules, including H2O, H2S, O2 and CO2, in molecular state and dissociative state. Since the molecular adsorption of H2S is unstable against partial dissociation, we only considered the adsorption of H2S in a partial dissociative or complete dissociative state, denoted respectively as (D
We calculated the total energies of reduced TiO2(110) after surface adsorption. The energies are denoted as
In order to understand the difference between the two different categories of adsorbates, the density of states (DOS) of r-TiO2(110) with different adsorbates were compared. We found that the structures have similar
In order to further verify the conclusion, we charged the polarized and non-polarized s-TiO2(110) surface by simply adding and removing electrons, and then calculated the energy difference
![]()
Figure 10.(Color online) The change of the total energy of the s-TiO2(110) surface induced by the in-plane polarization under strain along
8. Summary
In this paper, we have reviewed our work within recent ten years on the strain engineering of TiO2 surface properties. We found that when suitable external strain is applied in specific directions, the surface and bulk properties undergo a significant change, due to the difference in the elastic properties of different surface structures. We thus propose that the external strain is a good way to engineer the physical and chemical properties of TiO2 and to further improve the catalytic reactivity. We hope our work will stimulate more experimental studies.
Acknowledgements
The numerical calculations were carried out at the High Performance Computing Center of Nanjing University. This work was supported by the National Natural Science Foundation of China (Grants No. 11974164).
References
[1] R Asahi, T Morikawa, T Ohwaki et al. Visible-light photocatalysis in nitrogen-doped titanium oxides. Science, 293, 269(2001).
[2] C T Campbell, S C Parker, D E Starr. The effect of size-dependent nanoparticle energetics on catalyst sintering. Science, 298, 811(2002).
[3] O Bikondoa, C L Pang, R Ithnin et al. Direct visualization of defect-mediated dissociation of water on TiO2(110). Nat Mater, 5, 189(2006).
[4] M Gratzel. Photoelectrochemical cells. Nature, 414, 338(2001).
[5] G Lu, A Linsebigler, J T Yates. Photooxidation of ch3cl on TiO2(110) — a mechanism not involving H 2O. J Phys Chem, 99, 7626(1995).
[6] X Y Pan, M Q Yang, X Z Fu et al. Defective TiO2 with oxygen vacancies: Synthesis, properties and photocatalytic applications. Nanoscale, 5, 3601(2013).
[7] B Yang, F Liu, M G Lagally. Local strain-mediated chemical potential control of quantum dot self-organization in heteroepitaxy. Phys Rev Lett, 92, 4(2004).
[8] G H Lu, M Cuma, F Liu. First-principles study of strain stabilization of Ge(105) facet on Si(001). Phys Rev B, 72, 6(2005).
[9] W J Yin, S Y Chen, J H Yang et al. Effective band gap narrowing of anatase TiO2 by strain along a soft crystal direction. Appl Phys Lett, 96, 3(2010).
[10] D J Shu, S T Ge, M Wang et al. Interplay between external strain and oxygen vacancies on a rutile TiO2(110) surface. Phys Rev Lett, 101, 4(2008).
[11] Z W Wang, D J Shu, M Wang et al. Diffusion of oxygen vacancies on a strained rutile TiO2(110) surface. Phys Rev B, 82, 7(2010).
[12] Z W Wang, D J Shu, M Wang et al. Strain effect on diffusion properties of oxygen vacancies in bulk and subsurface of rutile TiO2. Surf Sci, 606, 186(2012).
[13] L Jia, D J Shu, M Wang. Tuning the area percentage of reactive surface of TiO2 by strain engineering. Phys Rev Lett, 109, 5(2012).
[14] L Yang, D J Shu, S C Li et al. Influence of strain on water adsorption and dissociation on rutile TiO2(110) surface. Phys Chem Chem Phys, 18, 14833(2016).
[15] Z W Wang, D J Shu. Intrinsic interaction between in-plane ferroelectric polarization and surface adsorption. Phys Chem Chem Phys, 21, 18680(2019).
[16] E Shojaee, M R Mohammadizadeh. First-principles elastic and thermal properties of TiO2: A phonon approach. J Phys Condens Matter, 22, 8(2010).
[17] H Sato, K Ono, T Sasaki et al. First-principles study of two-dimensional titanium dioxides. J Phys Chem B, 107, 9824(2003).
[18] D G Isaak, J D Carnes, O L Anderson et al. Elasticity of TiO2rutile to 1800 K. Phys Chem Miner, 26, 31(1998).
[19] R C Cammarata. Surface and interface stress effects in thin-films. Prog Surf Sci, 46, 1(1994).
[20] A Vittadini, M Casarin, A Selloni. Chemistry of and on TiO2-anatase surfaces by dft calculations: a partial review. Theor Chem Acc, 117, 663(2007).
[21] M Lazzeri, A Vittadini, A Selloni. Structure and energetics of stoichiometric TiO2anatase surfaces. Phys Rev B, 63, 9(2001).
[22] U Diebold, N Ruzycki, G S Herman et al. One step towards bridging the materials gap: surface studies of TiO2 anatase. Catal Today, 85, 93(2003).
[23] S Wendt, R Schaub, J Matthiesen et al. Oxygen vacancies on TiO2(110) and their interaction with H2O and O2: A combined high-resolution STM and DFT study. Surf Sci, 598, 226(2005).
[24] T L Thompson, J T Yates. Surface science studies of the photoactivation of TiO2 – new photochemical processes. Chem Rev, 106, 4428(2006).
[25] C M Yim, C L Pang, G Thornton. Oxygen vacancy origin of the surface band-gap state of TiO2(110). Phys Rev Lett, 104, 4(2010).
[26] U Diebold, J Lehman, T Mahmoud et al. Intrinsic defects on a TiO2(110)(1x1) surface and their reaction with oxygen: A scanning tunneling microscopy study. Surf Sci, 411, 137(1998).
[27] E Wahlstrom, N Lopez, R Schaub et al. Bonding of gold nanoclusters to oxygen vacancies on rutile TiO2(110). Phys Rev Lett, 90, 4(2003).
[28] R Schaub, E Wahlstrom, A Ronnau et al. Oxygen-mediated diffusion of oxygen vacancies on the TiO2(110) surface. Science, 299, 377(2003).
[29] Z R Zhang, R Rousseau, J L Gong et al. Vacancy-assisted diffusion of alkoxy species on rutile TiO2(110). Phys Rev Lett, 101, 4(2008).
[30] G A Kimmel, N G Petrik. Tetraoxygen on reduced TiO2(110): Oxygen adsorption and reactions with bridging oxygen vacancies. Phys Rev Lett, 100, 4(2008).
[31] D O Klenov, W Donner, B Foran et al. Impact of stress on oxygen vacancy ordering in epitaxial (La0.5Sr0.5)CoO3-partial derivative thin films. Appl Phys Lett, 82, 3427(2003).
[32] H Kamisaka, K Yamashita. The surface stress of the (110) and (100) surfaces of rutile 14 and the effect of water adsorbents. Surf Sci, 601, 4824(2007).
[33] K Hameeuw, G Cantele, D Ninno et al. Influence of surface and subsurface defects on the behavior of the rutile TiO2(110) surface. Phys Status Solidi A, 203, 2219(2006).
[34] T Bredow, L Giordano, F Cinquini et al. Electronic properties of rutile TiO2ultrathin films: Odd-even oscillations with the number of layers. Phys Rev B, 70, 6(2004).
[35] C Lee, P Ghosez, X Gonze. Lattice-dynamics and dielectric-properties of incipient ferroelectric TiO2 rutile. Phys Rev B, 50, 13379(1994).
[36] R A Parker. Static dielectric constant of rutile (TiO2), 1.6–1060 °K. Phys Rev, 124, 1719(1961).
[37] J G Trayler, H G Smith, R M Nicklow et al. Lattice dynamics of rutile. Phys Rev B, 3, 3457(1971).
Set citation alerts for the article
Please enter your email address



