Wa GAO, Yujie XIONG, Congping WU, Yong ZHOU, Zhigang ZOU. Recent Progress on Photocatalytic CO2 Reduction with Ultrathin Nanostructures[J]. Journal of Inorganic Materials, 2022, 37(1): 3
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 37, Issue 1, 3 (2022)
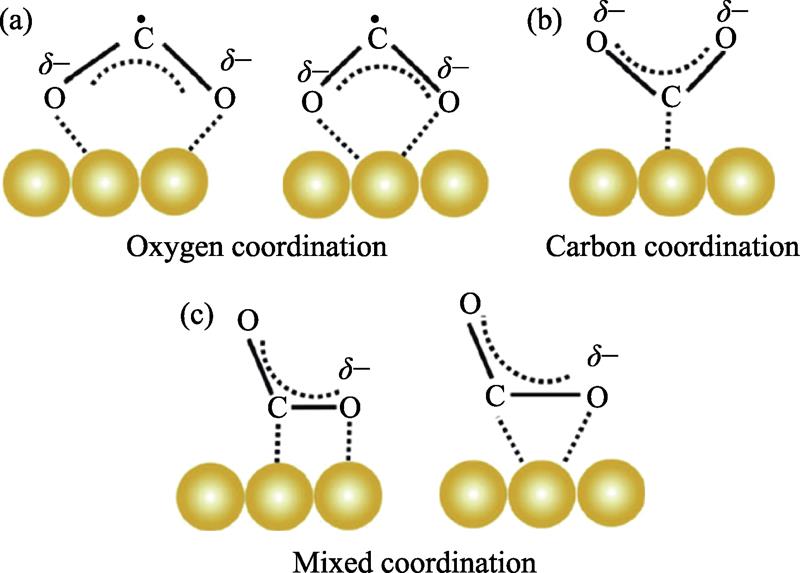
![Different CO2 adsorption modes on the surface of photocatalysts[4]](/richHtml/jim/2022/37/1/3/img_1.png)
. Different CO2 adsorption modes on the surface of photocatalysts[4]
![Possible reaction paths for CO2 reduction to produce HCHO, CH3OH, and CH4[35,36]](/richHtml/jim/2022/37/1/3/img_2.png)
. Possible reaction paths for CO2 reduction to produce C2H4, CH3CHO, and C2H5OH[35]
. (a) Calculated band positions of the WO3 nanosheet and commercial WO3, relative to the redox potential of CO2/CH4 in the presence of water, and (b) CH4 generation over the nanosheet and commercial powder as a function of visible light irradiation time (λ≥420 nm)[38]
. Height images of (a) atomically thin InVO4 nanosheet, (b) nanocube, and (c) bulk materials obtained by conventional solid-state reaction, surface photovoltage spectroscopy (SPV) images in (a′), (b′), and (c′) displaying differential images between potential images under light and in the dark, and (d) surface photovoltage change by subtracting the potential under dark conditions from that under illumination (SPV, ΔCPD = CPDdark - CPDlight)[13]
. Band structures of PCMT@In2O3/ZIS[47]
. Photocatalytic (a) CO and (b) CH4 output changing with light irradiation time, (c) comparison of photocatalytic activity over different samples, (d)schematic illustration of the photocatalytic CO2 reduction for ZnIn2S4/BiVO4 nanocomposite, schematic representation of (e) Z-scheme electron-hole transfer mechanisms, and (f) heterojunction-type electron-hole transfer mechanisms under light irradiation[50]
. TEM images of (a, b) poly(methylmethacrylate) spheres coated with (protonic polyethylenimine (PEI)/Ti0.91O2/ PEI/GO)5, (c, d) (G-Ti0.91O2)5 hollow spheres, and (e)comparation of the average product formation rates[53]
. (a, b) SEM images of InVO4/Ti3C2Tx at higher magnification, (c) HRTEM images of InVO4/Ti3C2Tx, (d)scheme for spatial charge separation and transport during the photocatalytic reduction of CO2 over hierarchical InVO4/Ti3C2Tx heterosystem, and (e)energy level alignment of InVO4/Ti3C2Tx hybrid[56]
. Schematic illustration of the preparation procedure of the Au-TiO2 composites (b), schematic illustration of charge separation and transfer in the Au-TiO2 system and photoreduction of CO2 into different products[57]
. (a) Scheme of the electronic band structures of Vo-rich WO3 atomic layers and WO3 atomic layers, and (b) in situ FT-IR spectra for the IR light-driven CO2 reduction process on the Vo-rich WO3 atomic layers[60]
|
Table 1. Standard potentials of convert CO2 to various C1 and C2 products in aqueous solutions at standard conditions (1.01×105 Pa and 25 ℃) [34]
Set citation alerts for the article
Please enter your email address



