Dandan YANG, Xiaoming LI, Cuifang MENG, Jiaxin CHEN, Haibo ZENG. Research Progress on the Stability of CsPbX3 Nanocrystals [J]. Journal of Inorganic Materials, 2020, 35(10): 1088
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 35, Issue 10, 1088 (2020)
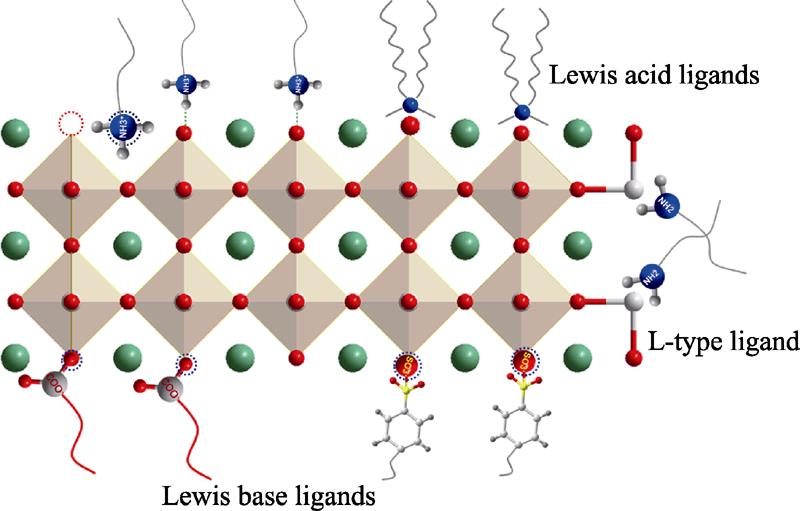
1. Schematic diagram of passivation strategies for different ligands on the surface of CsPbX3 nanocrystals
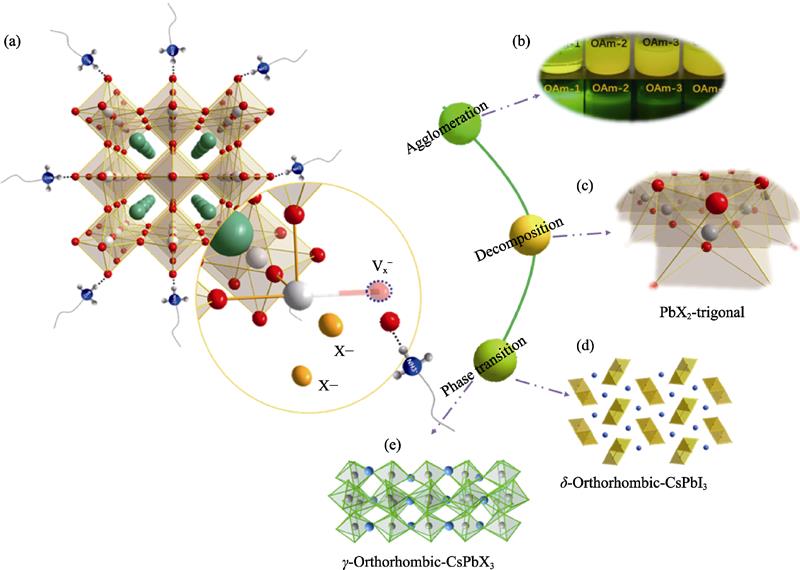
2. (a) Schematic diagram of instability mechanism of CsPbX3 nanocrystals, (b) agglomeration and (c) decomposing product of CsPbX3 nanocrystals, and schematic diagram of (d) phase transition (non-perovskite phase) and (e) phase transition (perovskite phase)
3. (a) Passivation strategy based on hard Lewis acid ligands (left), images under 365 nm UV light (middle), and photoluminescence decay curves (right) of nanocrystals with high and low defect densities[25]; (b) Schematic diagram of DETAI3 surface passivation strategy (left), absorption curves (middle) and long-term phase stability (right) of CsPbI3‧x DETAI3 thin films[29]
4. (a) Schematic diagram of Lewis base surface passivation strategy for CsPbX3 nanocrystals; (b) Theoretical calculation of the binding energy of mono- and dicarboxylic acids on the surface of CsPbI3 nanocrystals[34]; (c) Schematic diagram of OPA and OAm-CsPbX3 surface passivation strategies and photos after multiple purifications[35]; (d) Surface passivation strategy with zwitterionic ligands (sulfobetaines, phosphocholines and γ -amino acids)[36]
5. (a) Passivation strategies with different ligands (OAm, OA and DBSA) on the surface of CsPbBr3 nanocrystals and (b) the corresponding exciton recombination processes[37]; (c) Schematic diagram of PbBrx -rich surface of OAm-CsPbBr3 nanocrystals (left) and Br-rich surface of OAm/OA-CsPbBr3 nanocrystals (right)[11]; (d) Photographs showing the resistance of different samples against water treatment of OAm/OA-CsPbBr3 nanocrystals (above) and OAm-CsPbBr3 nanocrystals (below)[11]
6. (a) Schematic diagram of CPB-DBAE@SiO2 preparation process[51]; (b) Transmission electron microscopy (TEM) and high resolution transmission electron microscopy (HRTEM) images of CsPbBr3@SiO2 nanocrystals and photographs of water stability[60]; (c) Schematic representation (left), HRTEM image (middle), and plot of emission intensity under continuous pulsed laser irradiation (right) of CsPbBr3/CdS nanocrystals[67]
|
Table 1. Surface passivation strategies of Lewis acid ligands
|
Table 2. Surface passivation strategies of Lewis base ligands
| ||||||||||||||||||||||||||||||||||||||
Table 3. Characteristics, advantages and disadvantages of different passivation strategies

Set citation alerts for the article
Please enter your email address



