- Journal of Semiconductors
- Vol. 42, Issue 10, 101607 (2021)
Abstract
1. Introduction
Owing to the flexibility, portability, transparency, and low manufacturing cost, polymer solar cells (PSCs) have great potential for commercial production and application[
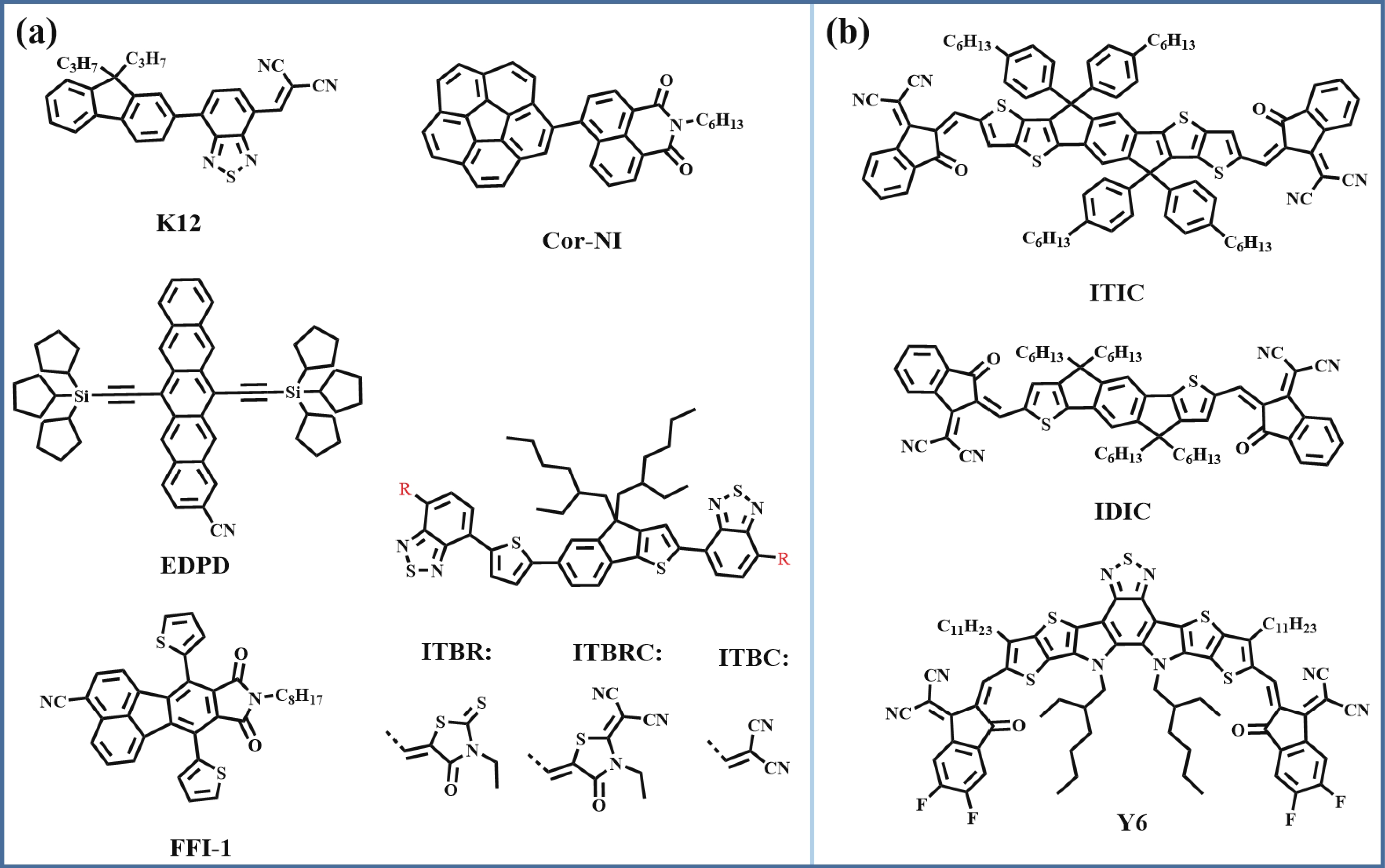
From 2011–2014, plenty of NF-SMAs have been designed with various electron-withdrawing groups such as indanedione[
![]()
Figure 1.Chemical structures of (a) early reports of A-NF-SMAs, (b) early reports of small molecule acceptors.
The asymmetric design of the molecule helps increase the dipole moment and dielectric constant and reduces the exciton binding energy, which is very beneficial to exciton dissociation and charge transport[
Generally speaking, Voc and Jsc are two iconic device performance parameters, and the increase of the value of Voc ×Jsc generally shows the improvement of device performance. Further optimize the value of Voc × Jsc through asymmetric molecular design to obtain higher device performance, which provides the latest research ideas and unique insights for PSCs research[
![]()
Figure 2.(a) A typical current–voltage
There have been related review articles summarizing the latest development of A-NF-SMAs, but its research content focused on the PSCs with a five-membered indaceno[1,2-b:5,6-b']dithiophene (IDT) series of A-NF-SMAs[
This review summarizes asymmetric non-fullerene polymer solar cells (A-NF-PSCs)-related content from three aspects: the structural advantages of the asymmetric system, the development history, and the research results in recent years. Meanwhile, we have also made a particular outlook on future research prospects, hoping to provide some guiding insights on the development of PSCs in the future.
2. Photophysical properties of OSCs
The structure and mechanism of photovoltaic devices have been discussed and studied in many excellent studies[
![]()
Figure 3.Simplified schematic of photoconversion in OSCs with the processes of photon absorption, exciton diffusion, exciton dissociation by charge transfer, and charge carrier collection denoted. Reproduced with permission from Ref. [
![]()
Figure 4.Chemical structures of polymer donors.
The photophysical characteristics of OSCs are generally represented by current density–voltage curves (J–V)[
(1) Voc
Under light conditions, Voc is the voltage at which the positive and negative poles of the OSCs are in an open circuit state, that is, the maximum output voltage of the OSCs[
(2) Jsc
Jsc is the current per unit area when the positive and negative electrodes of the OSCs are in a short-circuit state, that is, the maximum output current density of the OSCs in the light environment. As the band-gap of the material decreases, the value of Jsc increases and can be affected by the electron and hole transport efficiency of the active material[
(3) Fill factor (FF)
FF is a dimensionless physical quantity, which is the ratio of the maximum output power (Pmax) of the OSCs device to the product of Voc and Jsc, which can be expressed by Eq. (1).
FF is mainly related to the transfer and collection process of charge carriers. The balance of hole/electron mobility in the active layer, the degree of recombination during carrier transport, and the collection efficiency of carriers reaching the buffer layer will all affect the FF value of OSCs. The FF suggests how swiftly the charges can be removed from the cells and the ideal value is 1.0. Several factors can affect the FF of OSCs and they often interact in intricate ways[
(4) Power conversion efficiency (PCE)
PCE is the percentage of incident light energy converted into effective electric energy, which can be expressed by the ratio of Pmax of OSCs to incident power (Pin). In this case, the current density and applied bias voltage are expressed by Jmax and Vmax respectively, the calculation formula of PCE is as follow[
It can be seen that the PCE is jointly determined by Voc, Jsc, and FF, and represents the ability of OSCs to convert solar energy into electrical energy.
(5) External quantum efficiency (EQE)
The ratio of the number of electrons that can be collected under a certain wavelength of radiation to the number of incident photons at that wavelength can be expressed by the following Eq. (3):
Among them, λ is the wavelength of the incident light, and Pin is the power of the incident light. EQE is the product of light absorption efficiency, exciton diffusion and dissociation efficiency, charge transfer efficiency, and charge collection efficiency in the photoelectric conversion process. The EQE and Jsc can be mutually verified, and high EQE is the prerequisite for realizing high-efficiency OSCs[
3. Asymmetric non-fullerene acceptors based on A–D–A structures
Previous studies on A-NF-PSCs have been reviewed and published[
3.1. A-NF-SMAs with asymmetric cores
The structure of A-D-A is usually composed of one electron-donating unit and two electron-withdrawing units. This push–pull electron behavior in the molecule contributes to ICT so that the molecule has a strong transition dipole and wide spectral absorption range[
In the related research on the asymmetry of the core unit, the dipole moment and arrangement of the molecule can be adjusted by changing the number of asymmetric thiophene units and introducing heavy atoms. In the core unit of electron donation, the IDT core and the heptacyclic indacenodithieno[3,2-b]thiophene (IDTT) core play an important foundational role in the design of high-performance non-fullerene receptors, a new type of asymmetric ladder-type thiophene-phenylenethieno[3,2-b]thiophene-fused (TPTT) core combines the structural features of IDT and IDTT. At the same time, the π conjugate length of TPTT and the electron-donating capacity are all between IDT and IDTT. TPTT-IC (its structure is the same as T-TT), TPTT-2F, T-TT-4F, T-TT-4Cl, IDT6CN and IDT6CN-M are all molecules designed based on the TPTT core (Fig. 5). They have terminal groups which are different from each other. Their maximum absorption wavelengths range from 693 nm to 808 nm, and their LUMO energy levels range from –3.37 to –4.04 eV[
![]()
Figure 5.Chemical structures of A–D–A asymmetric non-fullerene acceptors without nitrogen.
In the combination of monothiophene and trithiophene, extending the IDT nuclear conjugation length can expand the spectral absorption, shift the LUMO energy level up, and improve electron mobility. At the same time, it can also enhance the π–π stacking between molecules. Compared with PSCs containing dithiophene-based molecules, the PSCs containing these trithiophene NF-SMAs TPTTT-2F, α-IT and MeIC1 (Fig. 5) have improved PSC performance parameters, including Voc, Jsc and FF[
The asymmetric structures of 4,4,9,9-tetrakis(4-hexylphenyl)-4,9-dihydro-s-indaceno[1,2-b]thiophene[3,2-b]thiophenealt-[5,6-d]dithieno[3,2-b:2’,3’-d]thiophene (IDT8) core give full play to extended conjugation and asymmetric core advantages. This asymmetric structure can not only expand the spectral absorption and shift the LUMO energy level up but also increase the dipole moment and fine-tunes relevant parameters. Gao et al. designed and synthesized two asymmetrical small molecule acceptors (IDT6CN-M and IDT8CN-M) (Fig. 5). The pure film for IDT8CN-M and IDT6CN-M comparable absorption coefficient of 1.35 × 105 and 1.29 × 105 cm–1 with distinguishing maximum absorption wavelength values of 699 vs. 693 nm. It can be seen that IDT8CN-M has a long conjugation length and a large maximum absorption wavelength[
Compared with thiophene-based NF-SMA, selenophene-based NF-SMA has received much less attention, and the performance of the related device is also poor. To promote the absorption of non-fullerene acceptor small molecules, reduce their bandgap, and increase their LUMO energy level, Sun's research group designed an asymmetric small molecule acceptor SePT-IN by introducing a single-sided selenium atom into the core unit of the symmetric small molecule acceptor TPT-IN (Fig. 5)[
Wang et al. and Gao et al. designed and synthesized A-NF-SMAs with the thienobenzodithiophene structure as the core TBDB-Na, TBDB-Ph, and a-BTTIC (Fig. 5)[
The electronegativity of nitrogen and sulfur atoms are 3.04 and 2.58, respectively. Compared with non-N-functionalized molecules in an asymmetric system, the introduction of nitrogen can further adjust the molecules' dipole moment. As shown in Fig. 6, the change of the sulfur atom to a nitrogen atom in the middle of the trithiophene unit determines the design of the dithieno[3,2-b:2',3'-d]pyrrole (DTP) unit. The ability to take advantage of sp2 hybrid nitrogen is the most prominent feature of the DTP unit. Besides, the lone pair of electrons on the vertical π orbital can delocalize along the π orbital of the molecule. Extending the delocalized π-electron system can narrow the optical energy bandgap (Egopt) and strengthen the π–π stacking between molecules[
![]()
Figure 6.Chemical structures of A–D–A asymmetric non-fullerene acceptors with nitrogen.
In contrast to C-type IPTT-2F, S-type IPT-2F and IPTTT-2F (Fig. 6) mixed membranes have fewer traps to avoid recombination, conducive to proper phase separation and formation of better active layer morphology. When using PBDB-T as a polymer donor, the PCEs of PSCs based on IPT-2F and IPTTT-2F were 14% and 12.3%, respectively, which were higher than those of PSCs based on C-type molecule IPTT-2F. Later, Yang et al. designed and synthesized four molecules with different aggregation characteristics IPT-4F, IPT-4Cl, IPTBO-4F, and IPTBO-4Cl (Fig. 6) by introducing different N-alkyl chains and terminal groups[
After introducing the N atom, Cao et al. combined selenophene and DTP building blocks to prepare an asymmetric heptacyclic electron-donating core DTPPSe[
Guo et al. and Luo et al. synthesized S-type seven-ring IDTP-4F and C-type eight-ring IDTTP-4F based on DTP and realized the adjustment of molecular conformation by changing the number of thiophenes on the right side (Fig. 6). IDTP-4F and IDTTP-4F both have good planar backbone conducive to intermolecular π–π packing[
Cao et al. introduced two-dimensional (2D) conjugated side chains into IPT core. They developed a 2D conjugated fused ring core semiconductor IPT2F-TCl (Fig. 6) as an electron acceptor for high-efficiency PSCs. The extended conjugate length in the ring core can optimize device performance[
In addition to changing the number of thiophene and introducing nitrogen and selenium atoms, the asymmetric design of the molecules can also be achieved by flipping the configuration for A–D–A-based non-fullerene acceptors. Luo et al. developed a novel NF-SMA ITCNTC (Fig. 7) with an asymmetric core by adjusting the configuration of thiophene[
![]()
Figure 7.Chemical structures of other asymmetric non-fullerene acceptors based on the A–D–A structure and the molecules that are associated.
Jiao et al. designed and synthesized an A–D–A typed acceptor CC10 with asymmetric donor units by introducing alkylbenzene units into CC5 (Fig. 7)[
![]()
Figure 8.(a) Chemical structure of CC5 and CC10. (b) Optimized geometries and the corresponding intermolecular binding energies by DFT calculations of CC5 and CC10 dimers. Reproduced with the permission of Ref. [
Among various core transformation engineering methods for designing asymmetric core units based on symmetrical analogs, the simplest method is to construct structural asymmetry by cutting off the side chains of the symmetric core. Many studies have been conducted on acceptors (ITOTC, ITUTC, ITUIC, IEPC, IOPC, IETC, IOTC, PhITBD, MeITBD, ITDI and CDTDI) that match to donor PBDB-T, and acceptors (PhITBD, TIDT-BT-R2, TIDT-BT-R6, ITBR, ITBRC and ITBC) match to donor PTB7-Th (Fig. 7). Zhang et al. synthesized three A–D–A type A-NF-SMAs (ITOTC, ITUTC and ITUIC)[
The molecules (TIDT-BT-R2 and TIDT-BT-R6) with the same molecular core and different terminal groups to PhITBD were reported by Bai et al.[
3.2. A-NF-SMAs with asymmetric terminal groups
Different terminal groups can induce the permanent dipole moment of A–D–A typed A-NF-SMAs molecules to regulate intermolecular interactions, leading to a more diverse aggregation tendency of the molecules affecting the π–π stacking, crystallization characteristics, and final photovoltaic performance[
ITIC is a typical NF-SMA with the advantages of easy synthesis, strong absorption in the visible light region, adjustable energy level and good stability. Adjusting terminal group substituents is a recent research hotspot in asymmetric terminal group strategy. Lai et al., Aldrich et al., Li et al., and Gao et al. used ITIC as the main body and changed the terminal substituents (chlorine, fluorine, hydrogen, and methoxy) to synthesize four different asymmetric molecules α-ITIC-2Cl, ITIC-2F (its structure is the same as a-IT-2F), ITIC-3F (its structure is the same as IT-3F), and a-IT-2OM (Fig. 9)[
![]()
Figure 9.The chemical structures of asymmetric non-fullerene acceptors are based on asymmetric terminal groups and their molecules.
Ye et al. synthesized an A-NF-SMA, named IDTT-2F-Th[
With dithienocy-clopentaindenoindene (ZIT) as the core, Zhang et al., through one-pot Knoevenagel reaction, synthesized ZITI-m, a mixture of A-NF-SMA ZITI-3F and S-NF-SMA ZITI-4F (Fig. 9)[
These great performances of PSCs based on molecules with asymmetric terminal groups reveal that it is meaningful to adjust the molecular polarity and stack morphology through asymmetric modification of the terminal groups.
3.3. A-NF-SMAs with asymmetric side chains
Side-chain engineering is crucial for device performance, which can alter crystallinity, miscibility, and intermolecular interactions. A suitable side chain can endow the acceptor molecules with good solubility so that the blended film has an appropriate nanoscale phase separation. It can also prevent small molecules from forming hydrogen aggregates and increase the charge transport rate.
Alkyl and alkaryl groups are generally used as side chains of A-NF-SMAs. Compared to acceptors with alkyl aryl groups, alkyl-substituted acceptor molecules have a shorter π–π stacking distance than alkyl aryl groups. Feng et al. synthesized two A-NF-SMAs IDT-OB and IDTT-OB (Fig. 10) through side-chain engineering[
![]()
Figure 10.Chemical structure of asymmetric non-fullerene acceptors based on asymmetric branched chains and the molecules associated with them.
Lee et al. synthesized A-D-A-type side-chain asymmetric small molecules p-IO1 and o-IO1 (Fig. 10)[
Chen et al. designed an A-NF-SMA TOBDT (Fig. 10) based on the benzo[1,2-b:4,5-b']dithiophene (BDT) fused central core with asymmetrical alkoxy and thienyl side chains[
It can be seen that the introduction of asymmetric side chains can increase the solubility of acceptor molecules, make the acceptor molecules densely packed in a dislocation manner, and form good phase separation and good device performance.
Table 1 covers the performance parameters of the acceptor–donor–acceptor (A–D–A) typed A-NF-PSCs.
4. Asymmetric non-fullerene acceptors based on A–D–A–D–A structures
A molecule with an A–D–A–D–A structure as the main body has two donor units and three acceptor units, which will cause this type of molecule to have a wider ultraviolet-visible absorption range than the A–D–A type structure. A–D–A–D–A type molecule has more donor units and acceptor units than the A–D–A type molecule, which can provide more frontier molecular orbitals to receive electrons from excited donors[
4.1. A-NF-SMAs with asymmetric cores
Imitating the design idea of A–D–A molecules, Cai et al. designed and synthesized two new Y-series non-fullerene acceptors Y21 and Y22 (Fig. 11), with asymmetric cores and used them in the study of PSCs[
![]()
Figure 11.Chemical structures of asymmetric non-fullerene acceptors based on A1–D–A2–D–A1 structure and the molecules that are associated with them.
![]()
Figure 12.(a) Molecular conformation of TB-4Cl and Y6. (b) Chemical structures of TB-4Cl and Y6. Models of (c) TB-4Cl-2T2, (d) TB-4Cl-1T1, and (e) Y6-dimer in front view and side view. Reproduced with the permission of Ref. [
4.2. A-NF-SMAs with asymmetric terminal groups
Different terminal groups are applied to the A–D–A–D–A structure molecules, and Liu et al. developed three kinds of A-NF-SMAS by replacing the fluorine atoms on the terminal groups of Y6 with chlorine atoms (Fig. 11), namely SY1 (two F atoms and one Cl atom), SY2 (two F atoms and two Cl atoms) and SY3 (three Cl atoms). Meanwhile, Y6 with four fluorine atoms substituted terminal groups and Y6-4Cl with four chlorine atoms substituted terminal groups were synthesized as control molecules[
Luo et al. developed a new asymmetric small molecule with one terminal group of BTP-4F and one terminal group of BTP-2ThCl, namely BTP-2F-ThCl (Fig. 11)[
4.3. A-NF-SMAs with asymmetric side chains
The branched alkyl chain has a significant influence on the solubility of the molecule and morphology of the mixed film, which will make the final device have different properties. Chen et al. applied asymmetric alkyl and alkoxy substitution strategies to the most advanced y-series non-fullerene acceptors and obtained a type of A-NF-SMA named Y6-1O (Fig. 11)[
The study of A–D–A–D–A typed asymmetric non-fullerene small molecules thoroughly explored the critical role of molecular conformation regulation on morphology and device efficiency, which has important guiding significance for the molecular design of NF-SMAs.
Table 2 shows the performance parameters of A–D–A–D–A typed A-NF-PSCs.
5. Conclusion and future outlook
For A–D–A typed asymmetric non-fullerene small molecules:
a) In the asymmetry of the core, molecules with DTP units show great potential. The device fabricated based on A-NF-SMA TPIC-4Cl with DTP unit obtained a PCEmax of 15.31%, which is the highest PCEmax in the A–D–A-type A-NF-PSCs so far. b) The "one-pot synthesis of mixed materials" strategy provides new ideas for molecular design, which can significantly reduce the device complexity of the traditional ternary strategy. ZITI-m is a mixture of A-NF-SMA ZITI-3F and S-NF-SMA ZITI-4F synthesized by the "one-pot Knoevenagel reaction". Compared with ZITI-3F and ZITI-4F, the PSC of the hybrid materials ZITI-m and J71 shows a very high PCEmax (13.65%). c) Side-chain asymmetric molecules are expected to further improve device performance, and research in this area should receive more attention. So far, reports on side-chain asymmetry only include 6 molecules IDT-OB, IDTT-OB, P-IO1, O-IO1, TOBDT and Y6-1O. Relative to core asymmetry and terminal group asymmetry, the number of studies related to these molecules is very small. However, the PCEmax values of these molecular-related devices are 10.12%, 11.19%, 10.80%, 13.10%, and 11.30%, which are all greater than 10%, showing great potential in the field of PSCs.
For A–D–A–D–A typed asymmetric non-fullerene small molecules:
a) Substituting thiophene for the benzene ring at the fluorinated IC terminal to form a new terminal T-IC, and using both the fluorinated IC terminal and the T-IC terminal group in the asymmetric A unit in A-NF-SMA has broad application prospects. The BTP-2F-ThCl synthesized by this move and the PSC fabricated after blending with the donor PM6 have reached the best PCEmax (17.06%) of the recent binary device. b) Fabrication of ternary PSCs also is a promising strategy further to improve the photovoltaic performance of binary PSCs. This article refers to the PCEmax of PSCs of the ternary blend (PM6:Y6:BTP-S2) was 17.43%, which improved the binary device's performance of PSCs based on PM6:BTP-S2 and PM6:Y6 (PCEmax = 16.37%, 15.79%).
In short, the asymmetric design of the molecule has the following advantages:
1) It helps increase the dipole moment and dielectric constant of the molecule and reduces the binding energy of excitons, which is very beneficial for exciton dissociation and charge transport. 2) The asymmetric structure design will also fine-tune the molecular energy level to adjust the Voc further. The influence on the absorption range and absorption intensity will cause the Jsc to change. The Voc × Jsc value can be further optimized through the asymmetric molecular design, resulting in higher device performance. 3) The effect on molecular aggregation and molecular stacking can directly change the microscopic morphology, phase separation size, and the active layer's crystallinity.
However, the synthesis of A-NF-SMAs is more complicated and costly, which is a big problem that scientists will face in the future. The box chart of PCE distribution for A-NF-SMAs is shown in Fig. 13, which records the latest developments in A-NF-SMAs based on different structures. The structural modularity of small molecules facilitates molecular tailoring and property regulation. This advantage can enable the continuous development of A-NF-SMAs in interface engineering, shape control and device structure optimization research, which will further promote the development of PSCs.
![]()
Figure 13.The box chart of PCE distribution for A-NF-SMAs based on different structures.
Acknowledgments
The authors acknowledge financial support from the National Key R&D Program of "Strategic Advanced Electronic Materials" (No.2016YFB0401100), the National Natural Science Foundation of China (Grant No.61574077), Major Program of Natural Science Foundation of the Higher Education Institutions of Jiangsu Province, China (No.19KJA460005) and Natural Science Foundation of Jiangsu Province (BK20170961).
References
[1] K N Winzenberg, P Kemppinen, F H Scholes et al. Indan-1, 3-dione electron-acceptor small molecules for solution-processable solar cells: A structure-property correlation. Chem Commun, 49, 6307(2013).
[2] T L Zhou, T Jia, B N Kang et al. Nitrile-substituted QA derivatives: New acceptor materials for solution-processable organic bulk heterojunction solar cells. Adv Energy Mater, 1, 431(2011).
[3] Y Fang, A K Pandey, A M Nardes et al. A narrow optical gap small molecule acceptor for organic solar cells. Adv Energy Mater, 3, 54(2013).
[4] C B Nielsen, E Voroshazi, S Holliday et al. Efficient truxenone-based acceptors for organic photovoltaics. J Mater Chem A, 1, 73(2013).
[5] J T Bloking, X Han, A T Higgs et al. Solution-processed organic solar cells with power conversion efficiencies of 2.5% using benzothiadiazole/imide-based acceptors. Chem Mater, 23, 5484(2011).
[6] Y Z Lin, H F Wang, Y F Li et al. A star-shaped electron acceptor based on 5, 5'-bibenzothiadiazole for solution processed solar cells. J Mater Chem A, 1, 14627(2013).
[7] Y Z Lin, P Cheng, Y F Li et al. A 3D star-shaped non-fullerene acceptor for solution-processed organic solar cells with a high open-circuit voltage of 1.18 V. Chem Commun, 48, 4773(2012).
[8] J F Li, C K Sun, A L Tang et al. Utilizing an electron-deficient thieno[3, 4-c]pyrrole-4, 6-dione (TPD) unit as a π-bridge to improve the photovoltaic performance of A–π–D–π–A type acceptors. J Mater Chem C, 8, 15981(2020).
[9] J Y Wang, X W Zhan. Fused-ring electron acceptors for photovoltaics and beyond. Acc Chem Res, 54, 132(2021).
[10] W Q Chen, Q C Zhang. Recent progress in non-fullerene small molecule acceptors in organic solar cells (OSCs). J Mater Chem C, 5, 1275(2017).
[11] H Sun, X Song, J Xie et al. PDI derivative through fine-tuning the molecular structure for fullerene-free organic solar cells. ACS Appl Mater Interfaces, 9, 29924(2017).
[12] X B Fan, J H Gao, W Wang et al. Ladder-type nonacyclic arene bis(thieno[3, 2-b]thieno)cyclopentafluorene as a promising building block for non-fullerene acceptors. Chem Asian J, 14, 1814(2019).
[13] Q S Liu, Y F Jiang, K Jin et al. 18% efficiency organic solar cells. Sci Bull, 65, 272(2020).
[14] Y Z Lin, J Y Wang, Z G Zhang et al. An electron acceptor challenging fullerenes for efficient polymer solar cells. Adv Mater, 27, 1170(2015).
[15] Y Z Lin, Y F Li, X W Zhan. A solution-processable electron acceptor based on dibenzosilole and diketopyrrolopyrrole for organic solar cells. Adv Energy Mater, 3, 724(2013).
[16] H Y Li, T Earmme, G Q Ren et al. Beyond fullerenes: Design of nonfullerene acceptors for efficient organic photovoltaics. J Am Chem Soc, 136, 14589(2014).
[17] A Sharenko, C M Proctor, T S van der Poll et al. A high-performing solution-processed small molecule: Perylene diimide bulk heterojunction solar cell. Adv Mater, 25, 4403(2013).
[18] Y Z Lin, F W Zhao, Y Wu et al. Mapping polymer donors toward high-efficiency fullerene free organic solar cells. Adv Mater, 29, 1604155(2017).
[19] X Chen, S H Zheng. On the study of influence of molecular arrangements and dipole moment on exciton binding energy in solid state. Int J Quantum Chem, 121, e26511(2021).
[20] L Benatto, K R de Almeida Sousa, M Koehler. Driving force for exciton dissociation in organic solar cells: The influence of donor and acceptor relative orientation. J Phys Chem C, 124, 13580(2020).
[21] J R Cao, S Y Qu, L Q Yang et al. An asymmetric acceptor enabling 77.51% fill factor in organic solar cells. Sci Bull, 65, 1876(2020).
[22] R J Ma, T Liu, Z H Luo et al. Adding a third component with reduced miscibility and higher LUMO level enables efficient ternary organic solar cells. ACS Energy Lett, 5, 2711(2020).
[23] L X Yin, Q Q Yuan, Y Q Li. D–A–A'-type asymmetric small molecules based on triphenylamine-diketopyrrolopyrrole/5, 6-difluoro-2, 1, 3-benzothiadiazole backbone for organic photovoltaic materials. New J Chem, 44, 13319(2020).
[24] D N Congreve, J Lee, N J Thompson et al. External quantum efficiency above 100% in a singlet-exciton-fission-based organic photovoltaic cell. Science, 340, 334(2013).
[25] P E Schwenn, K Gui, A M Nardes et al. A small molecule non-fullerene electron acceptor for organic solar cells. Adv Energy Mater, 1, 73(2011).
[26] Y Shu, Y F Lim, Z Li et al. A survey of electron-deficient pentacenes as acceptors in polymer bulk heterojunction solar cells. Chem Sci, 2, 363(2011).
[27] Y Zhou, L Ding, K Shi et al. A non-fullerene small molecule as efficient electron acceptor in organic bulk heterojunction solar cells. Adv Mater, 24, 957(2012).
[28] C Li, H T Fu, T Xia et al. Asymmetric nonfullerene small molecule acceptors for organic solar cells. Adv Energy Mater, 9, 1900999(2019).
[29] C Q Tang, S C Chen, Q Shang et al. Asymmetric indenothiophene-based non-fullerene acceptors for efficient polymer solar cells. Sci China Mater, 60, 707(2017).
[30] J Yuan, Y Q Zhang, L Y Zhou et al. Single-junction organic solar cell with over 15% efficiency using fused-ring acceptor with electron-deficient core. Joule, 3, 1140(2019).
[31] X Guo, Q P Fan, J N Wu et al. Optimized active layer morphologies via ternary copolymerization of polymer donors for 17.6 % efficiency organic solar cells with enhanced fill factor. Angew Chem Int Ed, 60, 2322(2021).
[32] S X Li, L L Zhan, Y Z Jin et al. Asymmetric electron acceptors for high-efficiency and low-energy-loss organic photovoltaics. Adv Mater, 32, 2001160(2020).
[33] C X Zhang, R G Liu, C H Mak et al. Photophysics of organic photovoltaic devices: A review. J Photonics Energy, 8, 021001(2018).
[34] R Ilmi, A Haque, M S Khan. High efficiency small molecule-based donor materials for organic solar cells. Org Electron, 58, 53(2018).
[35] B Qi, J Wang. Fill factor in organic solar cells. Phys Chem Chem Phys, 15, 8972(2013).
[36] Y Z Chen, F J Bai, Z X Peng et al. Asymmetric alkoxy and alkyl substitution on nonfullerene acceptors enabling high-performance organic solar cells. Adv Energy Mater, 11, 2003141(2021).
[37] C Li, Y P Xie, B B Fan et al. A nonfullerene acceptor utilizing a novel asymmetric multifused-ring core unit for highly efficient organic solar cells. J Mater Chem C, 6, 4873(2018).
[38] J L Song, C Li, L L Ye et al. Extension of indacenodithiophene backbone conjugation enables efficient asymmetric A–D–A type non-fullerene acceptors. J Mater Chem A, 6, 18847(2018).
[39] S N Liu, B F Zhao, Z Y Cong et al. Influences of the terminal groups on the performances of asymmetric small molecule acceptors-based polymer solar cells. Dyes Pigments, 178, 108388(2020).
[40] W Gao, T Liu, C Zhong et al. Asymmetrical small molecule acceptor enabling nonfullerene polymer solar cell with fill factor approaching 79%. ACS Energy Lett, 3, 1760(2018).
[41] X S Li, C Li, L L Ye et al. Asymmetric A–D–π–A-type nonfullerene small molecule acceptors for efficient organic solar cells. J Mater Chem A, 7, 19348(2019).
[42] W Gao, M Zhang, T Liu et al. Asymmetrical ladder-type donor-induced polar small molecule acceptor to promote fill factors approaching 77% for high-performance nonfullerene polymer solar cells. Adv Mater, 30, 1800052(2018).
[43] W Gao, F Wu, T Liu et al. Multifunctional asymmetrical molecules for high-performance perovskite and organic solar cells. J Mater Chem A, 7, 2412(2019).
[44] W Gao, T Liu, R Sun et al. Dithieno[3, 2-b: 2ʹ, 3ʹ-d]pyrrol-fused asymmetrical electron acceptors: A study into the effects of nitrogen-functionalization on reducing nonradiative recombination loss and dipole moment on morphology. Adv Sci, 7, 1902657(2020).
[45] W Gao, Q S An, C Zhong et al. Designing an asymmetrical isomer to promote the LUMO energy level and molecular packing of a non-fullerene acceptor for polymer solar cells with 12.6% efficiency. Chem Sci, 9, 8142(2018).
[46] C Li, J L Song, L L Ye et al. High-performance eight-membered indacenodithiophene-based asymmetric A-D-A type non-fullerene acceptors. Sol RRL, 3, 1800246(2019).
[47] C Li, J L Song, Y H Cai et al. Heteroatom substitution-induced asymmetric A-D-A type non-fullerene acceptor for efficient organic solar cells. J Energy Chem, 40, 144(2020).
[48] C Li, T Xia, J L Song et al. Asymmetric selenophene-based non-fullerene acceptors for high-performance organic solar cells. J Mater Chem A, 7, 1435(2019).
[49] X C Wang, J H Han, H X Jiang et al. Regulation of molecular packing and blend morphology by finely tuning molecular conformation for high-performance nonfullerene polymer solar cells. ACS Appl Mater Interfaces, 11, 44501(2019).
[50] W Gao, T Liu, J W Li et al. Simultaneously increasing open-circuit voltage and short-circuit current to minimize the energy loss in organic solar cells via designing asymmetrical non-fullerene acceptor. J Mater Chem A, 7, 11053(2019).
[51] X M Zhang, M M Li, Q Wang et al. Near-infrared absorbing non-fullerene acceptors with dithienopyrrole as π spacer for organic solar cells. Chin J Appl Chem, 36, 1023(2019).
[52] Y F Geng, A L Tang, K Tajima et al. Conjugated materials containing dithieno[3, 2-b: 2', 3'-d]pyrrole and its derivatives for organic and hybrid solar cell applications. J Mater Chem A, 7, 64(2019).
[53] L Q Yang, X Song, J S Yu et al. Tuning of the conformation of asymmetric nonfullerene acceptors for efficient organic solar cells. J Mater Chem A, 7, 22279(2019).
[54] L Q Yang, Z H Hu, Z H Zhang et al. Molecular engineering of acceptors to control aggregation for optimized nonfullerene solar cells. J Mater Chem A, 8, 5458(2020).
[55] R J Ma, G Li, D D Li et al. Understanding the effect of end group halogenation in tuning miscibility and morphology of high-performance small molecular acceptors. Sol RRL, 4, 2000250(2020).
[56] G Li, D D Li, R J Ma et al. Efficient modulation of end groups for the asymmetric small molecule acceptors enabling organic solar cells with over 15% efficiency. J Mater Chem A, 8, 5927(2020).
[57] J R Cao, S Y Qu, J S Yu et al. 13.76% efficiency nonfullerene solar cells enabled by selenophene integrated dithieno[3, 2-b:2', 3'-d]pyrrole asymmetric acceptors. Mater Chem Front, 4, 924(2020).
[58] Q Guo, R J Ma, J Hu et al. Over 15% efficiency polymer solar cells enabled by conformation tuning of newly designed asymmetric small-molecule acceptors. Adv Funct Mater, 30, 2000383(2020).
[59] Z H Luo, R J Ma, Y Q Xiao et al. Conformation-tuning effect of asymmetric small molecule acceptors on molecular packing, interaction, and photovoltaic performance. Small, 16, 2001942(2020).
[60] Z H Zhang, L Q Yang, Z H Hu et al. Charge density modulation on asymmetric fused-ring acceptors for high-efficiency photovoltaic solar cells. Mater Chem Front, 4, 1747(2020).
[61] Z H Luo, G H Li, K L Wu et al. Asymmetric thieno[2, 3-b]thiophene-based electron acceptor featuring a seven fused-ring electron donor unit as core for nonfullerene organic photovoltaics. Org Electron, 62, 82(2018).
[62] C C Jiao, Z Q Guo, B Q Sun et al. An acceptor–donor–acceptor type non-fullerene acceptor with an asymmetric backbone for high performance organic solar cells. J Mater Chem C, 8, 6293(2020).
[63] W Hu, X Y Du, W L Zhuang et al. Axisymmetric and asymmetric naphthalene-bisthienothiophene based nonfullerene acceptors: On constitutional isomerization and photovoltaic performance. ACS Appl Energy Mater, 3, 5734(2020).
[64] M Q Zhang, Y L Ma, Q D Zheng. Asymmetric indenothienothiophene-based unfused core for A-D-A type nonfullerene acceptors. Dyes Pigments, 180, 108495(2020).
[65] B Huang, L Chen, X F Jin et al. Alkylsilyl functionalized copolymer donor for annealing-free high performance solar cells with over 11% efficiency: Crystallinity induced small driving force. Adv Funct Mater, 28, 1800606(2018).
[66] Z J Kang, Y L Ma, Q D Zheng. Asymmetric indenothiophene-based nonfullerene acceptors for binary- and ternary-blend polymer solar cells. Dyes Pigments, 170, 107555(2019).
[67] L Hong, H F Yao, R N Yu et al. Investigating the trade-off between device performance and energy loss in nonfullerene organic solar cells. ACS Appl Mater Interfaces, 11, 29124(2019).
[68] W Y Bai, X P Xu, Q Y Li et al. Efficient nonfullerene polymer solar cells enabled by small-molecular acceptors with a decreased fused-ring core. Small Methods, 2, 1700373(2018).
[69] Y Un Kim, G Eun Park, S Choi et al. A new n-type semiconducting molecule with an asymmetric indenothiophene core for a high-performing non-fullerene type organic solar cell. J Mater Chem C, 5, 7182(2017).
[70] Q Y Li, J Y Xiao, L M Tang et al. Thermally stable high performance non-fullerene polymer solar cells with low energy loss by using ladder-type small molecule acceptors. Org Electron, 44, 217(2017).
[71] J Y Xiao, Z M Chen, G C Zhang et al. Efficient device engineering for inverted non-fullerene organic solar cells with low energy loss. J Mater Chem C, 6, 4457(2018).
[72] Z J Kang, S C Chen, Y L Ma et al. Push-pull type non-fullerene acceptors for polymer solar cells: Effect of the donor core. ACS Appl Mater Interfaces, 9, 24771(2017).
[73] Y Zhang, Y Wang, Z Y Xie et al. Preparation of non-fullerene acceptors with a multi-asymmetric configuration in a one-pot reaction for organic solar cells. J Mater Chem C, 8, 17229(2020).
[74] D Q Hu, Q G Yang, Y J Zheng et al. 15.3% efficiency all-small-molecule organic solar cells achieved by a locally asymmetric F, Cl disubstitution strategy. Adv Sci, 8, 2004262(2021).
[75] F Pan, X J Li, S Bai et al. High electron mobility fluorinated indacenodithiophene small molecule acceptors for organic solar cells. Chin Chem Lett, 32, 1257(2021).
[76] H J Lai, H Chen, J D Zhou et al. 3D interpenetrating network for high-performance nonfullerene acceptors via asymmetric chlorine substitution. J Phys Chem Lett, 10, 4737(2019).
[77] T J Aldrich, M Matta, W Zhu et al. Fluorination effects on indacenodithienothiophene acceptor packing and electronic structure, end-group redistribution, and solar cell photovoltaic response. J Am Chem Soc, 141, 3274(2019).
[78] M Li, Y Y Zhou, J Q Zhang et al. Tuning the dipole moments of nonfullerene acceptors with an asymmetric terminal strategy for highly efficient organic solar cells. J Mater Chem A, 7, 8889(2019).
[79] B W Gao, H F Yao, J X Hou et al. Multi-component non-fullerene acceptors with tunable bandgap structures for efficient organic solar cells. J Mater Chem A, 6, 23644(2018).
[80] L L Ye, Y P Xie, Y Q Xiao et al. Asymmetric fused-ring electron acceptor with two distinct terminal groups for efficient organic solar cells. J Mater Chem A, 7, 8055(2019).
[81] H J Lai, H Chen, Y Shen et al. Using chlorine atoms to fine-tune the intermolecular packing and energy levels of efficient nonfullerene acceptors. ACS Appl Energy Mater, 2, 7663(2019).
[82] J Y Zhang, W R Liu, S S Chen et al. One-pot synthesis of electron-acceptor composite enables efficient fullerene-free ternary organic solar cells. J Mater Chem A, 6, 22519(2018).
[83] T N Duan, L C Hou, J H Fu et al. An asymmetric end-capping strategy enables a new non-fullerene acceptor for organic solar cells with efficiency over 10%. Chem Commun, 56, 6531(2020).
[84] Y Zhao, Z H Luo, G H Li et al. De novo design of small molecule acceptors via fullerene/non-fullerene hybrids for polymer solar cells. Chem Commun, 54, 9801(2018).
[85] S Y Feng, C E Zhang, Y H Liu et al. Fused-ring acceptors with asymmetric side chains for high-performance thick-film organic solar cells. Adv Mater, 29, 1703527(2017).
[86] S Y Feng, C Zhang, Z Z Bi et al. Controlling molecular packing and orientation via constructing a ladder-type electron acceptor with asymmetric substituents for thick-film nonfullerene solar cells. ACS Appl Mater Interfaces, 11, 3098(2019).
[87] J Lee, S Song, J F Huang et al. Bandgap tailored nonfullerene acceptors for low-energy-loss near-infrared organic photovoltaics. ACS Mater Lett, 2, 395(2020).
[88] X B Chen, B Kan, Y Y Kan et al. As-cast ternary organic solar cells based on an asymmetric side-chains featured acceptor with reduced voltage loss and 14.0% efficiency. Adv Funct Mater, 30, 1909535(2020).
[89] X Z Liu, Y N Wei, X Zhang et al. An A-D-A'-D-A type unfused nonfullerene acceptor for organic solar cells with approaching 14% efficiency. Sci China Chem, 64, 228(2021).
[90] T Liu, Y D Zhang, Y M Shao et al. Asymmetric acceptors with fluorine and chlorine substitution for organic solar cells toward 16.83% efficiency. Adv Funct Mater, 30, 2000456(2020).
[91] Z H Luo, R J Ma, T Liu et al. Fine-tuning energy levels via asymmetric end groups enables polymer solar cells with efficiencies over 17%. Joule, 4, 1236(2020).
[92] J S Zhang, Y F Han, W X Zhang et al. High-efficiency thermal-annealing-free organic solar cells based on an asymmetric acceptor with improved thermal and air stability. ACS Appl Mater Interfaces, 12, 57271(2020).
[93] F F Cai, C Zhu, J Yuan et al. Efficient organic solar cells based on a new "Y-series" non-fullerene acceptor with an asymmetric electron-deficient-core. Chem Commun, 56, 4340(2020).
[94] F F Cai, H J Peng, H G Chen et al. An asymmetric small molecule acceptor for organic solar cells with a short circuit current density over 24 mA cm–2. J Mater Chem A, 8, 15984(2020).
[95] M Zhang, W Gao, F J Zhang et al. Efficient ternary non-fullerene polymer solar cells with PCE of 11.92% and FF of 76. 5%. Energy Environ Sci, 11, 841(2018).
[96] C H Jeong, Y U Kim, C G Park et al. Improved performance of non-fullerene polymer solar cells by simple structural change of asymmetric acceptor based on indenothiophene. Synth Met, 246, 164(2018).
Set citation alerts for the article
Please enter your email address



