Jing Long, Zhao Ye, Yong Du, Xu-ming Zheng, Jia-dan Xue. Direct Observation of Transient Species Generated from Protonation and Deprotonation of the Lowest Triplet of p-Nitrophenylphenol†[J]. Chinese Journal of Chemical Physics, 2020, 33(5): 635
Search by keywords or author
- Chinese Journal of Chemical Physics
- Vol. 33, Issue 5, 635 (2020)
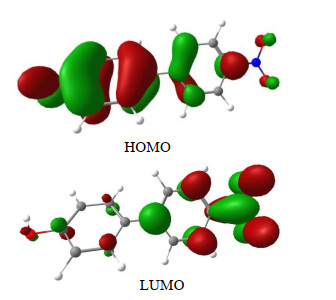
Fig. 1. The transient absorption spectra obtained immediately after the laser excitation of HO-Bp-NO2 in acetonitrile (MeCN) solution without acid (orange) and with 0.38 mol/mL HClO\begin{document}$ _4 $\end{document} \begin{document}$ ^3 $\end{document} \begin{document}$ ^+ $\end{document}

Fig. 1. The molecular structure diagrams corresponding to the abbreviations mentioned in text.
Fig. 1. Plot of HOMO and LUMO of HO-Bp-NO2 corresponding to S0\begin{document}$ \to $\end{document}
Fig. 2. Transient absorption spectrum obtained immediately after 355 nm laser pulse photolysis of HO-Bp-NO2 in cyclohexane (CHX) compared to that in acetonitrile (MeCN).
Fig. 2. [in Chinese]
Fig. 2. Nanosecond transient absorption spectra upon laser excitation of HO-Bp-NO2 in acetonitrile in the presence of 0.1 mmol/L acid. Insert: (black) kinetics monitored at 660 and 450 nm, and (red) exponential curves fitted with a single decay (660 nm) and a combined (growth and decay) functions respectively.
Fig. 3. Nanosecond transient absorption spectra obtained immediately after laser excitation of Bp-NO2 in acetonitrile solution containing various concentrations of HClO\begin{document}$ _4 $\end{document}
Fig. 3. [in Chinese]
Fig. 3. Molecular structure of (top) close-shell and (middle) open-shell singlet of O-Bp-NO2H. (bottom) The spin density contribution for O-Bp-NO2H, plotted with Multiwfn software1.
Fig. 4. Nanosecond transient absorption spectra obtained immediately after laser excitation of HO-Bp-NO2 in acetonitrile solution containing various concentrations of acid. Symbol star indicates the residual of excitation laser pulse.
Fig. 4. Nanosecond transient absorption spectra upon laser excitation of HO-Bp-NO2 in acetonitrile in the presence of various concentration of acid.
Fig. 5. (a) Nanosecond transient absorption spectra recorded at various time delays after laser pulse excitation of HO-Bp-NO2 (0.05 mmol/L) in neat acetonitrile. (b) Difference spectra obtained by subtraction of scaled 0 ns spectra from those recorded at post time delays which have been labeled in the graph, with the criterion of completely removing the absorption of \begin{document}$ ^3 $\end{document} \begin{document}$ {\rm{ \mathsf{ μ} }} $\end{document}
Fig. 5. Kinetics (black) monitored at 450 nm obtained upon excitation of HO-Bp-NO2 in acetonitrile solution containing various concentration of acid, and simulated curves (red) fitted with exponential functions.
Fig. 6. pKa*=3.1 for 3Bp-NO2H+ obtained by using the dual-wavelength spectrophotometry measured at characteristic absorption bands at 625 (neutral T1) and 740 nm (triplet cation).
Fig. 7. Kinetics (black) monitored at 650, 450 and 380 nm obtained in neat acetonitrile solution with different concentrations of HO-Bp-NO2, and simulated curves (red) fitted with exponential functions.
Fig. 8. (a) UV-visible absorption spectra of HO-Bp-NO2 in solutions containing MeCN:buffered water (9:1/v:v), and (b) pK a =10.7 for HO-Bp-NO2 obtained by using the dual-wavelength spectrophotometry measured at characteristic absorptions 328 and 426 nm.
|
Table 1. Pseudo first order decay or/and growth time constants fitted with exponential functions at wavelengths of 660 and 450 nm obtained upon excitation of HO-Bp-NO2 in acetonitrile containing various concentration of acid.
|
Table 2. Pseudo first order decay or/and growth time constants fitted with exponential functions at wavelengths of 650, 450 and 380 nm obtained in neat acetonitrile with different concentration of HO-Bp-NO2 in mol/L.
|
Table 3. Fitted parameters in the rate constant equation, $ r_{ \rm{T}_1} $ $ ^{\rm{a}} $ $ k $ $ \times $ $ b $
|
Table 4. Vertical transitions predicted by TD-DFT calculations on the open-shell singlet of O-Bp-NO2H
Set citation alerts for the article
Please enter your email address







