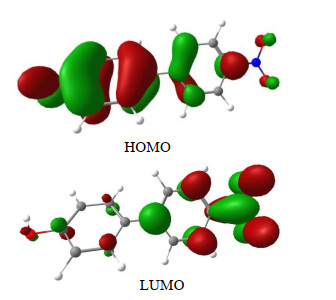
Jing Long, Zhao Ye, Yong Du, Xu-ming Zheng, Jia-dan Xue. Direct Observation of Transient Species Generated from Protonation and Deprotonation of the Lowest Triplet of p-Nitrophenylphenol†[J]. Chinese Journal of Chemical Physics, 2020, 33(5): 635
Search by keywords or author
- Chinese Journal of Chemical Physics
- Vol. 33, Issue 5, 635 (2020)
Abstract
Set citation alerts for the article
Please enter your email address