- Journal of Semiconductors
- Vol. 41, Issue 9, 091708 (2020)
Abstract
1. Introduction
The sharply increasing threat posed by energy shortage and environmental pollution exerts a substantial impact on modern economic development and human living environment[
Traditionally, inorganic semiconductors have been widely employed in the field of photocatalysis, e.g. TiO2[
Recently, emerging organic semiconductors have aroused substantial attention owing to flexible structural modification, changeable electronic energy bands, tunable morphologies, superior light absorption, excellent chemical stability and versatile functions[
Textile dyes have a long history of ca. 150 years as the first products in most of the largest chemical companies founded at that time[
Herein, we present the overview from the electronic structural modulation to photocatalytic applications of PDI self-assembly-based photocatalysts. Firstly, the basic characteristics of PDI molecules and PDI self-assembly are introduced. Next, the possible electronic modulation approaches are discussed, including modifying perylene areas, tuning Π–Π stacking via side-chain substituents, constructing PDI self-assembly-based composites and the role of PDI anion/dianion radicals. Subsequently, some practical applications are exemplified to highlight the significance of the organic self-assembled materials, including photocatalytic degradation of pollutants, water splitting into H2/O2, organic synthesis and disease therapy, with high efficiency of solar-light utilization stemming from particular Π–Π stacking structures. Finally, the outlooks and perspectives on further development of PDI self-assembly-based photocatalysts are envisioned.
2. Basic characteristics of PDI molecules
Individual PDI molecule derivatives consist of two primary motifs-PDI skeleton and side chains. The PDI skeleton can be considered as being composed of a nonpolar perylene ring and two polar cyclic amides. Π-conjugated perylene fragment endows PDI molecules a strong self-assembled propensity and electron delocalization possibility along the Π–Π stacking direction via intermolecular orbital overlapping. Viewing from the bond lengths of PDI molecules (Figs. 1(a) and 1(b)), the two Csp2–Csp2 bonds that connect two naphthalene half units appear to be longer than the other Csp2–Csp2 bonds and would undoubtedly generate steric strain in the coplanar perylene core. Consequently, two naphthalene half units in the perylene ring are twisted with a torsional angle[
![]()
Figure 1.(Color online) Bond lengths of PDI molecules (a) without side chains and (b) with side chains obtained via DFT calculations. (c) Frontier orbital energy levels of PDI molecules (1), (2), (3) and (4). (Method and basic set: B3LYP 6-31+G*).
3. Aggregation characteristics in thermodynamics
The packing characters have been deeply discussed in the reviews of Würthner’s group[
![]()
Figure 2.(Color online) (a) Model diagram of the PDI Π–Π stacking-assembled structure with permission from Ref. [
4. Electronic structure of PDI self-assembly
4.1. Identification of band-like electronic structure of PDI self-assembly
The PDI molecule belongs to a type of dye whose energy level gap measured by UV–vis absorption spectra corresponds to ca. 2.5 eV, similar with the theoretical value calculated through DFT calculations. The light adsorption of PDI originates from perylene chromophore, and the polarity of cyclic amide leads to the red-shift of absorption peak. Hence, its frontier orbital energy levels consist of the orbital correlation of C and O atoms in the two units. Generally, there exists three different electronic transition modes for monomeric PDI molecules, and thus three corresponding characteristic absorption (400–550 nm) and emission peaks shown up in UV–vis absorption and fluorescence spectra, respectively. According to DFT calculations, the energy levels of the frontier orbits of parent PDI molecules are positioned at –6.11 eV (LUMO) and –3.57 eV (HOMO). The PDI molecule structure does not deliver continuous electronic energy band structure, since only the crystals form band-like electronic structures. The band gap of PDI self-assembly is dependent upon the packing degree and length in PDI self-assembly with PDI molecules as building blocks[
![]()
Figure 3.(Color online) (a) Mott–Schottky curves and (b) XPS valence-band spectrum of the self-assembled PDI supramolecular system. (c) Schematic illustration of the electronic energy level structure of PDI self-assembly with permission from Ref. [
4.2. Approaches to tune the energy bands of PDI self-assembly
With an aim of purposeful utilization of the sunlight and improvement of photocatalytic redox reactions, regulating electronic energy bands of semiconductors is critical and effective. For example, narrowing energy band gap can extend light absorption range to the entire visible-light wavelength so as to efficiently utilize the solar energy as far as possible; adjusting the CB and VB position can make it possible to reach the potentials required for redox reactions and initiate overall reaction kinetics. In theory, for photocatalytic water splitting, when the potentials of CBs approach 0.5 to –1.5 V vs. NHE, the semiconductors can photocatalyze water into H2; and if the VB potentials reach 1.0–3.5 V vs. NHE, the semiconductors perform a strong capability to realize water oxidation[
4.2.1. Electronic modification of isolated PDI molecules
Modification to bay areas of PDI molecules with electron-defective or electron-rich substituents is an effective approach to adjust electronic structures of isolated PDI molecules and follow-up PDI self-assembly. The approach is firstly mastered by Seybold et al., via incorporating four phenoxy groups to substituted chlorine groups at bay positions[
The modification to bay areas of PDI molecules cannot only modify electronic energy levels of PDI molecules, but also transform Π–Π stacking arrangements in PDI self-assembly. Both of these would lead to the change in electronic structures of PDI self-assembly. When electron-withdrawing or electron-donating groups are introduced into perylene bays of PDI molecules, the substituents electronically interact with PDI molecular orbital, causing the charge density redistribution in PDI molecules, which influences LUMO and HOMO levels of PDI molecules. In electrochemical measurements, PDI molecules undergo a transformation in three types of existent states (PDI, PDI– and PDI2–), and hence the reduction potentials of PDI molecules can be observed (Fig. 4(a)). Seifert et al. found out via electrochemical measurements that two reversible reduction peaks of highly electron-deficient PDI shifted to a more positive potential, and the reduction potentials of the PDI derivatives with substituents at the bays were compared as following: Cl4PDI > Br 4Cl4PDI > (CN) 4Cl4PDI[
![]()
Figure 4.(Color online) (a) Schematic representation of the equilibrium between reduced form (PDI2–), intermediate (PDI–) and fully oxidized form (PDI). (b) Cyclic voltammograms of Br4Cl4PDI (PDI 2) and (CN)4Cl4PDI (PDI 3) in dichloromethane with permission from Ref. [
In addition, increasing the degree of Π-conjugation of aromatic rings can also effectively tune electronic structures of PDI anlogues. The perylene ring of PDI molecules was expanded by Lee et al. to obtain terrylenetetracarboxylic diimide (TDI), and quaterrylenecarboxylic diimide (QDI) counterparts[
4.2.2. Tuning PDI self-assembled array via side-chain substituents
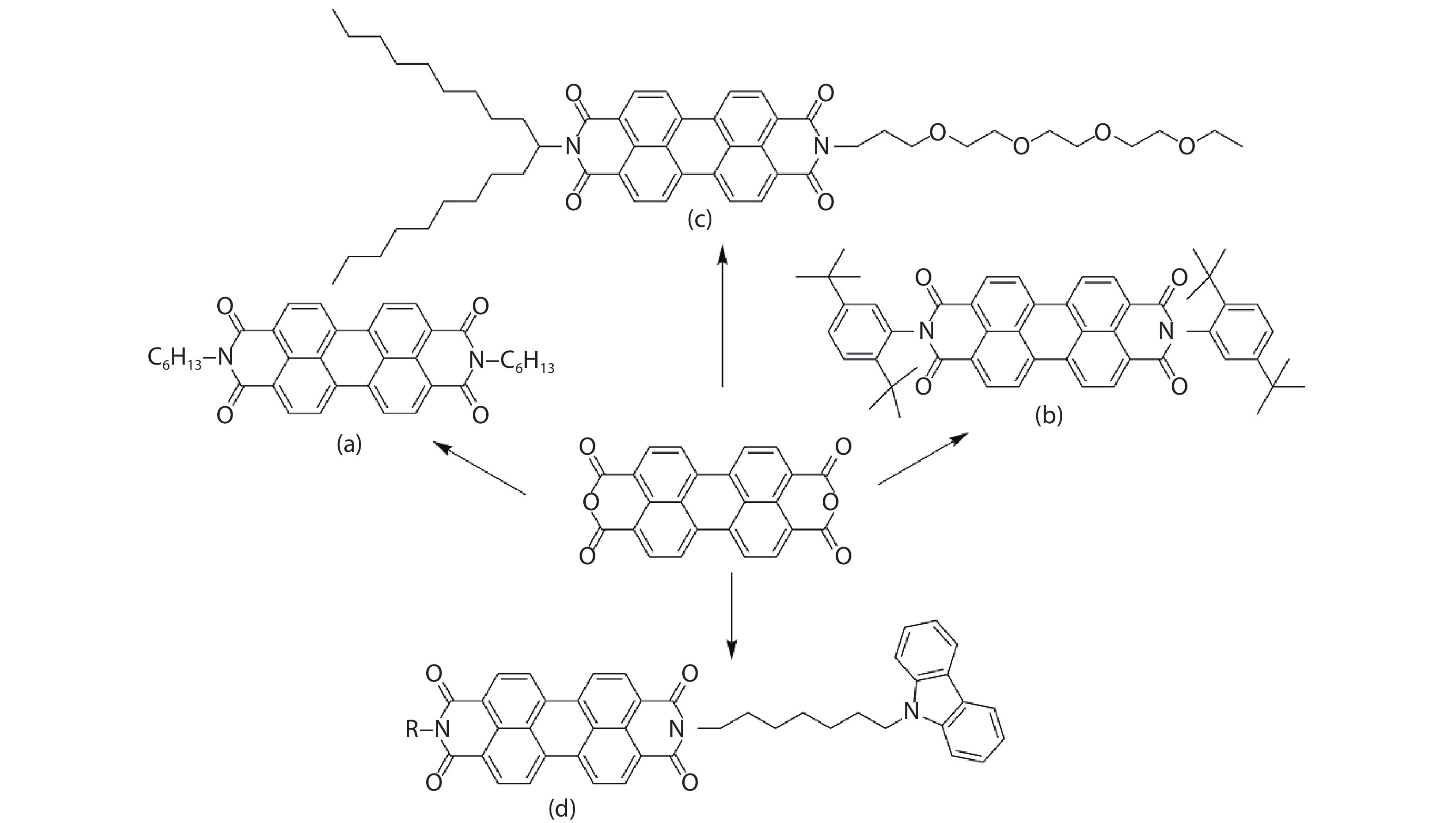
Suitable substituents introduced at imide nitrogen of PDI molecules are necessary for controlling the self-assembled process to obtain desirable well-defined supramolecular structures. It is not only because substituents provide H-bonds/side-to-side chain interactions for lateral growth of PDI self-assembly, but also sterically hindered counterparts would weaken Π–Π stacking interaction between perylene rings, permitting a pathway-controlled self-assembly. For example, PDI-[GY]2 (GY = glycine-tyrosine) forms chiral nanofibers while PDI-[GD]2 (GD = glycine-aspartic acid) aggregates into spherical morphology, which depends on the nature of substituents at side chains[
![]()
Figure 5.(Color online) (a) Diagrams showing the approaches relative to binary solvent mixing and metal-ligand-coordination-directed method. TEM images of (b) bulk PDI and (c) nano PDI with permission from Ref. [
Current methods for preparing PDI self-assemblies are various, generally including substrate-supported in situ self-assembly, solvent-phase interfacial self-assembly, vapor-triggered self-assembly at the interface of solid/gas or liquid/gas systems, pH triggered self-assembly in aqueous solution, and chemical reaction-mediated self-assembly of unsubstituted PTCDA and perylene diimide, etc.[
![]()
Figure 6.(Color online) (a) Fluorescence decay transients measured at 470 nm for 2%, 8%, and 80% hybrids with permission from Ref. [
Alternatively, another possible approach is metal-ligand-coordination (Fig. 5(a)). Zeng et al. fabricated the single crystalline metal-organic polymer based on PDI self-assembly using this method[
Though side chains at the nodes of PDI molecules do not obviously alter electronic structures of PDI molecules, it can modulate Π–Π stacking modes of PDI self-assembly, thereby acting on its electronic structure, which has been verified via DFT calculations using a series of PDI self-assemblies with different side-chain substituents[
The PDI molecules can act as electron acceptors that can exchange electrons with some other molecules/materials with electron-donating capability. This interaction can effectively regulate the conduction of photo-generated charges in PDI self-assembly[
4.2.3. Combining with other materials
The construction of PDI self-assembly-based composites can be employed for the modification of electronic structure of PDI self-assembly. The Π–Π stacking interaction between PDI molecules is usually much stronger than the interaction between PDI molecules and supporting materials, unless the template is modified with appropriate polar groups so as to build a strong coupling effect with functional groups of PDI molecules. Adsorption of a few PDI molecules on templates favors connection of more PDI molecules and follow-up formation of PDI self-assembly on the surface. Via pH-induced aggregation, PDI self-assembly can be loaded on the surface of inorganic semiconductor nanoparticles, forming the core-shell structure[
Π–Π stacking permutations of PDI self-assembly are closely pertinent to external environments, mainly because the PDI self-assembly is a dynamic balance process between aggregation and disaggregation[
![]()
Figure 7.(Color online) Proposed mechanism of electron transmission in PDI self-assembly, wherein the HOMO and LUMO levels are obtained via DFT calculations.
4.2.4. Electronic modulation role of PDI anion/dianion radicals
Since the PDI molecules act as an electron acceptor[
4.3. Electron/energy transfer in PDI self-assembly
In the process of electron transfer, the PDI molecules have very small reorganization energy (0.15 eV), which promotes the electron/energy transfer between PDI molecules/motifs and other chemical compounds/motifs under the built-in electric field. A series of multifunctional integrated systems with extended tetracationic cyclophane/PDI systems as different constituent units were successfully designed by Scheman’s research group[
The charge-transfer event in photo-excited PDI self-assembly is roughly as following: firstly, under the visible-light irradiation, PDI self-assembly produces photogenerated electrons and holes, and the minimum excitation energy of photogenerated charges depends on the energy band gap of PDI self-assembly; secondly, due to the Π–Π stacking interaction between PDI molecules, photo-generated charges can be rapidly separated along the Π–Π stacking direction in PDI self-assembly. The fluorescence lifetime of these photogenerated charges in organic semiconductors is around 1–100 ns[
Inspired by the Marcus principle, the charge transfer process is actually analogue to a redox process, the redox mechanism between PDI molecules has been adopted to explain the charge transfer mechanism inside PDI self-assembly (Fig. 7)[
![]()
Figure 8.(Color online) (a) Photodegradation curves for phenol (5 ppm) over H-PDI and J-PDI under visible light with permission from Ref. [
Electron transfer along Π–Π stacking direction can smoothly proceed in a short range. Long-range electron conduction in the bulk may result in relative fast electron–hole recombination. The main reasons may be: a) the transfer of photogenerated electrons in PDI self-assembly is bidirectional, and thus electron conduction to a certain distance would inevitably terminate or shift in reverse direction; b) when PDI molecules are stacked to a certain extent, the subsequent stacking of PDI molecules cannot affect the electronic structure of PDI self-assembly.
In order to prevent the overly long Π–Π stacking dimension of PDI self-assembly, we could adopt some trade-off strategies on the basis of the crystal growth modes: (1) The common method is to change the side chains of PDI molecules, combined with the adjustment of the external environment, i.e., the side chains containing acidic/alkaline groups can be introduced for different acid-base equilibrium constants, followed by tuning the pH value of the solution to change its aggregation degree; as for the side chains with different lipophilicity/hydrophilicity, we can change the polarity of the mixed solvent to control the Π–Π stacking length. (2) As described above, building Π–Π stacking indentation inside PDI self-assembly is also an alternative method; (3) Appropriate templates/additives can be considered to control the growth rate or scale of PDI crystal nuclei.
5. Photocatalytic examples based on PDI self-assembly
5.1. Environmental remedy
5.1.1. Pure PDI self-assembly
PDI self-assembly is an efficient visible-light photocatalyst because it possesses appropriate electronic energy band structure, highly ordered Π–Π stacking structure, and strong adsorption interaction with organic compounds. The electron "clouds" of Π-orbits of PDI molecules overlap with each other, and this structure significantly narrows the energy band gap of PDI self-assembly, so that its light absorption range extends to the entire visible-light region. Due to the existence of molecular polar electric field, PDI molecules exhibit a high extinction coefficient, giving it a strong visible-light absorption ability. The Π–Π stacking structure of PDI self-assembly enables the rapid transfer of photo-generated carriers to realize spatial separation of photo-generated electrons and holes. The surface of nano-shaped PDI self-assembly has a strong adsorption effect on some negative ions in aqueous solution, hence the PDI self-assembly surface would be negatively charged and covered with hydration film[
![]()
Figure 9.(Color online) (a) The photocatalytic process with charge transfer and accumulation on the surface of PDI self-assembly. (b) H2 production histogram of CA gels prepared with NaCl, PDDA, CaCl2 and ascorbic acid compared to insoluble protonated CA with permission from Ref. [
5.1.2. Composites
However, photogenerated charges in PDI self-assembly would also recombine and annihilate, severely corroding its photocatalytic activity. To boost visible-light photocatalytic activity of PDI self-assembly, some feasible approaches need to be developed. Composite photocatalysts based on PDI self-assembly are therefore developed to reduce the recombination of photogenerated charges. Wei et al. prepared the PDI/P25 core-shell structure, which greatly improved the photocatalytic activity of PDI self-assembly[
Besides of combination with TiO2, composite construction of PDI self-assembly and other suitable materials (e.g. inorganic semiconductors, carbon materials, metal promoters) is also conducive to the improvement of photocatalytic performance. Yang et al. constructed p-Ag2S/n-PDI composite materials to obtain a superior photocatalyst with full-spectrum utilization[
5.1.3. Photoelectrocatalysis
Photoelectrocatalysis is one of the most promising fields for our developing society to study how to effectively utilize the solar energy. Like traditional catalysts, the role of photocatalysts is to reduce the activation energy of the reaction and change the reaction pathway. Photoelectrocatalytic technology, as a new technology combined with photocatalysis and electrochemistry, has drawn increasing attention, along with the characteristics of photocatalysis and electrocatalysis[
The effect of applied electric field further promotes the effective separation and transfer of photogenerated charges in photocatalysts. Since photogenerated electrons and holes are accompanied by equal amount, when they are in direct contact, simple recombination occurs consequently. This recombination leads to the phenomenon of short circuit galvanic cells on the surface of photocatalysts, which greatly reduces the efficiency of photon utilization. To effectively utilize the light energy and improve the efficiency of photocatalytic degradation, it is necessary to subject a positive potential bias to the catalyst electrode system. Zhu’s group made their efforts to combine the photocatalysis of PDI self-assembly and applied electric field to achieve more robust photocatalytic performance compared with the photocatalytic and electrocatalytic processes (Fig. 8(c))[
5.2. Energy production
5.2.1. O2 evolution
Photocatalytic dioxygen production is a key scientific problem that needs to be solved urgently in the field of photocatalysis, mainly because dioxygen production acts as the decisive half-step for the overall water splitting reaction. The photocatalytic hydrolysis to produce dioxygen requires four electrons to participate in photocatalytic reactions, that is, the accumulation of holes on the heterogeneous photocatalyst, which determines photocatalytic reaction kinetics. Moreover, its overpotential is relatively higher, and often requires a lower VB potential to fit the requirement of water oxidation. It has been found that PDI self-assembly has a deep VB, higher than the oxidation potential of H2O/O2, and meanwhile the construction of PDI self-assembly favors the delocalization and accumulation of photogenerated holes, hence it can potentially catalyze dioxygen generation with four-electron transfer driven by visible light. In 2004, Kirner et al. reported that phosphonate-functionalized PDI-sensitized CoOx as an effective photoelectrocatalyst to realize oxidation of H2O, wherein PDI is not a photocatalyst[
![]()
Figure 10.(Color online) (a) In vitro viability of HeLa cells with different concentrations of H-PDI and J-PDI at 600 ± 15 nm with permission from Ref. [
5.2.2. H2 evolution
Generally, the potential of the CB of PDI self-assembly is lower than the reduction potential of H+/H2; therefore, the kinetic process of its photocatalytic dihydrogen production is unfavorable in thermodynamics. In terms of photocatalytic dihydrogen production, PDIs were initially considered as dye-sensitizers or co-catalysts to assist photocatalytic hydrolysis of host photocatalysts, mainly benefiting from its strong visible-light absorption, photochemical stability and energy level matching with Zn0.5Cd0.5S, TiO2 or C3N4[
It is worth mentioning (perylene monoimide) PMI molecules whose structure is mostly similar to PDI molecules. Unlike the PDI scaffold, PMI molecules bear only one part of amide and side chain, but it can also form a supramolecular self-assembly by Π–Π stacking behaviors, and Π-electrons inside can migrate along the Π–Π stacking direction. The photocatalytic hydrogen production driven by hydrogel scaffold built with PMI supramolecular self-assembly was attempted by Stupp's group in 2014[
5.3. Organic synthesis
In past decades, visible light-mediated organic synthesis has been derived as a very useful tool for building various natural products, medicine chemicals and organic functional materials[
Under visible-light excitation, PDI molecules would undergo direct single-electron transfer with some reducing agents to propagate some active intermediate species-PDI anion radicals with strong reducing ability. Accordingly, Ghosh et al. used PDI as a homogeneous photocatalyst to reduce a series of chlorinated compounds, achieving the effect of dehalogenation. After that, Schanze’s group provided a direct evidence for the photoinduced electron-transfer process from excited PDI anion radicals to aromatic halogen via transient absorption spectra[
However, the reducing capability of PDI self-assembly is rather weak, so it needs some extra driving conditions to achieve the effect of photocatalytic organic reduction. First, we can use some electron-donating chemicals (such as triethylamine or ammonia) to stabilize photo-generated electrons in PDI self-assembly and extend their lifetimes rest on the surface of PDI self-assembly; second, PDI generates a few PDI anion radicals in PDI self-assembly under the excitation of visible light. Upon re-excitation by light, PDI anion radicals absorb photons once again to form an excited state with stronger reducing power. DFT calculations showcase that the reduction potentials of PDI anion radicals and their excited states are –1.22 and +1.22 eV, respectively, which are much higher than the potentials of LUMO and HOMO levels of PDI molecules corresponding to –3.57 and –6.11 eV, respectively[
There are still some difficulties that need to be resolved in photocatalytic organic synthesis over PDI self-assembly. For example, if PDI molecules are electronically coupled with metal ions, photogenerated electrons and active intermediate species may be quenched due to electron/energy transfer, which would in turn reduce photocatalytic efficacy of PDI self-assembly. Therefore, it is critical to select a suitable metal ion as a coupling agent with PDI. On the other hand, the aforementioned heterogeneous PDI self-assembly presents excellent photocatalytic efficiency, mainly because of Π–Π stacking structure. However, in real photocatalytic process, PDI self-assembly easily depolymerizes due to the change in the surrounding environment, which distorts the highly ordered Π–Π stacking structure.
5.4. Photodynamic/photothermal therapy
PDI self-assembly displays excellent photogenerated charge separation capability, generating photogenerated electrons and holes. Moreover, along the Π–Π stacking direction in PDI self-assembly, photogenerated electrons and holes can be separated spatially. Through energy/electron transfer, photogenerated electrons on the surface of PDI self-assembly would interact with O2 to form singlet oxygen species (1O2) and superoxide radicals (•O2–), which play an important role in photocatalytic degradation. The holes in PDI self-assembly can oxidize organic pollutants and water molecules. Cancer is undoubtedly a huge threat to human physical health. Cancer treatment is a great challenge encountered in modern medical field. The currently used cancer treatment methods generally include surgical resection, chemotherapy, radiotherapy, endocrine therapy, and immunotherapy. But these traditional treatments may cause irreparable harm to the organizations. PDI self-assembly is a relatively safe and biocompatible chemical[
In addition, PDI-based organic semiconductors are widely used in photoacoustic imaging and photothermal therapy because of their thermal stability, high light-to-heat conversion, and simple modifiability[
6. Summary and outlook
As a common pigment, the structure of the PDI molecule contains a large Π-conjugated perylene, cyclic amides and side chains. DFT calculations present that the C=C bond between the two naphthalene units of PDI molecule has the characteristics of the C–C bond, so that certain rotational distortion comes up. Unlike most of dyes, PDI molecules have one important feature, that is, through Π–Π stacking interaction and side-to-side chain effect, PDI molecules can aggregate to form supramolecular self-assembly with PDI molecules as building blocks. We can change PDI molecule structures by modification to perylene bay and side chains of PDI molecules. For one thing, the solubility of PDI molecules in organic solvents can thus be increased. On the other hand, the self-assembly mode of PDI molecules can be changed obviously, thereby effectively regulating morphology of PDI self-assembly. In a suitable solvent, the self-assembly process of PDI molecules occurs with controllable morphology through Π–Π stacking and side-to-side chain interactions. The PDI self-assembly methods include modifying PDI molecules with substituents, introducing metal ions, and changing dispersion solvents. The self-assembling of PDI molecules is a dynamic equilibrium process between aggregation and disaggregation, which is largely affected by PDI molecule structures and the external environment. According to this, the morphologies of PDI self-assembly can be effectively modulated to obtain desired well-defined supramolecular architectures. The PDI self-assembly has a band-like electronic structure with deep VB. In addition, the Π–Π stacking structure is conducive to the transfer of photogenerated charges along the Π–Π stacking direction, thereby reducing the recombination of photogenerated electrons and holes. Therefore, PDI self-assembly can be used in photocatalytic degradation, photocatalytic water splitting into H2/O2, photocatalytic organic synthesis and light-driven disease therapy. The emergency of the active species-PDI anion radicals in PDI self-assembly makes photocatalytic reduction possible in organic synthesis. A series of PDI self-assembly-based composites have been fabricated to further extend light absorption range, reduce recombination of photogenerated charges in PDI self-assembly, and increase the specific surface area of photocatalytic systems.
Whilst PDI self-assembly is a highly ordered supramolecular structure formed by PDI molecules through Π–Π stacking interaction, unlike C3N4, it is currently difficult to completely verify the clear structure of PDI self-assembly through theoretical calculations and experimental means, partly because non-covalent bonds make supramolecular structures some large variables[
The overlapping of PDI molecular orbitals is beneficial to the formation of band-like electronic energy band structure. Upon excitation by visible light, the electrons in the VB of PDI self-assembly transition to the conduction band, forming excited charge carriers. The charge carriers can move rapidly along the Π–Π stacking direction, thereby realizing the spatial separation of photogenerated electrons and holes. As an electron-defective pigment, neutral PDI molecules can be converted into PDI anion/dianion radicals, as well as existence in PDI self-assembly, but its real role in photocatalysis remains elusive except of photocatalytic organic synthesis, e.g. the reduction function of the excited states of anion/dianion radicals albeit the shorter lifetimes[
In general, the electronic structure of PDI self-assembly can be further adjusted by modifying the electronic structure of the individual molecule. Since the Π–Π stacking interaction between PDI molecules has a significant effect on the electronic structure of PDI self-assembly, it is also a very effective way to modulate the PDI self-assembled mode. Such stacking arrangements are diversified, hence flexible electronic structure control can be achieved[
To further optimize the photocatalytic performance of the PDI self-assembly, we can proceed from three main aspects—enhancing the utilization of the full-spectrum sunlight, promoting the further separation of photogenerated charges, and building a stronger VB and CB potential. Though PDI self-assembly exhibits excellent visible light absorption performance, it lacks effective utilization of the ultraviolet region. This problem can be solved by modifying the molecular structures of PDI or combining with ultraviolet photocatalysts[
Especially for the photocatalytic water splitting performance over PDI self-assembly, compared with other inorganic semiconductor materials, there is still a big gap in photocatalytic performance of PDI self-assembly. Under the interaction with water molecules, photo-generated charge conduction in PDI self-assembles may be hindered. On the other hand, the overpotentials of the photolytic water over PDI self-assembly are large, which is not conducive to the transport of photogenerated charges at the solid-liquid interface. To clarify the specific mechanism of the photocatalytic process is uncovering one of the keys of this door to explore the key factors that affect its photocatalytic performance. Using PDI self-assembly as a photocatalyst is a promising alternative to traditional photocatalysts. It can be believed that in the near future, this type of photocatalysts will make a greater breakthrough progress in photocatalysis and function as traditional photocatalysts.
Acknowledgements
We acknowledge the financial support from the National Natural Science Foundation of China (No. 21972052). We appreciate Prof. Yongfa Zhu from Tsinghua University for his suggestions to this review paper. S.O. thanks the financial support from the “Guizi Scholar” Program of Central China Normal University.
References
[1] A J Bard. Photoelectrochemistry. Science, 207, 139(1980).
[2] M R Hoffmann, S T Martin, W Choi et al. Environmental applications of semiconductor photocatalysis. Chem Rev, 95, 69(1995).
[3] P D Quay, B Tilbrook, C S Wong. Oceanic uptake of fossil fuel CO2: Carbon-13 evidence. Science, 256, 74(1992).
[4] O Gustafsson, M Krusa, Z Zencak et al. Brown clouds over south asia: biomass or fossil fuel combustion. Science, 323, 495(2009).
[5] T M L Wigley. Could reducing fossil-fuel emissions cause global warming. Nature, 349, 503(1991).
[6] H Tong, S Ouyang, Y P Bi et al. Nano-photocatalytic materials: Possibilities and challenges. Adv Mater, 24, 229(2012).
[7] A Kudo, Y Miseki. Heterogeneous photocatalyst materials for water splitting. Chem Soc Rev, 38, 253(2009).
[8] W Q Wei, D Liu, Z Wei et al. Short-range π–π stacking assembly on P25 TiO2 nanoparticles for enhanced visible-light photocatalysis. ACS Catal, 7, 652(2017).
[9] W Q Wei, Z Wei, D Liu et al. Enhanced visible-light photocatalysis via back-electron transfer from palladium quantum dots to perylene diimide. Appl Catal B, 230, 49(2018).
[10] W Q Wei, Y F Zhu. TiO2@perylene diimide full-spectrum photocatalysts via semi-core–shell structure. Small, 15, 1903933(2019).
[11] W Hu, L Lin, R Q Zhang et al. Highly efficient photocatalytic water splitting over edge-modified phosphorene nanoribbons. J Am Chem Soc, 139, 15429(2017).
[12] H Fujito, H Kunioku, D Kato et al. Layered perovskite oxychloride Bi4NbO8Cl: A stable visible light responsive photocatalyst for water splitting. ChemInform, 138, 2082(2016).
[13] Z Wei, M L Liu, Z J Zhang et al. Efficient visible-light-driven selective oxygen reduction to hydrogen peroxide by oxygen-enriched graphitic carbon nitride polymers. Energy Environ Sci, 11, 2581(2018).
[14] G T Zeng, J Qiu, Z Li et al. CO2 reduction to methanol on TiO2-passivated GaP photocatalysts. ACS Catal, 4, 3512(2014).
[15] X Li, J G Yu, M Jaroniec et al. Cocatalysts for selective photoreduction of CO2 into solar fuels. Chem Rev, 119, 3962(2019).
[16] L Zeng, T Liu, C He et al. Organized aggregation makes insoluble perylene diimide efficient for the reduction of aryl halides via consecutive visible light-induced electron-transfer processes. J Am Chem Soc, 138, 3958(2016).
[17] I Ghosh, T Ghosh, J I Bardagi et al. Reduction of aryl halides by consecutive visible light-induced electron transfer processes. Science, 346, 725(2014).
[18] P K J Robertson, J M C Robertson, D W Bahnemann. Removal of microorganisms and their chemical metabolites from water using semiconductor photocatalysis. J Hazard Mater, 211/212, 161(2012).
[19] J Wang, D Liu, Y F Zhu et al. Supramolecular packing dominant photocatalytic oxidation and anticancer performance of PDI. Appl Catal B, 231, 251(2018).
[20] A Fujishima, K Honda. Electrochemical photolysis of water at a semiconductor electrode. Nature, 238, 37(1972).
[21] Z G Chai, T T Zeng, Q Li et al. Efficient visible light-driven splitting of alcohols into hydrogen and corresponding carbonyl compounds over a Ni-modified CdS photocatalyst. J Am Chem Soc, 138, 10128(2016).
[22] J Q Hu, A L Liu, H L Jin et al. A versatile strategy for shish-kebab-like multi-heterostructured chalcogenides and enhanced photocatalytic hydrogen evolution. J Am Chem Soc, 137, 11004(2015).
[23] H Song, X G Meng, S Y Wang et al. Direct and selective photocatalytic oxidation of CH4 to oxygenates with O2 on cocatalysts/ZnO at room temperature in water. J Am Chem Soc, 141, 20507(2019).
[24] W W He, H K Kim, W G Wamer et al. Photogenerated charge carriers and reactive oxygen species in ZnO/Au hybrid nanostructures with enhanced photocatalytic and antibacterial activity. J Am Chem Soc, 136, 750(2014).
[25] K Zhang, J L Liu, L Y Wang et al. Near-complete suppression of oxygen evolution for photoelectrochemical H2O oxidative H2O2 synthesis. J Am Chem Soc, 142, 8641(2020).
[26] Y Y Yu, K Ma, R Zhuang et al. Hydroxyl-mediated formation of highly dispersed SnO2/TiO2 heterojunction via pulsed chemical vapor deposition to enhance photocatalytic activity. Ind Eng Chem Res, 58, 14655(2019).
[27] Y Y Wang, W J Jiang, W J Luo et al. Ultrathin nanosheets g-C3N4@Bi2WO6 core-shell structure via low temperature reassembled strategy to promote photocatalytic activity. Appl Catal B, 237, 633(2018).
[28] J J Yang, D M Chen, Y Zhu et al. 3D–3D porous Bi2WO6/ graphene hydrogel composite with excellent synergistic effect of adsorption-enrichment and photocatalytic degradation. Appl Catal B, 205, 228(2017).
[29] A Iwase, S Yoshino, T Takayama et al. Water splitting and CO2 reduction under visible light irradiation using Z-scheme systems consisting of metal sulfides, CoO
[30] Z G Zou, J H Ye, K Sayama et al. Direct splitting of water under visible light irradiation with an oxide semiconductor photocatalyst. Nature, 414, 625(2001).
[31] B Weng, M Y Qi, C Han et al. Photocorrosion inhibition of semiconductor-based photocatalysts: Basic principle, current development, and future perspective. ACS Catal, 9, 4642(2019).
[32] S Ghosh, N A Kouame, L Ramos et al. Conducting polymer nanostructures for photocatalysis under visible light. Nat Mater, 14, 505(2015).
[33] F X Yang, S S Cheng, X T Zhang et al. 2D organic materials for optoelectronic applications. Adv Mater, 30, 1702415(2018).
[34] S W Cao, J Low, J G Yu et al. Polymeric photocatalysts based on graphitic carbon nitride. Adv Mater, 27, 2150(2015).
[35] N N Zhao, L M Yan, X Y Zhao et al. Versatile types of organic/inorganic nanohybrids: From strategic design to biomedical applications. Chem Rev, 119, 1666(2019).
[36] L L Li, Y Chen, J J Zhu. Recent advances in electrochemiluminescence analysis. Anal Chem, 89, 358(2017).
[37] I Choudhuri, P Bhauriyal, B Pathak. Recent advances in graphene-like 2D materials for spintronics applications. Chem Mater, 31, 8260(2019).
[38] W H Niu, Y Yang. Graphitic carbon nitride for electrochemical energy conversion and storage. ACS Energy Lett, 3, 2796(2018).
[39] Z Meng, R M Stolz, L Mendecki et al. Electrically-transduced chemical sensors based on two-dimensional nanomaterials. Chem Rev, 119, 478(2019).
[40] W J Ong, L L Tan, Y H Ng et al. Graphitic carbon nitride (g-C3N4)-based photocatalysts for artificial photosynthesis and environmental remediation: Are we a step closer to achieving sustainability. Chem Rev, 116, 7159(2016).
[41] Z H Wang, X Hu, Z Z Liu et al. Recent developments in polymeric carbon nitride-derived photocatalysts and electrocatalysts for nitrogen fixation. ACS Catal, 9, 10260(2019).
[42] X C Wang, K Maeda, A Thomas et al. A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat Mater, 8, 76(2009).
[43] H Takeda, H Kamiyama, K Okamoto et al. Highly efficient and robust photocatalytic systems for CO2 reduction consisting of a Cu(I) photosensitizer and Mn(I) catalysts. J Am Chem Soc, 140, 17241(2018).
[44] R F Higgins, S M Fatur, S G Shepard et al. Uncovering the roles of oxygen in Cr(III) photoredox catalysis. J Am Chem Soc, 138, 5451(2016).
[45] D C Hong, T Kawanishi, Y Tsukakoshi et al. Efficient photocatalytic CO2 reduction by a Ni(II) complex having pyridine pendants through capturing a Mg2+ ion as a lewis-acid cocatalyst. J Am Chem Soc, 141, 20309(2019).
[46] D Zhang, L Z Wu, L Zhou et al. Photocatalytic hydrogen production from hantzsch 1, 4-dihydropyridines by platinum(II) terpyridyl complexes in homogeneous solution. J Am Chem Soc, 126, 3440(2004).
[47] S Fernández, F Franco, C Casadevall et al. A unified electro- and photocatalytic CO2 to CO reduction mechanism with aminopyridine cobalt complexes. J Am Chem Soc, 142, 120(2020).
[48] B Xu, L Troian-Gautier, R Dykstra et al. Photocatalyzed diastereoselective isomerization of cinnamyl chlorides to cyclopropanes. J Am Chem Soc, 142, 6206(2020).
[49] M Elvington, J Brown, S M Arachchige et al. Photocatalytic hydrogen production from water employing a Ru, Rh, Ru molecular device for photoinitiated electron collection. J Am Chem Soc, 129, 10644(2007).
[50] P L Cheung, S C Kapper, T Zeng et al. Improving photocatalysis for the reduction of CO2 through non-covalent supramolecular assembly. J Am Chem Soc, 141, 14961(2019).
[51] E J Rabe, K L Corp, A L Sobolewski et al. Proton-coupled electron transfer from water to a model heptazine-based molecular photocatalyst. J Phys Chem Lett, 9, 6257(2018).
[52] C Wang, Z G Xie, K E de Krafft et al. Doping metal–organic frameworks for water oxidation, carbon dioxide reduction, and organic photocatalysis. J Am Chem Soc, 133, 13445(2011).
[53] X J Yang, T Liang, J X Sun et al. Template-directed synthesis of photocatalyst-encapsulating metal-organic frameworks with boosted photocatalytic activity. ACS Catal, 9, 7486(2019).
[54] M B Chambers, X Wang, L Ellezam et al. Maximizing the photocatalytic activity of metal–organic frameworks with aminated-functionalized linkers: Substoichiometric effects in MIL-125-NH2. J Am Chem Soc, 139, 8222(2017).
[55] P F Wei, M Z Qi, Z P Wang et al. Benzoxazole-linked ultrastable covalent organic frameworks for photocatalysis. J Am Chem Soc, 140, 4623(2018).
[56] Y Wan, L Wang, H Xu et al. A simple molecular design strategy for two-dimensional covalent organic framework capable of visible-light-driven water splitting. J Am Chem Soc, 149, 4508(2020).
[57] Q Z Luo, L L Bao, D S Wang et al. Preparation and strongly enhanced visible light photocatalytic activity of TiO2nanoparticles modified by conjugated derivatives of polyisoprene. J Phys Chem C, 116, 25806(2012).
[58] D Floresyona, F Goubard, P H Aubert et al. Highly active poly(3-hexylthiophene) nanostructures for photocatalysis under solar light. Appl Catal B, 209, 23(2017).
[59] M Zhang, W D Rouch, R D McCulla. Conjugated polymers as photoredox catalysts: Visible-light-driven reduction of aryl aldehydes by poly(p-phenylene). Eur J Org Chem, 2012, 6187(2012).
[60] B Muktha, G Madras, T N Guru Row et al. Conjugated polymers for photocatalysis. J Phys Chem B, 111, 7994(2007).
[61] S Ghosh, A K Mallik, R N Basu. Enhanced photocatalytic activity and photoresponse of poly(3, 4-ethylenedioxythiophene) nanofibers decorated with gold nanoparticle under visible light. Sol Energy, 159, 548(2018).
[62] L W Li, Z X Cai, Q H Wu et al. Rational design of porous conjugated polymers and roles of residual palladium for photocatalytic hydrogen production. J Am Chem Soc, 138, 7681(2016).
[63] Z J Zhang, Y F Zhu, X J Chen et al. A full-spectrum metal-free porphyrin supramolecular photocatalyst for dual functions of highly efficient hydrogen and oxygen evolution. Adv Mater, 31, 1806626(2019).
[64] D Liu, J Wang, X J Bai et al. Self-assembled PDINH supramolecular system for photocatalysis under visible light. Adv Mater, 28, 7284(2016).
[65] Z Wei, J S Hu, K J Zhu et al. Self-assembled polymer phenylethnylcopper nanowires for photoelectrochemical and photocatalytic performance under visible light. Appl Catal B, 226, 616(2018).
[66] F Würthner, C R Saha-Möller, B Fimmel et al. Perylene bisimide dye assemblies as archetype functional supramolecular materials. Chem Rev, 116, 962(2016).
[67]
[68] F Wuerthner. Perylene bisimide dyes as versatile building blocks for functional supramolecular architectures. ChemInform, 35, 1564(2004).
[69] G Saito, Y Yoshida. Development of conductive organic molecular assemblies: Organic metals, superconductors, and exotic functional materials. ChemInform, 38, 1(2007).
[70] A S Weingarten, R V Kazantsev, L C Palmer et al. Self-assembling hydrogel scaffolds for photocatalytic hydrogen production. Nat Chem, 6, 964(2014).
[71] D M Ke, C L Zhan, S P Xu et al. Self-assembled hollow nanospheres strongly enhance photoluminescence. J Am Chem Soc, 133, 11022(2011).
[72] K Balakrishnan, A Datar, T Naddo et al. Effect of side-chain substituents on self-assembly of perylene diimide molecules: Morphology control. J Am Chem Soc, 128, 7390(2006).
[73] S Bai, S Debnath, N Javid et al. Differential self-assembly and tunable emission of aromatic peptide bola-amphiphiles containing perylene bisimide in polar solvents including water. Langmuir, 30, 7576(2014).
[74] J Wang, W Shi, D Liu et al. Supramolecular organic nanofibers with highly efficient and stable visible light photooxidation performance. Appl Catal B, 202, 289(2017).
[75] K Balakrishnan, A Datar, R Oitker et al. Nanobelt self-assembly from an organic n-type semiconductor: Propoxyethyl-PTCDI. J Am Chem Soc, 127, 10496(2005).
[76] P Jonkheijm, P van der Schoot, A P H J Schenning et al. Probing the solvent-assisted nucleation pathway in chemical self-assembly. Science, 313, 80(2006).
[77] Q Y Gong, J Xing, Y J Huang et al. Perylene diimide oligomer nanoparticles with ultrahigh photothermal conversion efficiency for cancer theranostics. ACS Appl Bio Mater, 3, 1607(2020).
[78] L Zang. Interfacial donor–acceptor engineering of nanofiber materials to achieve photoconductivity and applications. Acc Chem Res, 48, 2705(2015).
[79] Y K Che, A Datar, X M Yang et al. Enhancing one-dimensional charge transport through intermolecular π-electron delocalization: Conductivity improvement for organic nanobelts. J Am Chem Soc, 129, 6354(2007).
[80] L Zang, Y K Che, J S Moore. One-dimensional self-assembly of planar π-conjugated molecules: Adaptable building blocks for organic nanodevices. Acc Chem Res, 41, 1596(2008).
[81] H Miao, J Yang, G L Peng et al. Enhancement of the degradation ability for organic pollutants via the synergistic effect of photoelectrocatalysis on a self-assembled perylene diimide (SA-PDI) thin film. Sci Bull, 64, 896(2019).
[82] J Yang, H Miao, Y X Wei et al. Π-Π Interaction between self-assembled perylene diimide and 3D graphene for excellent visible-light photocatalytic activity. Appl Catal B, 240, 225(2019).
[83] J Yang, H Miao, W L Li et al. Designed synthesis of a p-Ag2S/n-PDI self-assembled supramolecular heterojunction for enhanced full-spectrum photocatalytic activity. J Mater Chem A, 7, 6482(2019).
[84] H Miao, J Yang, Y X Wei et al. Visible-light photocatalysis of PDI nanowires enhanced by plasmonic effect of the gold nanoparticles. Appl Catal B, 239, 61(2018).
[85] Y X Wei, M G Ma, W L Li et al. Enhanced photocatalytic activity of PTCDI-C60 via π-π interaction. Appl Catal B, 238, 302(2018).
[86] Z J Zhang, J Wang, D Liu et al. Highly efficient organic photocatalyst with full visible light spectrum through π–π stacking of TCNQ–PTCDI. ACS Appl Mater Interfaces, 8, 30225(2016).
[87] S Chen, C Wang, B R Bunes et al. Enhancement of visible-light-driven photocatalytic H2 evolution from water over g-C3N4 through combination with perylene diimide aggregates. Appl Catal A, 498, 63(2015).
[88] S Chen, D L Jacobs, J K Xu et al. 1D nanofiber composites of perylene diimides for visible-light-driven hydrogen evolution from water. RSC Adv, 4, 48486(2014).
[89] Z Yang, W P Fan, J H Zou et al. Precision cancer theranostic platform by
[90] A Stergiou, N Tagmatarchis. Fluorene–perylene diimide arrays onto graphene sheets for photocatalysis. ACS Appl Mater Interfaces, 8, 21576(2016).
[91] S Chen, P Slattum, C Y Wang et al. Self-assembly of perylene imide molecules into 1D nanostructures: Methods, morphologies, and applications. Chem Rev, 115, 11967(2015).
[92] C Huang, S Barlow, S R Marder. Perylene-3, 4, 9, 10-tetracarboxylic acid diimides: Synthesis, physical properties, and use in organic electronics. J Org Chem, 76, 2386(2011).
[93] H Q Peng, L Y Niu, Y Z Chen et al. Biological applications of supramolecular assemblies designed for excitation energy transfer. Chem Rev, 115, 7502(2015).
[94] Y N Teo, E T Kool. DNA-multichromophore systems. Chem Rev, 112, 4221(2012).
[95] Z J Chen, M G Debije, T Debaerdemaeker et al. Tetrachloro-substituted perylene bisimide dyes as promising n-type organic semiconductors: Studies on structural, electrochemical and charge transport properties. ChemPhysChem, 5, 137(2004).
[96] F Würthner, A Sautter, J Schilling. Synthesis of diazadibenzoperylenes and characterization of their structural, optical, redox, and coordination properties. J Org Chem, 67, 3037(2002).
[97] P Yan, A Chowdhury, M W Holman et al. Self-organized perylene diimide nanofibers. J Phys Chem B, 109, 724(2005).
[98] Y K Che, A Datar, K Balakrishnan et al. Ultralong nanobelts self-assembled from an asymmetric perylene tetracarboxylic diimide. J Am Chem Soc, 129, 7234(2007).
[99] Y K Che, H L Huang, M Xu et al. Interfacial engineering of organic nanofibril heterojunctions into highly photoconductive materials. J Am Chem Soc, 133, 1087(2011).
[100] F Graser, E Hädicke. Kristallstruktur und Farbe Bei Perylen-3, 4: 9, 10-bis(dicarboximid)-Pigmenten. Liebigs Ann Chem, 1980, 1994(1980).
[101] F Graser, E Hädike. Kristallstruktur und Farbe Bei Perylen-3, 4: 9, 10-bis(dicarboximid)-Pigmenten, 2. Liebigs Ann Chem, 1984, 483(1984).
[102] C W Struijk, A B Sieval, J E J Dakhorst et al. Liquid crystalline perylene diimides: architecture and charge carrier mobilities. J Am Chem Soc, 122, 11057(2000).
[103] A Datar, K Balakrishnan, X M Yang et al. Linearly polarized emission of an organic semiconductor nanobelt. J Phys Chem B, 110, 12327(2006).
[104] H Yamagata, D S Maxwell, J Fan et al. HJ-aggregate behavior of crystalline 7, 8, 15, 16-tetraazaterrylene: Introducing a new design paradigm for organic materials. J Phys Chem C, 118, 28842(2014).
[105] Y C Chen, J W Y Lam, R T K Kwok et al. Aggregation-induced emission: Fundamental understanding and future developments. Mater Horiz, 6, 428(2019).
[106] Y K Che, X M Yang, G L Liu et al. Ultrathin n-type organic nanoribbons with high photoconductivity and application in optoelectronic vapor sensing of explosives. J Am Chem Soc, 132, 5743(2010).
[107] F Rodler, B Schade, C M Jäger et al. Amphiphilic perylene–calix. J Am Chem Soc, 137, 3308(2015).
[108] J L Wang, Y Yu, L Z Zhang. Highly efficient photocatalytic removal of sodium pentachlorophenate with Bi3O4Br under visible light. Appl Catal B, 136/137, 112(2013).
[109] Y M Liang, S Q Lan, P Deng et al. Regioregular and regioirregular poly(selenophene-perylene diimide) acceptors for polymer–polymer solar cells. ACS Appl Mater Interfaces, 10, 32397(2018).
[110] X Li, H Wang, H Nakayama et al. Multi-sulfur-annulated fused perylene diimides for organic solar cells with low open-circuit voltage loss. ACS Appl Energy Mater, 2, 3805(2019).
[111] S K Samanta, I Song, J H Yoo et al. Organic n-channel transistors based on. ACS Appl Mater Interfaces, 10, 32444(2018).
[112] J Yang, Y Yin, F Chen et al. Comparison of three n-type copolymers based on benzodithiophene and naphthalene diimide/perylene diimide/fused perylene diimides for all-polymer solar cells application. ACS Appl Mater Interfaces, 10, 23263(2018).
[113] M Woodhouse, C L Perkins, M T Rawls et al. Non-conjugated polymers for organic photovoltaics: Physical and optoelectronic properties of poly(perylene diimides). J Phys Chem C, 114, 6784(2010).
[114] J Zhang, Y Li, J Huang et al. Ring-fusion of perylene diimide acceptor enabling efficient nonfullerene organic solar cells with a small voltage loss. J Am Chem Soc, 139, 16092(2017).
[115] T C An, J B An, Y P Gao et al. Photocatalytic degradation and mineralization mechanism and toxicity assessment of antivirus drug acyclovir: Experimental and theoretical studies. Appl Catal B, 164, 279(2015).
[116] M Iwase, K Yamada, T Kurisaki et al. Visible-light photocatalysis with phosphorus-doped titanium(IV) oxide particles prepared using a phosphide compound. Appl Catal B, 132/133, 39(2013).
[117] S Kitano, K Hashimoto, H Kominami. Photocatalytic degradation of 2-propanol over metal-ion-loaded titanium(IV) oxide under visible light irradiation: Effect of physical properties of nano-crystalline titanium(IV) oxide. Appl Catal B, 101, 206(2011).
[118] Q Li, J K Shang. Composite photocatalyst of nitrogen and fluorine codoped titanium oxide nanotube arrays with dispersed palladium oxide nanoparticles for enhanced visible light photocatalytic performance. Environ Sci Technol, 44, 3493(2010).
[119] Q Shi, S Murcia-López, P Y Tang et al. Role of tungsten doping on the surface states in BiVO4 photoanodes for water oxidation: Tuning the electron trapping process. ACS Catal, 8, 3331(2018).
[120] L J An, H Onishi. Electron–hole recombination controlled by metal doping sites in NaTaO3 photocatalysts. ACS Catal, 5, 3196(2015).
[121] X Liu, S Gao, H Xu et al. Stable blue TiO2–
[122] Q Zhu, Y Peng, L Lin et al. Green synthetic approach for Ti3+ self-doped TiO2–
[123] H W Huang, C Zhou, X C Jiao et al. Subsurface defect engineering in single-unit-cell Bi2WO6 monolayers boosts solar-driven photocatalytic performance. ACS Catal, 10, 1439(2020).
[124] D Jiang, W Z Wang, L Zhang et al. Insights into the surface-defect dependence of photoreactivity over CeO2 nanocrystals with well-defined crystal facets. ACS Catal, 5, 4851(2015).
[125] S K Cushing, F K Meng, J Y Zhang et al. Effects of defects on photocatalytic activity of hydrogen-treated titanium oxide nanobelts. ACS Catal, 7, 1742(2017).
[126] G Seybold, G Wagenblast. New perylene and violanthrone dyestuffs for fluorescent collectors. Dye Pigment, 11, 303(1989).
[127] M Sadrai, L Hadel, R R Sauers et al. Lasing action in a family of perylene derivatives: Singlet absorption and emission spectra, triplet absorption and oxygen quenching constants, and molecular mechanics and semiempirical molecular orbital calculations. J Phys Chem, 96, 7988(1992).
[128] M J Ahrens, M J Fuller, M R Wasielewski. Cyanated perylene-3, 4-dicarboximides and perylene-3, 4: 9, 10-bis(dicarboximide): Facile chromophoric oxidants for organic photonics and electronics. Chem Mater, 15, 2684(2003).
[129] Y Y Zhao, M R Wasielewski. 3, 4: 9, 10-Perylenebis(dicarboximide) chromophores that function as both electron donors and acceptors. Tetrahedron Lett, 40, 7047(1999).
[130] A S Lukas, Y Y Zhao, S E Miller et al. Biomimetic electron transfer using low energy excited states: A green perylene-based analogue of chlorophylla. J Phys Chem B, 106, 1299(2002).
[131] J I Yoshida, K Kataoka, R Horcajada et al. Modern strategies in electroorganic synthesis. Chem Rev, 108, 2265(2008).
[132] C Kingston, M D Palkowitz, Y Takahira et al. A survival guide for the "electro-curious". Acc Chem Res, 53, 72(2020).
[133] A Ruffoni, F Juliá, T D Svejstrup et al. Practical and regioselective amination of arenes using alkyl amines. Nat Chem, 11, 426(2019).
[134] J Bariwal, E van der Eycken. C–N bond forming cross-coupling reactions: An overview. Chem Soc Rev, 42, 9283(2013).
[135] K D Moeller. Synthetic applications of anodic electrochemistry. Tetrahedron, 56, 9527(2000).
[136] Q L Yang, X Y Wang, J Y Lu et al. Copper-catalyzed electrochemical C–H amination of arenes with secondary amines. J Am Chem Soc, 140, 11487(2018).
[137] T Morofuji, A Shimizu, J I Yoshida. Electrochemical C–H amination: Synthesis of aromatic primary amines viaN-arylpyridinium ions. J Am Chem Soc, 135, 5000(2013).
[138] W S Ham, J Hillenbrand, J Jacq et al. Divergent late-stage (hetero)aryl C–H amination by the pyridinium radical cation. Angew Chem Int Ed, 58, 532(2019).
[139] R Hayashi, A Shimizu, J I Yoshida. The stabilized cation pool method: Metal- and oxidant-free benzylic C–H/aromatic C–H cross-coupling. J Am Chem Soc, 138, 8400(2016).
[140] Z W Hou, Z Y Mao, Y Y Melcamu et al. Back cover: Electrochemical synthesis of imidazo-fused N-heteroaromatic compounds through a C–N bond-forming radical. Angew Chem Int Ed, 57, 1722(2018).
[141] Z W Hou, Z Y Mao, H B Zhao et al. Frontispiece: electrochemical C–H/N–H functionalization for the synthesis of highly functionalized (aza)indoles. Angew Chem Int Ed, 55, 9168(2016).
[142] S R Waldvogel, M Selt. Electrochemical allylic oxidation of olefins: Sustainable and safe. Angew Chem Int Ed, 55, 12578(2016).
[143] Y Y Jiang, K Xu, C C Zeng. Use of electrochemistry in the synthesis of heterocyclic structures. Chem Rev, 118, 4485(2018).
[144] M Yan, Y Kawamata, P S Baran. Synthetic organic electrochemical methods since 2000: On the verge of a renaissance. Chem Rev, 117, 13230(2017).
[145] A Jutand. Contribution of electrochemistry to organometallic catalysis. Chem Rev, 108, 2300(2008).
[146] R Z Feng, J A Smith, K D Moeller. Anodic cyclization reactions and the mechanistic strategies that enable optimization. Acc Chem Res, 50, 2346(2017).
[147] E Krieg, H Weissman, E Shimoni et al. Understanding the effect of fluorocarbons in aqueous supramolecular polymerization: Ultrastrong noncovalent binding and cooperativity. J Am Chem Soc, 136, 9443(2014).
[148] Q L Zhao, S Zhang, Y Liu et al. Tetraphenylethenyl-modified perylene bisimide: Aggregation-induced red emission, electrochemical properties and ordered microstructures. J Mater Chem, 22, 7387(2012).
[149] A D Hendsbee, J P Sun, W K Law et al. Synthesis, self-assembly, and solar cell performance of N-annulated perylene diimide non-fullerene acceptors. Chem Mater, 28, 7098(2016).
[150] G Li, Y B Zhao, J B Li et al. Synthesis, characterization, physical properties, and OLED application of single BN-fused perylene diimide. J Org Chem, 80, 196(2015).
[151] S Seifert, D Schmidt, F Würthner. An ambient stable core-substituted perylene bisimide dianion: Isolation and single crystal structure analysis. Chem Sci, 6, 1663(2015).
[152] N J Schuster, L A Joyce, D W Paley et al. The structural origins of intense circular dichroism in a waggling helicene nanoribbon. J Am Chem Soc, 142, 7066(2020).
[153] S K Lee, Y B Zu, A Herrmann et al. Electrochemistry, spectroscopy and electrogenerated chemiluminescence of perylene, terrylene, and quaterrylene diimides in aprotic solution. J Am Chem Soc, 121, 3513(1999).
[154] A D Zhang, W Jiang, Z H Wang. Fulvalene-embedded perylene diimide and its stable radical anion. Angew Chem, 132, 762(2020).
[155] B A Jones, A Facchetti, M R Wasielewski et al. Tuning orbital energetics in arylene diimide semiconductors. materials design for ambient stability of n-type charge transport. J Am Chem Soc, 129, 15259(2007).
[156] S R Peurifoy, E Castro, F Liu et al. Three-dimensional graphene nanostructures. J Am Chem Soc, 140, 9341(2018).
[157] G P Gao, N N Liang, H Geng et al. Spiro-fused perylene diimide arrays. J Am Chem Soc, 139, 15914(2017).
[158] B Liu, M Böckmann, W Jiang et al. Perylene diimide-embedded double. J Am Chem Soc, 142, 7092(2020).
[159] H Langhals. Cyclic carboxylic imide structures as structure elements of high stability. Novel developments in perylene dye chemistry. Heterocycles, 1, 477(1995).
[160] W Wang, L Q Wang, B J Palmer et al. Cyclization and catenation directed by molecular self-assembly. J Am Chem Soc, 128, 11150(2006).
[161] T A Barendt, L Ferreira, I Marques et al. Anion- and solvent-induced rotary dynamics and sensing in a perylene diimide. J Am Chem Soc, 139, 9026(2017).
[162] C M Pochas, K A Kistler, H Yamagata et al. Contrasting photophysical properties of star-shaped vs linear perylene diimide complexes. J Am Chem Soc, 135, 3056(2013).
[163] J Wang, Z Yang, X X Gao et al. Core-shell g-C3N4@ZnO composites as photoanodes with double synergistic effects for enhanced visible-light photoelectrocatalytic activities. Appl Catal B, 217, 169(2017).
[164] C C You, F Würthner. Self-assembly of ferrocene-functionalized perylene bisimide bridging ligands with Pt(II) corner to electrochemically active molecular squares. J Am Chem Soc, 125, 9716(2003).
[165] M C R Delgado, E G Kim, D A D S Filho et al. Tuning the charge-transport parameters of perylene diimide single crystals via end and/or core functionalization: A density functional theory investigation. J Am Chem Soc, 132, 3375(2010).
[166] Y J Kim, Y Lee, K Park et al. Hierarchical self-assembly of perylene diimide (PDI) crystals. J Phys Chem Lett, 11, 3934(2020).
[167] E J Zhou, J Z Cong, Q S Wei et al. Berichtigung: all-polymer solar cells from perylene diimide based copolymers: Material design and phase separation control. Angew Chem, 123, 8120(2011).
[168] Z H Luo, F Wu, T Zhang et al. Designing a perylene diimide/fullerene hybrid as effective electron transporting material in inverted perovskite solar cells with enhanced efficiency and stability. Angew Chem Int Ed, 58, 8520(2019).
[169] L F Dössel, V Kamm, I A Howard et al. Synthesis and controlled self-assembly of covalently linked hexa-peri-hexabenzocoronene/perylene diimide dyads as models to study fundamental energy and electron transfer processes. J Am Chem Soc, 134, 5876(2012).
[170] S B Jin, M Supur, M Addicoat et al. Creation of superheterojunction polymers via direct polycondensation: Segregated and bicontinuous donor–acceptor π-columnar arrays in covalent organic frameworks for long-lived charge separation. J Am Chem Soc, 137, 7817(2015).
[171] S Prathapan, S I Yang, J Seth et al. Synthesis and excited-state photodynamics of perylene–porphyrin dyads. 1. parallel energy and charge transfer via a diphenylethyne linker. J Phys Chem B, 105, 8237(2001).
[172] M P O'Neil, M P Niemczyk, W A Svec et al. Picosecond optical switching based on biphotonic excitation of an electron donor-acceptor-donor molecule. Science, 257, 63(1992).
[173] T van der Boom, R T Hayes, Y Y Zhao et al. Charge transport in photofunctional nanoparticles self-assembled from zinc 5, 10, 15, 20-tetrakis(perylenediimide)porphyrin building blocks. J Am Chem Soc, 124, 9582(2002).
[174] J Baram, E Shirman, N Ben-Shitrit et al. Control over self-assembly through reversible charging of the aromatic building blocks in photofunctional supramolecular fibers. J Am Chem Soc, 130, 14966(2008).
[175] C Jung, B K Müller, D C Lamb et al. A new photostable terrylene diimide dye for applications in single molecule studies and membrane labeling. J Am Chem Soc, 128, 5283(2006).
[176] V Marcon, D W Breiby, W Pisula et al. Understanding structure–mobility relations for perylene tetracarboxydiimide derivatives. J Am Chem Soc, 131, 11426(2009).
[177] R N Dsouza, U Pischel, W M Nau. Fluorescent dyes and their supramolecular host/guest complexes with macrocycles in aqueous solution. Chem Rev, 111, 7941(2011).
[178] X M Yin, Q X Wang, Y J Zheng et al. Molecular alignment and electronic structure of N, N'-dibutyl-3, 4, 9, 10-perylene-tetracarboxylic-diimide molecules on MoS2 surfaces. ACS Appl Mater Interfaces, 9, 5566(2017).
[179] L Gigli, R Arletti, G Tabacchi et al. Structure and host-guest interactions of perylene-diimide dyes in zeolite L nanochannels. J Phys Chem C, 122, 3401(2018).
[180] N Liu, M M Shi, X W Pan et al. Photoinduced electron transfer and enhancement of photoconductivity in silicon nanoparticles/perylene diimide composites in a polymer matrix. J Phys Chem C, 112, 15865(2008).
[181] A F Xie, B Liu, J E Hall et al. Self-assembled photoactive polyelectrolyte/perylene-diimide composites. Langmuir, 21, 4149(2005).
[182] D Gosztola, M P Niemczyk, W Svec et al. Excited doublet states of electrochemically generated aromatic imide and diimide radical anions. J Phys Chem A, 104, 6545(2000).
[183] O O Adegoke, I H Jung, M Orr et al. Effect of acceptor strength on optical and electronic properties in conjugated polymers for solar applications. J Am Chem Soc, 137, 5759(2015).
[184] S Shoaee, T M Clarke, C Huang et al. Acceptor energy level control of charge photogeneration in organic donor/acceptor blends. J Am Chem Soc, 132, 12919(2010).
[185] R K Dubey, M Niemi, K Kaunisto et al. Direct evidence of significantly different chemical behavior and excited-state dynamics of 1, 7- and 1, 6-regioisomers of pyrrolidinyl-substituted perylene diimide. Chem Eur J, 19, 6791(2013).
[186] S T Ryan, R M Young, s J J Henkelis et al. Energy and electron transfer dynamics within a series of perylene diimide/cyclophane systems. J Am Chem Soc, 137, 15299(2015).
[187] A M Ramos, E H A Beckers, T Offermans et al. Photoinduced multistep electron transfer in an oligoaniline–oligo(p-phenylene vinylene)–perylene diimide molecular array. J Phys Chem A, 108, 8201(2004).
[188] S T J Ryan, J del Barrio, I Ghosh et al. Efficient host–guest energy transfer in polycationic cyclophane–perylene diimide complexes in water. J Am Chem Soc, 136, 9053(2014).
[189] E R D Santos, J Pina, T Venâncio et al. Photoinduced energy and electron-transfer reactions by polypyridine ruthenium(II) complexes containing a derivatized perylene diimide. J Phys Chem C, 120, 22831(2016).
[190] Y Song, W Zhang, S J He et al. Perylene diimide and luminol as potential-resolved electrochemiluminescence nanoprobes for dual targets immunoassay at low potential. ACS Appl Mater Interfaces, 11, 33676(2019).
[191] Y W Huang, L N Fu, W J Zou et al. Ammonia sensory properties based on single-crystalline micro/nanostructures of perylenediimide derivatives: Core-substituted effect. J Phys Chem C, 115, 10399(2011).
[192] Y K Che, X M Yang, S Loser et al. Expedient vapor probing of organic amines using fluorescent nanofibers fabricated from an n-type organic semiconductor. Nano Lett, 8, 2219(2008).
[193] N J Schuster, D W Paley, S Jockusch et al. Electron delocalization in perylene diimide helicenes. Angew Chem Int Ed, 55, 13519(2016).
[194] M L Ball, B Y Zhang, Q Z Xu et al. Influence of molecular conformation on electron transport in giant, conjugated macrocycles. J Am Chem Soc, 140, 10135(2018).
[195] P R L Malenfant, C D Dimitrakopoulos, J D Gelorme et al. N-type organic thin-film transistor with high field-effect mobility based on a N, N'-dialkyl-3, 4, 9, 10-perylene tetracarboxylic diimide derivative. Appl Phys Lett, 80, 2517(2002).
[196] X Y Wang, J Q Meng, X Yang et al. Fabrication of a perylene tetracarboxylic diimide–graphitic carbon nitride heterojunction photocatalyst for efficient degradation of aqueous organic pollutants. ACS Appl Mater Interfaces, 11, 588(2019).
[197] W Q Liu, S Bobbala, C L Stern et al. XCage: A tricyclic octacationic receptor for perylene diimide with picomolar affinity in water. J Am Chem Soc, 142, 3165(2020).
[198] Q C Zhang, L Jiang, J Wang et al. Photocatalytic degradation of tetracycline antibiotics using three-dimensional network structure perylene diimide supramolecular organic photocatalyst under visible-light irradiation. Appl Catal B, 277, 119122(2020).
[199] P Chen, L Blaney, G Cagnetta et al. Degradation of ofloxacin by perylene diimide supramolecular nanofiber sunlight-driven photocatalysis. Environ Sci Technol, 53, 1564(2019).
[200] D M Guldi. Fullerene–porphyrin architectures; photosynthetic antenna and reaction center models. Chem Soc Rev, 31, 22(2002).
[201] L Liu, M T Yue, J R Lu et al. The enrichment of photo-catalysis via self-assembly perylenetetracarboxylic acid diimide polymer nanostructures incorporating TiO2 nano-particles. Appl Surf Sci, 456, 645(2018).
[202] R F Araújo, C J R Silva, M C Paiva et al. Efficient dispersion of multi-walled carbon nanotubes in aqueous solution by non-covalent interaction with perylene bisimides. RSC Adv, 3, 24535(2013).
[203] Y Liu, E W Zhu, L Y Bian et al. Robust graphene dispersion with amphiphlic perylene-polyglycidol. Mater Lett, 118, 188(2014).
[204] C Oelsner, C Schmidt, F Hauke et al. Interfacing strong electron acceptors with single wall carbon nanotubes. J Am Chem Soc, 133, 4580(2011).
[205] Y Tsarfati, V Strauss, S Kuhri et al. Dispersing perylene diimide/SWCNT hybrids: Structural insights at the molecular level and fabricating advanced materials. J Am Chem Soc, 137, 7429(2015).
[206] K Zhang, J Wang, W J Jiang et al. Self-assembled perylene diimide based supramolecular heterojunction with Bi2WO6 for efficient visible-light-driven photocatalysis. Appl Catal B, 232, 175(2018).
[207] Q Y Ji, Z Xu, W M Xiang et al. Enhancing the performance of pollution degradation through secondary self-assembled composite supramolecular heterojunction photocatalyst BiOCl/PDI under visible light irradiation. Chemosphere, 253, 126751(2020).
[208] Q Z Gao, J Xu, Z P Wang et al. Enhanced visible photocatalytic oxidation activity of perylene diimide/g-C3N4 n–n heterojunction via π–π interaction and interfacial charge separation. Appl Catal B, 271, 118933(2020).
[209] H L Wang, L L Zhao, X Q Liu et al. Novel hydrogen bonding composite based on copper phthalocyanine/perylene diimide derivatives p–n heterojunction with improved photocatalytic activity. Dye Pigment, 137, 322(2017).
[210] W G Zeng, T Cai, Y T Liu et al. An artificial organic-inorganic Z-scheme photocatalyst WO3@Cu@PDI supramolecular with excellent visible light absorption and photocatalytic activity. Chem Eng J, 381, 122691(2020).
[211] Y Cheng, R Q Song, K Wu et al. The enhanced visible-light-driven antibacterial performances of PTCDI-PANI(Fe(III)-doped) heterostructure. J Hazard Mater, 383, 121166(2020).
[212] X M Gao, K L Gao, X B Li et al. Hybrid PDI/BiOCl heterojunction with enhanced interfacial charge transfer for a full-spectrum photocatalytic degradation of pollutants. Catal Sci Technol, 10, 372(2020).
[213] T H Jeon, M S Koo, H Kim et al. Dual-functional photocatalytic and photoelectrocatalytic systems for energy- and resource-recovering water treatment. ACS Catal, 8, 11542(2018).
[214] C M Ding, J Y Shi, Z L Wang et al. Photoelectrocatalytic water splitting: Significance of cocatalysts, electrolyte, and interfaces. ACS Catal, 7, 675(2017).
[215] K R Brereton, A G Bonn, A J M Miller. Molecular photoelectrocatalysts for light-driven hydrogen production. ACS Energy Lett, 3, 1128(2018).
[216] Y Q Sheng, H Miao, J F Jing et al. Perylene diimide anchored graphene 3D structure via π–π interaction for enhanced photoelectrochemical degradation performances. Appl Catal B, 272, 118897(2020).
[217] J T Kirner, J J Stracke, B A Gregg et al. Visible-light-assisted photoelectrochemical water oxidation by thin films of a phosphonate-functionalized perylene diimide plus CoO
[218] J T Kirner, R G Finke. Sensitization of nanocrystalline metal oxides with a phosphonate-functionalized perylene diimide for photoelectrochemical water oxidation with a CoO
[219]
[220] V Kunz, V Stepanenko, F Würthner. Embedding of a ruthenium(ii) water oxidation catalyst into nanofibers via self-assembly. Chem Commun, 51, 290(2015).
[221] J X Li, Z J Li, C Ye et al. Visible light-induced photochemical oxygen evolution from water by 3, 4, 9, 10-perylenetetracarboxylic dianhydride nanorods as an n-type organic semiconductor. Catal Sci Technol, 6, 672(2016).
[222] Z Zhong, R F Li, W L Lin et al. One-dimensional nanocrystals of cobalt perylene diimide polymer with
[223] R J Zheng, M Zhang, X Sun et al. Perylene-3, 4, 9, 10-tetracarboxylic acid accelerated light-driven water oxidation on ultrathin indium oxide porous sheets. Appl Catal B, 254, 667(2019).
[224] M T Vagnini, A L Smeigh, J D Blakemore et al. Ultrafast photodriven intramolecular electron transfer from an iridium-based water-oxidation catalyst to perylene diimide derivatives. PNAS, 109, 15651(2012).
[225] S Chen, Y X Li, C Y Wang. Visible-light-driven photocatalytic H2 evolution from aqueous suspensions of perylene diimide dye-sensitized Pt/TiO2 catalysts. RSC Adv, 5, 15880(2015).
[226] T Sun, J G Song, J Jia et al. Real roles of perylenetetracarboxylic diimide for enhancing photocatalytic H2-production. Nano Energy, 26, 83(2016).
[227] Y Z Chen, A X Li, X Q Yue et al. Facile fabrication of organic/inorganic nanotube heterojunction arrays for enhanced photoelectrochemical water splitting. Nanoscale, 8, 13228(2016).
[228] L W Li, Z X Cai. Structure control and photocatalytic performance of porous conjugated polymers based on perylene diimide. Polym Chem, 7, 4937(2016).
[229] M C Nolan, J J Walsh, L L E Mears et al. pH dependent photocatalytic hydrogen evolution by self-assembled perylene bisimides. J Mater Chem A, 5, 7555(2017).
[230] R Wang, G Li, A D Zhang et al. Efficient energy-level modification of novel pyran-annulated perylene diimides for photocatalytic water splitting. Chem Commun, 53, 6918(2017).
[231] K Y Kong, S C Zhang, Y M Chu et al. A self-assembled perylene diimide nanobelt for efficient visible-light-driven photocatalytic H2 evolution. Chem Commun, 55, 8090(2019).
[232] J J Concepcion, R L House, J M Papanikolas et al. Chemical approaches to artificial photosynthesis. PANS, 109, 15560(2012).
[233] Y C Xu, J X Zheng, J O Lindner et al. Consecutive charging of a perylene bisimide dye by multistep low-energy solar-light-induced electron transfer towards H2 evolution. Angew Chem Int Ed, 59, 10363(2020).
[234] X Li, X Lv, Q Q Zhang et al. Self-assembled supramolecular system PDINH on TiO2 surface enhances hydrogen production. J Colloid Interface Sci, 525, 136(2018).
[235] L Yao, N Guijarro, F Boudoire et al. Establishing stability in organic semiconductor photocathodes for solar hydrogen production. J Am Chem Soc, 142, 7795(2020).
[236] A S Weingarten, R V Kazantsev, L C Palmer et al. Supramolecular packing controls H2photocatalysis in chromophore amphiphile hydrogels. J Am Chem Soc, 137, 15241(2015).
[237] R V Kazantsev, A J Dannenhoffer, A S Weingarten et al. Crystal-phase transitions and photocatalysis in supramolecular scaffolds. J Am Chem Soc, 139, 6120(2017).
[238] R V Kazantsev, A J Dannenhoffer, T Aytun et al. Molecular control of internal crystallization and photocatalytic function in supramolecular nanostructures. Chem, 4, 1596(2018).
[239] A S Weingarten, A J Dannenhoffer, R V Kazantsev et al. Chromophore dipole directs morphology and photocatalytic hydrogen generation. J Am Chem Soc, 140, 4965(2018).
[240] H Sai, A Erbas, A Dannenhoffer et al. Chromophore amphiphile-polyelectrolyte hybrid hydrogels for photocatalytic hydrogen production. J Mater Chem A, 8, 158(2020).
[241] O Dumele, J H Chen, J V Passarelli et al. Supramolecular energy materials. Adv Mater, 32, 1907247(2020).
[242] C K Prier, D A Rankic, D W C MacMillan. Visible light photoredox catalysis with transition metal complexes: Applications in organic synthesis. Chem Rev, 113, 5322(2013).
[243]
[244] C J IV Zeman, S Kim, F Zhang et al. Direct observation of the reduction of aryl halides by a photoexcited perylene diimide radical anion. J Am Chem Soc, 142, 2204(2020).
[245] I Ghosh. Excited radical anions and excited anions in visible light photoredox catalysis. Phys Sci Rev, 4, 20170185(2019).
[246] J T Shang, H Y Tang, H W Ji et al. Synthesis, characterization, and activity of a covalently anchored heterogeneous perylene diimide photocatalyst. Chin J Catal, 38, 2094(2017).
[247] L W Wang, X Zhang, X Yu et al. An all-organic semiconductor C3N4/PDINH heterostructure with advanced antibacterial photocatalytic therapy activity. Adv Mater, 31, 1901965(2019).
[248] Z Yang, X Y Chen. Semiconducting perylene diimide nanostructure: Multifunctional phototheranostic nanoplatform. Acc Chem Res, 52, 1245(2019).
[249] X M Hu, F Lu, L Chen et al. Perylene diimide-grafted polymeric nanoparticles chelated with Gd3+ for photoacoustic/T1-weighted magnetic resonance imaging-guided photothermal therapy. ACS Appl Mater Interfaces, 9, 30458(2017).
[250] W Tang, Z Yang, S Wang et al. Organic semiconducting photoacoustic nanodroplets for laser-activatable ultrasound imaging and combinational cancer therapy. ACS Nano, 12, 2610(2018).
[251] Z Yang, R Tian, J J Wu et al. Impact of semiconducting perylene diimide nanoparticle size on lymph node mapping and cancer imaging. ACS Nano, 11, 4247(2017).
[252] Q L Fan, K Cheng, Z Yang et al. Photoacoustic imaging: Perylene-diimide-based nanoparticles as highly efficient photoacoustic agents for deep brain tumor imaging in living mice. Adv Mater, 27, 774(2015).
[253] B Lü, Y F Chen, P Y Li et al. Stable radical anions generated from a porous perylenediimide metal-organic framework for boosting near-infrared photothermal conversion. Nat Commun, 10, 767(2019).
[254] R Englman, J Jortner. The energy gap law for radiationless transitions in large molecules. Mol Phys, 18, 145(1970).
Set citation alerts for the article
Please enter your email address



