Lijia DONG, Xiaojie GUO, Xue LI, Chaogui CHEN, Yang JIN, Alsaedi AHMED, Hayat TASAWAR, Qingzhou ZHAO, Guodong SHENG. Microscopic Insights into pH-dependent Adsorption of Cd(II) on Molybdenum Disulfide Nanosheets [J]. Journal of Inorganic Materials, 2020, 35(3): 293
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 35, Issue 3, 293 (2020)
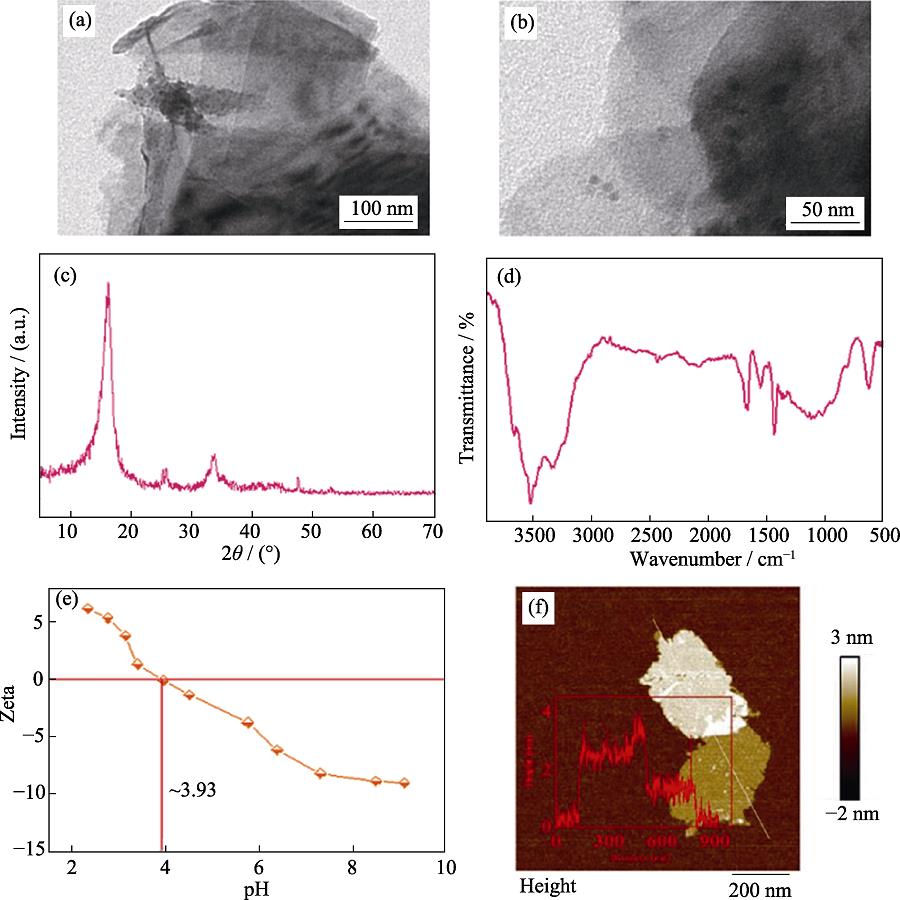
. SEM image (a), TEM image (b), XRD pattern (c), FT-IR spectrum (d), Zeta potentials (e), and height cross-section profile (inset) and corresponding AFM image (f) of MoS2 samples
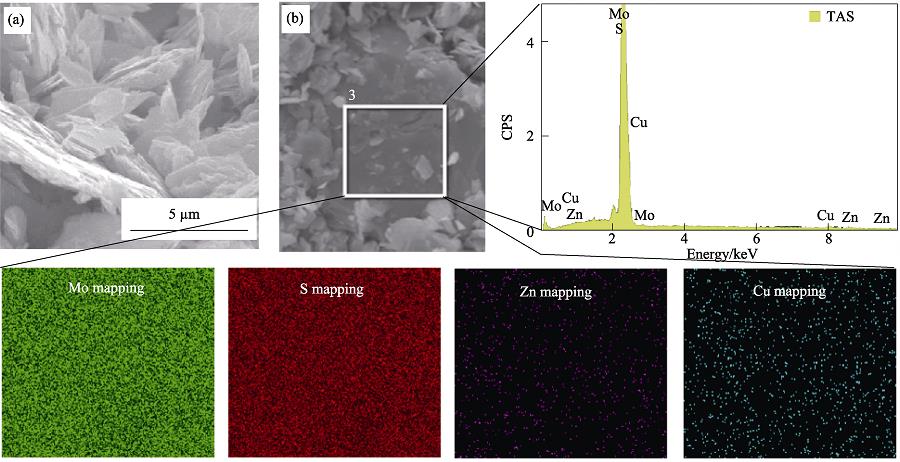
. SEM image (a), EDX spectra, and corresponding elemental maps (b) for MoS2 nanosheets
. Effects of pH and ionic strength on Cd(II) adsoprtion onto MoS2 nanosheets
. Cd(II) adsorption on MoS2 nanosheets as a function of contact time (a) and the fitting of pseudo-first-order kinetic model (b), pseudo-second-order kinetic model (c) and intra-particle diffusion model (d) at different pH
. Adsorption isotherms (a) and fitting results of Langmuir (b) and Freundlich (c) sorption isotherms of Cd(II) sorption on MoS2 at different temperatures and different pH
. Linear plots of lnK d versus Ce for Cd(II) at different temperatures and different pH(a), and linear regression plots of lnK θversus 1/T for Cd(II) sorption on MoS2 at different pH(b)
. Normalized, background-subtracted and k3-weighted EXAFS spectra (a) and corresponding RSF magnitudes and imaginary parts (b) of Cd reference samples
. EXAFS spectra (a) and the corresponding RSF magnitudes and imaginary parts (b) of Cd reference samples
. Schematic representation of possible sorption sites on the surface of MoS2
|
Table 1.
Parameters of kinetic models for the adsorption of Cd(II) on MoS2 as a function of pH
|
Table 1.
Parameters of Langmuir and Freundlich equations for the sorption of Cd(II) onto MoS2 at different temperatures and different pH
|
Table 2.
Parameters of thermodynamics for adsorption of Cd(II) onto MoS2 at 3 temperatures and different pH
|
Table 3.
Structural parameters of Cd(II) reference and sorption samples

Set citation alerts for the article
Please enter your email address



