Pengpeng LI, Bing WANG, Yingde WANG. Ultrafast CO Sensor Based on Flame-annealed Porous CeO2 Nanosheets for Environmental Application [J]. Journal of Inorganic Materials, 2021, 36(11): 1223
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 36, Issue 11, 1223 (2021)
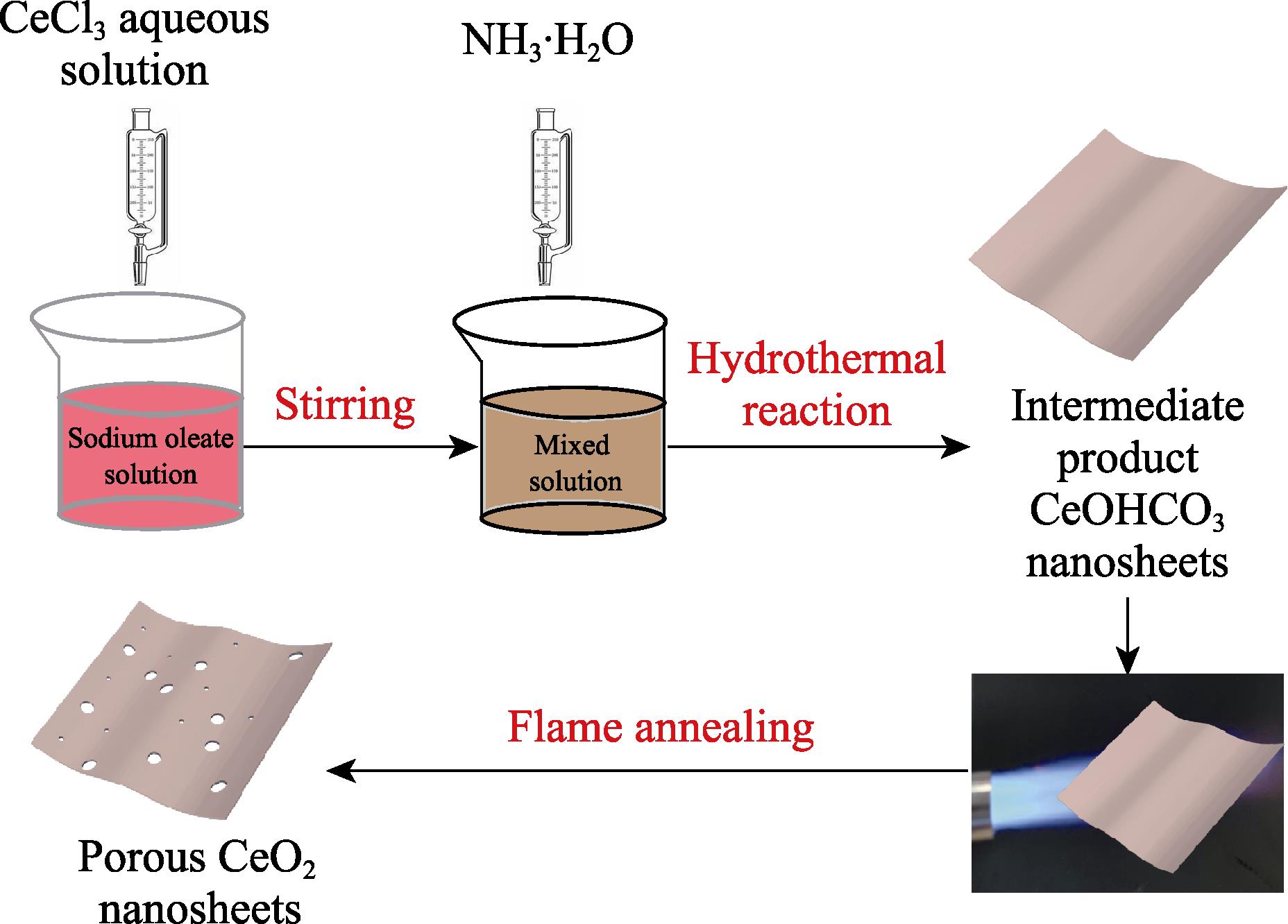
1. Schematic illustration of preparation of CeO2 nanosheets
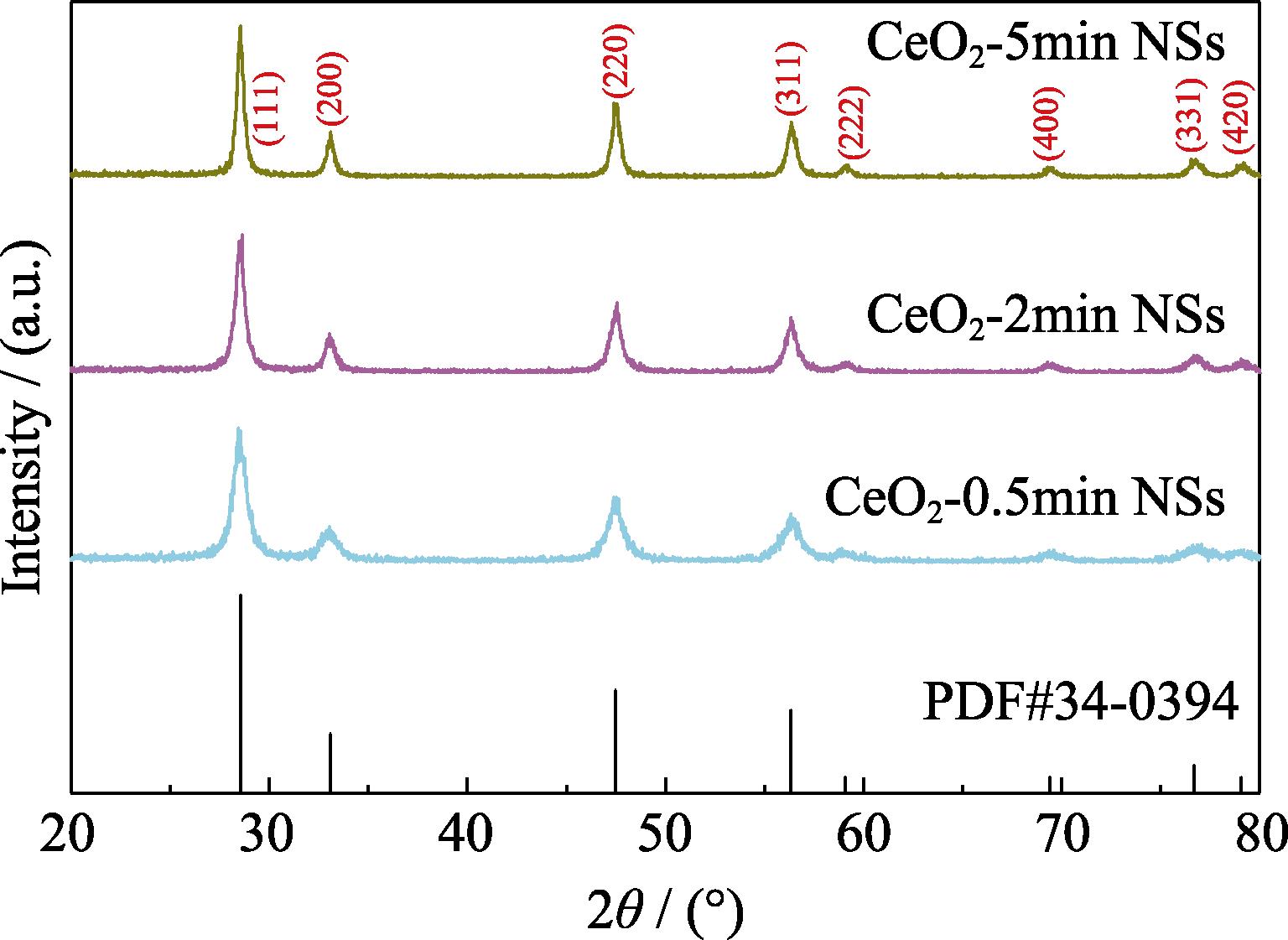
2. XRD patterns of the flame-annealed CeO2 nanosheets
3. XPS spectra of the CeO2 NSs
(a, b, c) high resolution spectra of O 1s of CeO2-0.5min NSs, CeO2-2min NSs and CeO2-5min NSs; (d, e, f) high resolution spectra of Ce 3d of CeO2-0.5min NSs, CeO2-2min NSs and CeO2-5min NSs
4. Morphologies and structure characterizations of the as-prepared samples(a, b, c) TEM images of CeO2-0.5min NSs, CeO2-2min NSs and CeO2-5min NSs (pores are labelled by red dotted circles); (d, e, f) HRTEM images of CeO2-0.5min NSs, CeO2-2min NSs and CeO2-5min NSs
5. (a) Nitrogen adsorption-desorption isotherms and (b) pore size distributions of CeO2 NSs
6. EPR spectra of CeO2 NSs
7. Gas sensing properties of CeO2 NSs
(a) Response of CeO2 NSs at different temperatures from 300 ℃ to 500 ℃ with an interval of 50 ℃ towards 500 μL/L CO; (b) Response of CeO2-2min NSs towards 500 μL/L CO at 450 ℃ for seven periods; (c) Transient response of CeO2-2min NSs towards different concentration from 10 μL/L to 10000 μL/L at 450 ℃; (d) Fitting of CO concentration and its corresponding response; (e) Determination of response/recovery time of CeO2-2min NSs towards 500 μL/L CO at 450 ℃; (f) Selectivity of CeO2-2min NSs towards CH4, H2, NH3, NO2 and CO at 450 ℃
8. Schematic illustration of CO gas sensing mechanism Yellow spheres represent O atoms; Orange spheres represent Ce atoms; Red rings represent the position of oxygen vacancies
S1. XRD pattern of the intermediate product
S2. Response of CeO2-2min NSs towards 500 μL/L CO at 450 ℃ under different humidities
S3. Long-term stability of CeO2-2min NSs towards 500 μL/L CO at 450 ℃ for two weeks
|
Table 1.
Percentage of each element species to the whole element content
|
Table 1.
Recent materials for in CO gas sensing
|
Table 2.
Summary of surface area and pore volume of CeO2 NSs

Set citation alerts for the article
Please enter your email address



