- Journal of Semiconductors
- Vol. 41, Issue 9, 091701 (2020)
Abstract
1. Introduction
Colloidal quantum dots (QDs) refer to the uniform small semiconductor particles, in which the charge carrier transportation is confined by three dimensions[
I–III–VI chalcogenides and their solid solutions have the advantages of low toxicity and continuously adjustable band gap covering the UV–vis to near infrared region which has attracted huge amount of attention as promising alternatives to the traditional cadmium- or lead-containing QDs[
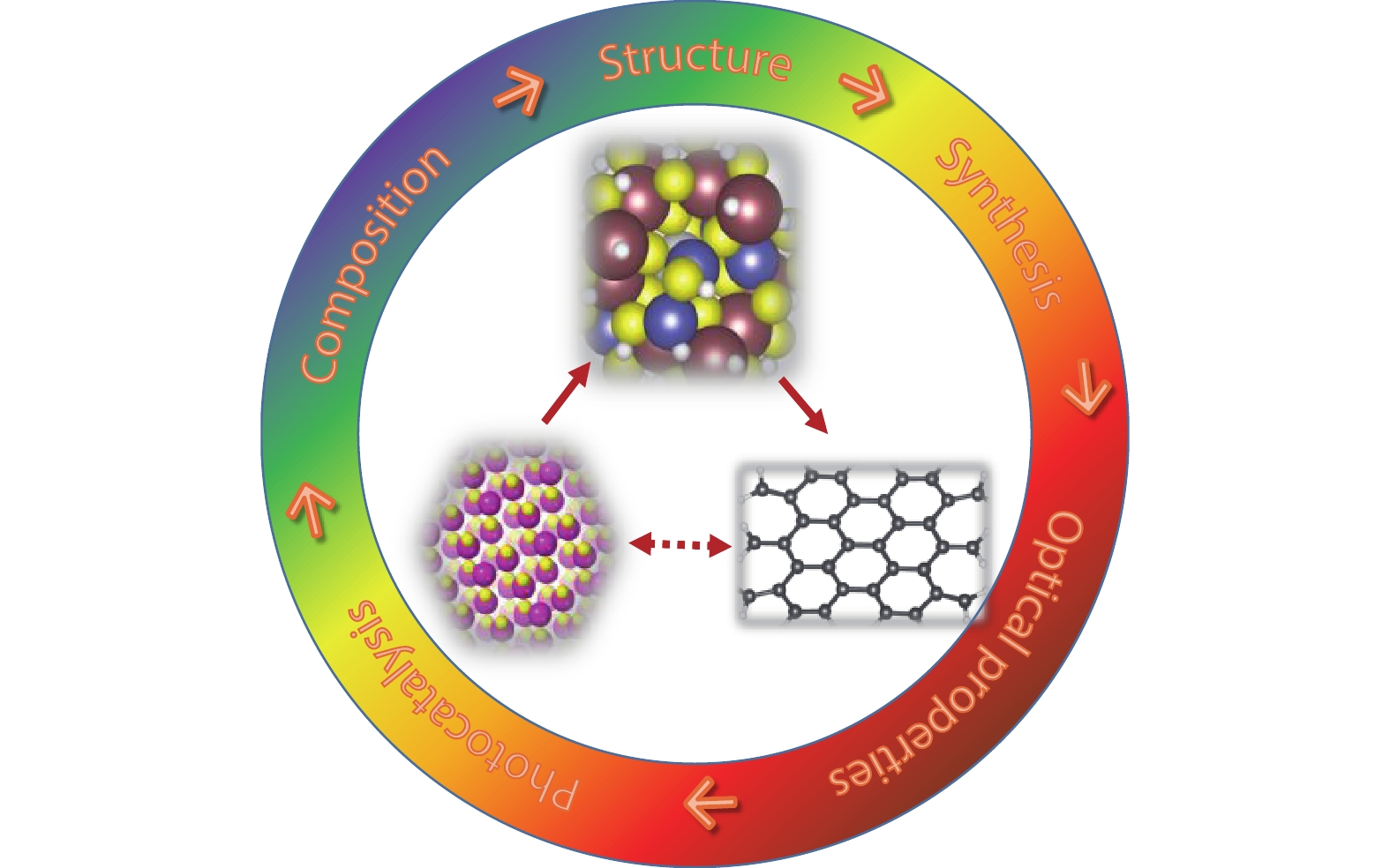
QDs have played a key role in many fields such as biology, devices and catalysis. Among these application, photocatalysis, as potentially the key solution to both environmental and energy problems, has been the most prominent and widely studied field not only for traditional QDs, but also for I–III–VI QDs and carbon dots[
![]()
Figure 1.(Color online) Schematic illustration for the bridging role of I–III–VI QDs between traditional II–VI QDs and emerging new carbon dots.
2. Structure
I–III–VI chalcogenides constitute a large class of semiconductor materials, whose band gap can be tuned not only by size and shape, but also by continuous composition manipulation[
The energy band structure of the multinary I−III−VI chalcogenide compounds can be adjusted by the alloy composition, thereby affecting the light absorption and reducing capability[
3. Synthesis
The preparation method and growth mechanism of ternary and multinary I–III–VI QDs are similar to traditional binary II–VI QDs, since the basic synthetic principles are similar. Currently, the hot injection method is still the main synthesis strategy of the complex I–III–VI QDs containing three or more elements. The metal precursors and chalcogen precursors can be formed in the same way capped with corresponding ligands. The main synthesis challenge of I–III–VI QDs is the reactivity balancing of multiple metal cations with strong capping ligands, which is a prerequisite for the formation of particles with uniform composition, phase and controllable structure (Fig. 2)[
![]()
Figure 2.(Color online) Schematic synthetic processes of I–III–VI QDs. Reprinted from Ref. [
Aqueous synthesis methods are receiving more and more attention because of the promising biological and catalytic applications of I–III–VI QDs. With mercaptoacetic acid, mercaptopropionic acid and other thiol-containing amines/alcohols as ligands, different types of I–III–VI QDs can be synthesized[
Semiconductor heterojunctions are critical in the bandgap engineering and wave function engineering of QDs. In the binary II–VI QD system, surface passivation by growing a larger band gap semiconductor shell, are used to decrease surface defects and improve PL quantum yield (QY)[
![]()
Figure 3.(Color online) Schematic alloying and selective cation exchange process of quaternary AgInS2–ZnS QDs. Reprinted from Ref. [
The declaration of particle growth mechanism played a crucial on the delicate control of QDs as well as the development of nanoscience and nanotechnology. In general, the related work of I–III–VI QDs preparation still cannot summarize out a clear nucleation and growth mechanism, especially the different aspects from that of II–VI QDs. It should be noted that although there have been a lot of reports on related research, the adjustment of the size distribution, surface structure and optical properties of I–III–VI QDs are still not comparable to that of II–VI QDs, and in-depth mechanism research is still lacking. This has brought a lot of challenges for the regulation of optoelectronic properties. Without compromising quality, mass production of ternary QDs remains a challenge, so optimized reactions that exceed laboratory scale is an urgent requirement. In order to achieve this goal, an in-depth understanding of chemical synthesis mechanism is necessary. From a basic scientific point of view, the precise structure and composition control is still challenging, which subsequently limits the manipulation and understanding of its optical properties. Comprehensive single-dot characterizations and spectroscopic measurements might be the key to clarify the correlation of optical performance and other physical parameters with structure. In addition, the synthesis and growth control research is carried out focusing on the ground state QDs, while the excited state mechanisms of these I–III–VI QDs are of great significance, especially their size, composition and surface/interface effects that are more critical in the design and preparation of photoelectric materials and clean energy applications (such as photovoltaics and photocatalysis).
4. Optical properties
4.1. Basic optical properties
There is no doubt that the I–III–VI QDs show the widest tunable optical properties due to changes in both size and composition. These materials also show special features, including longer excited state lifetimes, wider full width at half maximum (fwhm), larger Stokes shift, high quantum efficiency, along with simple and economical synthesis[
![]()
Figure 4.(Color online) (a) Theoretical calculated[
4.2. Mechanisms and defect states
The I–III–VI materials have photoelectric properties that are significantly different from traditional II–VI QDs, where the clarification of the excited state photophysical properties is crucial. The most important feature of I–III–VI QDs is their deep donor–acceptor pairs (DAPs, Fig. 5), which results in wide PL peaks and relatively large Stokes shifts. DAPs consist of different types of defects such as vacancies (VCu and VS), interstitial atoms (Cui) and substituting (CuIn), which are abundant in these ternary semiconductors due to the coexistence of multiple cations in the crystal lattice[
![]()
Figure 5.(Color online) Schematic defect states[
For chalcopyrite CuInS2 QDs, the main contributions of optical transitions are assigned to the electrons at VS and InCu, and holes at VCu[
5. Photocatalytic applications
5.1. General considerations
Band gap of I–III–VI QDs can be adjusted within a very wide range due to their composition manipulation capability, which represent the most promising visible-light-active photocatalysts for hydrogen production due to their excellent optoelectrical properties. Bulk I–III–VI sulfides were actually the first reported visible light photocatalysts with adjustable band gap used for photocatalytic water splitting[
5.2. Size and shape control
For multinary I–III–VI QDs, the small size of QDs and the narrow band gap of sulfides have important effects on the photocatalytic water splitting technology. As the size decreases, the band structure of QDs changes from continuous to discrete along with upshift of VB, resulting in the enhanced photoreduction capability, so reducing the size will usually facilitate photocatalytic H2 production. The reduction in particle size results in larger surface area and more active sites on the surface[
![]()
Figure 6.(Color online) Size- and composition-dependent photocatalytic properties of ZAIS QDs. Reprinted from Ref. [
5.3. Composition manipulation
Composition manipulation plays a critical role in I–III–VI QDs photocatalysts, not only for bang gap engineering, but also for catalytic activity. The band structure of the multinary compounds can be adjusted by composition, thereby affecting the light absorption ability and reduction ability to improve the photocatalytic performance (Fig. 6). Through theoretical calculation and analysis of energy band structure, it was found that Cu 3d or Ag 4d orbits play a critical role in the VB, while In 5s5p orbital constructs the CB of I–III–VI QDs[
5.4. Surface manipulation
The surface ligands not only determine the aqueous/organic dispersibility of the QDs, but may also serve as the surface catalytic center[
5.5. Heterojunctions
In heterojunctions formed by combining semiconductor materials with different band gaps and energy levels, especially type-II heterojunctions, photogenerated electrons can transfer from high CB materials to relatively low CB materials, while photogenerated holes can migrate reversibly. This band arrangement thereby achieves efficient separation of electrons and holes. CuIn5S8/Ag2S, CuInSe2/TiO2[
5.6. Cocatalysts
Various cocatalysts, especially noble metals, are widely used as high-efficiency promoters in water splitting systems to enhance charge separation and catalytic activity. The Fermi level of the noble metals are often lower than that of the semiconductors and the electrons generated by the semiconductor can smoothly migrate to the active site of the precious metal to reduce the surface adsorbed H+ to H2[
5.7. Stability and hole scarification
Photocorrosion caused by the oxidative holes is a problem faced by most narrow-band-gap sulfide photocatalysts[
6. Bridging between QDs and emerging carbon dots
As mentioned above, the QDs family has shown amazing developing vitality, and the connotation is also constantly expanding, bringing a series of new phenomena, new principles and new challenges to the QDs field. Now, as it expands from traditional II–VI semiconductors to carbon and other emerging materials, there is a huge gap in the composition and structure, leading to completely different synthetic chemistry, PL mechanisms and applications[
7. Conclusions
In summary, I–III–VI QDs own lots of unique structural and optical properties and play a key role in photoelectric fields, including photocatalysis. It has similar advantages and characteristics of traditional QDs, such as the size-dependent quantum confinement effect and high specific surface area. On one hand, traditional QDs have developed lots of useful strategies to improve light harvesting and charge separation, such as the delicate control over size, shape, surface exposure and heterostructures. On the other hand, these ternary or multinary I–III–VI QDs provide several characteristic advantages that traditional binary QDs do not have, especially the wide range regulation of composition and band gap, as well as the rich long-lived trap states, which greatly expands the design of solid solution QDs and composite photocatalysts with complex compositions and structures. However, the complicated composition also brought a series of challenges on the structure and synthesis of these QDs, such as the precise control of the composition, the balancing of cation reactivity, the unwanted cation exchange and diffusion for heterojunction construction. This may rely on the in-situ monitoring of the growing process and the deep understanding of the synthetic chemistry. Subsequently, more efforts are needed on the clarification of size- vs. composition-dependent band gap, the PL origin from the abundant trap states, the engineering and utilization of the long-lived charge carriers, which requires investigation and manipulation of the excited states by ultrafast spectroscopy. As mentioned above, all structure controled methods of traditional QDs photocatalysts have been applied to the I–III–VI QDs system, including particle size, composition, surface ligands, cocatalysts, hole sacrificial agents, etc., which provide systematic research on related strategies as very good model system. In addition, the performance shortcomings of the traditional chalcogenide photocatalysts usually can also be found in the I–III–VI QDs system, such as poor stability, slow hole extraction, low charge carrier utilization efficiency, etc. These problems rely on ultrafast spectroscopic research, which also provides a good inspiration for other photocatalytic systems. In principle, photocatalysis is a special form of electrocatalysis, where the electrons and holes are provided by light irradiation. The advantages of I–III–VI QDs in mechanism research can also be extended to the entire photo/electrocatalysis fields Furthermore, as a natural multi-composition and multi-interface catalyst, I–III–VI QDs can be useful in the distribution process of active species, the adsorption of active species, and electron transfer in other catalytic systems in a much broader way. The identification and change of the active sites and other issues may provide useful help as a suitable model system for understanding the catalyst from the perspective of physical and chemical interface engineering, for which more profound ultrafast and in situ spectroscopy studies are the key. In terms of the overall significance of photocatalysis and other catalytic studies, I–III–VI QDs are a class of materials that far surpasses traditional binary QDs. With the joint efforts of researchers in related fields, we hope that they will play an increasingly important role in catalysis research.
Acknowledgements
This work is supported by the National Natural Science Foundation of China (21908081, 21501072, 51972216, 51725204, 21771132 and 52041202), the National MCF Energy R&D Program (2018YFE0306105), Innovative Research Group Project of the National Natural Science Foundation of China (51821002), the Jiangsu Specially-Appointed Professors Program, and the Natural Science Foundation of Jiangsu Province (BK20190041, BK20190828 and BK20150489).
References
[1] S Q Li, X S Tang, Z G Zang et al. I–III–VI chalcogenide semiconductor nanocrystals: Synthesis, properties, and applications. Chin J Catal, 39, 590(2018).
[2] Y H Kuo, Y K Lee, Y Ge et al. Strong quantum-confined Stark effect in germanium quantum-well structures on silicon. Nature, 437, 1334(2005).
[3] E L Tae, K E Lee, J S Jeong et al. Synthesis of diamond-shape titanate molecular sheets with different sizes and realization of quantum confinement effect during dimensionality reduction from two to zero. J Am Chem Soc, 130, 6534(2008).
[4] Y Cao, J Guo, R Shi et al. Evolution of thiolate-stabilized Ag nanoclusters from Ag-thiolate cluster intermediates. Nat Commun, 9, 2379(2018).
[5] H T Ma, L J Pan, J Wang et al. Synthesis of AgInS2 QDs in droplet microreactors: Online fluorescence regulating through temperature control. Chin Chem Lett, 30, 79(2019).
[6] O Stroyuk, A Raevskaya, F Spranger et al. Origin and dynamics of highly efficient broadband photoluminescence of aqueous glutathione-capped size-selected Ag –In –S quantum dots. J Phys Chem C, 122, 13648(2018).
[7] O S Oluwafemi, B M M May, S Parani et al. Facile, large scale synthesis of water soluble AgInSe2/ZnSe quantum dots and its cell viability assessment on different cell lines. Mat Sci Eng C, 106, 110181(2020).
[8] X S Zhu, V G Demillo, S Q Chen et al. Development of non-cadmium I–III–VI quantum dots and their surface modification for biomedical applications. Mater Sci Forum, 915, 163(2018).
[9] D Kalinowska, M Drozd, I Grabowska-Jadach et al. The influence of selected omega-mercaptocarboxylate ligands on physicochemical properties and biological activity of Cd-free, zinccopperindium sulfide colloidal nanocrystals. Mater Sci Eng C, 97, 583(2019).
[10] B Chen, N Pradhan, H Zhong. From large-scale synthesis to lighting device applications of ternary I–III–VI semiconductor nanocrystals: Inspiring greener material emitters. J Phys Chem Lett, 9, 435(2018).
[11] S Y Yoon, J H Kim, K H Kim et al. High-efficiency blue and white electroluminescent devices based on non-Cd I−III−VI quantum dots. Nano Energy, 63, 103869(2019).
[12] W Rui, T Xin, A I Channa et al. Environment-friendly Mn-alloyed core/shell quantum dots for high-efficiency photoelectrochemical cells. J Mater Chem A, 8, 10736(2020).
[13] M Jalalah, M S Al-Assiri, J G Park. One-pot gram-scale, eco-friendly, and cost-effective synthesis of CuGaS2/ZnS nanocrystals as efficient UV-harvesting down-converter for photovoltaics. Adv Energy Mater, 8, 1703418(2018).
[14] O Yarema, M Yarema, V Wood. Tuning the composition of multicomponent semiconductor nanocrystals: The case of I–III–VI materials. Chem Mater, 30, 1446(2018).
[15] L L Tan, Y H Liu, B D Mao et al. Effective bandgap narrowing of Cu–In–Zn–S quantum dots for photocatalytic H2 production via cocatalyst-alleviated charge recombination. Inorg Chem Front, 5, 258(2018).
[16] J Feng, X S Yang, G W Feng et al. The experimental determination of composition-dependent molar extinction coefficient of nonstoichiometric Zn–Cu–In–S nanocrystals. Mater Res Express, 5, 075030(2018).
[17] J P Nong, G L Lan, W F Jin et al. Eco-friendly and high-performance photoelectrochemical anode based on AgInS2 quantum dots embedded in 3D graphene nanowalls. J Mater Chem C, 7, 9830(2019).
[18] B J Stanbery. Copper indium selenides and related materials for photovoltaic devices. Crit Rev Solid State, 27, 73(2002).
[19] T Feurer, P Reinhard, E Avancini et al. Progress in thin film CIGS photovoltaics - Research and development, manufacturing, and applications. Prog Photovolt, 25, 645(2017).
[20] T Kameyama, M Kishi, C Miyamae et al. Wavelength-tunable band-edge photoluminescence of nonstoichiometric Ag –In –S nanoparticles via Ga3+ Doping. ACS Appl Mater Inter, 10, 42844(2018).
[21] D Huang, J W Jiang, J Guo et al. General rules of the sub-band gaps in group-IV (Si, Ge, and Sn)-doped I–III–VI2-type chalcopyrite compounds for intermediate band solar cell: A first-principles study. Mater Sci Eng B, 236–237, 147(2018).
[22] M M Chen, H G Xue, S P Guo. Multinary metal chalcogenides with tetrahedral structures for second-order nonlinear optical, photocatalytic, and photovoltaic applications. Coordin Chem Rev, 368, 115(2018).
[23] Y H Liu, H Huang, W J Cao et al. Advances in carbon dots: from the perspective of traditional quantum dots. Mater Chem Front, 4, 1586(2020).
[24] W M Girma, M Z Fahmi, A Permadi et al. Synthetic strategies and biomedical applications of I–III–VI ternary quantum dots. J Mater Chem B, 5, 6193(2017).
[25] T Omata, K Nose, S Otsuka-Yao-Matsuo. Size dependent optical band gap of ternary I–III–VI2 semiconductor nanocrystals. J Appl Phys, 105, 073106(2009).
[26] D Aldakov, A Lefrancois, P Reiss. Ternary and quaternary metal chalcogenide nanocrystals: synthesis, properties and applications. J Mater Chem C, 1, 3756(2013).
[27] S H Wei, L G Ferreira, A Zunger. First-principles calculation of the order-disorder transition in chalcopyrite semiconductors. Phys Rev B, 45, 2533(1992).
[28] M Sandroni, K D Wegner, D Aldakov et al. Prospects of chalcopyrite-type nanocrystals for energy applications. Acs Energy Lett, 2, 1076(2017).
[29] H Nakamura, W Kato, M Uehara et al. Tunable photoluminescence wavelength of chalcopyrite CuInS2-based semiconductor nanocrystals synthesized in a colloidal system. Chem Mater, 18, 3330(2006).
[30] P J Whitham, A Marchioro, K E Knowles et al. Single-particle photoluminescence spectra, blinking, and delayed luminescence of colloidal CuInS2 nanocrystals. J Phys Chem C, 120, 17136(2016).
[31] K Takarabe, K Kawai, S Minomura et al. Electronic structure of some I–III–VI2 chalcopyrite semiconductors studied by synchrotron radiation. J Appl Phys, 71, 441(1992).
[32] L L Zhang, Z X Pan, W Wang et al. Copper deficient Zn–Cu–In–Se quantum dot sensitized solar cells for high efficiency. J Mater Chem A, 5, 21442(2017).
[33] A Bouich, B Hartiti, S Ullah et al. Experimental, theoretical, and numerical simulation of the performance of CuIn
[34] M S Pradeepkumar, A S Pal, A Singh et al. Phase separation in wurtzite CuIn
[35] P Ilaiyaraja, P S Mocherla, T K Srinivasan et al. Synthesis of Cu-deficient and Zn-graded Cu–In–Zn–S quantum dots and hybrid inorganic-organic nanophosphor composite for white light emission. ACS Appl Mater Interfaces, 8, 12456(2016).
[36] P Bujak, Z Wrobel, M Penkala et al. Highly luminescent Ag−In−Zn−S quaternary nanocrystals: Growth mechanism and surface chemistry elucidation. Inorg Chem, 58, 1358(2019).
[37] N Tsolekile, S Parani, M C Matoetoe et al. Evolution of ternary I–III–VI QDs: Synthesis, characterization and application. Nano-Struct Nano-Objects, 12, 46(2017).
[38] R L Zhang, P Yang, Y Q Wang. Facile synthesis of CuInS2/ZnS quantum dots with highly near-infrared photoluminescence via phosphor-free process. J Nanopart Res, 15, 1910(2013).
[39] Z Y Guan, A W Tang, P W Lv et al. New insights into the formation and color-tunable optical properties of multinary Cu –In –Zn-based chalcogenide semiconductor nanocrystals. Adv Optical Mater, 6, 1701389(2018).
[40] M X Jiao, Y Li, Y X Jia et al. Aqueously synthesized color-tunable quaternary Cu–In–Zn–S quantum dots for Cu(II) detection via mild and rapid cation exchange. Sens Actuators B, 294, 32(2019).
[41] L Yang, A Antanovich, A Prudnikau et al. Highly luminescent Zn –Cu –In –S/ZnS core/gradient shell quantum dots prepared from indium sulfide by cation exchange for cell labeling and polymer composites. Nanotechnology, 30, 395603(2019).
[42] S T Connor, C M Hsu, B D Weil et al. Phase transformation of biphasic Cu2S–CuInS2 to monophasic CuInS2 nanorods. J Am Chem Soc, 131, 4962(2009).
[43] T Torimoto, T Adachi, K Okazaki et al. Facile synthesis of ZnS–AgInS2 solid solution nanoparticles for a color-adjustable luminophore. J Am Chem Soc, 129, 12388(2007).
[44] Y Y Chen, Q Wang, T Y Zha et al. Green and facile synthesis of high-quality water-soluble Ag–In–S/ZnS core/shell quantum dots with obvious bandgap and sub-bandgap excitations. J Alloy Compd, 753, 364(2018).
[45] M Masab, H Muhammad, F Shah et al. Facile synthesis of CdZnS QDs: Effects of different capping agents on the photoluminescence properties. Mat Sci Semicond Proc, 81, 113(2018).
[46] M Mrad, T Ben Chaabane, H Rinnert et al. Aqueous synthesis for highly emissive 3-mercaptopropionic acid-capped AIZS quantum dots. Inorg Chem, 59, 6220(2020).
[47] B D Mao, B Wang, F R Yu et al. Hierarchical MoS2 nanoflowers on carbon cloth as an efficient cathode electrode for hydrogen evolution under all pH values. Int J Hydrogen Energ, 43, 11038(2018).
[48] Z Moradi Alvand, H R Rajabi, A Mirzaei et al. Rapid and green synthesis of cadmium telluride quantum dots with low toxicity based on a plant-mediated approach after microwave and ultrasonic assisted extraction: Synthesis, characterization, biological potentials and comparison study. Mater Sci Eng C, 98, 535(2019).
[49] X B Hu, T Chen, Y Q Xu et al. Hydrothermal synthesis of bright and stable AgInS2 quantum dots with tunable visible emission. J Lumin, 200, 189(2018).
[50] T Chen, X B Hu, Y Q Xu et al. Hydrothermal synthesis of highly fluorescent Ag –In –S/ZnS core/shell quantum dots for white light-emitting diodes. J Alloy Compd, 804, 119(2019).
[51] A Das, P T Snee. Synthetic developments of nontoxic quantum dots. Chemphyschem, 17, 598(2016).
[52] H Li, Z H Chen, L Zhao et al. Synthesis of TiO2@ZnIn2S4 hollow nanospheres with enhanced photocatalytic hydrogen evolution. Rare Met, 38, 420(2019).
[53] X J Kang, Y C Yang, L J Huang et al. Large-scale synthesis of water-soluble CuInSe2/ZnS and AgInSe2/ZnS core/shell quantum dots. Green Chem, 17, 4482(2015).
[54] W J Cao, Y L Qin, H Huang et al. Extraction of high-quality quantum dot photocatalysts via combination of size selection and electrochemiluminescence. Acs Sustain Chem Eng, 7, 20043(2019).
[55] M D Tessier, E A Baquero, D Dupont et al. Interfacial oxidation and photoluminescence of InP-based core/shell quantum dots. Chem Mater, 30, 6877(2018).
[56] Y M Li, J Liu, X D Wan et al. Surface passivation enabled-structural engineering of I–III–VI2 nanocrystal photocatalysts. J Mater Chem A, 8, 9951(2020).
[57] B M Saidzhonov, V F Kozlovsky, V B Zaytsev et al. Ultrathin CdSe/CdS and CdSe/ZnS core-shell nanoplatelets: The impact of the shell material on the structure and optical properties. J Lumin, 209, 170(2019).
[58] K Yu, Y Yang, J Wang et al. Ultrafast carrier dynamics and third-order nonlinear optical properties of AgInS2/ZnS nanocrystals. Nanotechnology, 29, 255703(2018).
[59] M Michalska, A Aboulaich, G Medjahdi et al. Amine ligands control of the optical properties and the shape of thermally grown core/shell CuInS2/ZnS quantum dots. J Alloy Compd, 645, 184(2015).
[60] B D Mao, C H Chuang, F Lu et al. Study of the partial Ag-to-Zn cation exchange in AgInS2/ZnS nanocrystals. J Phys Chem C, 117, 648(2012).
[61] J H Kim, K H Kim, S Y Yoon et al. Tunable emission of bluish Zn–Cu–Ga–S quantum dots by Mn doping and their electroluminescence. ACS Appl Mater Inter, 11, 8250(2019).
[62] S Paderick, M Kessler, T J Hurlburt et al. Synthesis and characterization of AgGaS2 nanoparticles: A study of growth and fluorescence. Chem Commun, 54, 62(2017).
[63] K W Zhang, Y H Liu, B Wang et al. Three-dimensional interconnected MoS2 nanosheets on industrial 316 L stainless steel mesh as an efficient hydrogen evolution electrode. Int J Hydrogen Energ, 44, 1555(2019).
[64] T Omata, K Nose, K Kurimoto et al. Electronic transition responsible for size-dependent photoluminescence of colloidal CuInS2 quantum dots. J Mater Chem C, 2, 6867(2014).
[65] B D Mao, C H Chuang, J W Wang et al. Synthesis and photophysical properties of ternary I–III–VI AgInS2 nanocrystals: Intrinsic versus surface states. J Phys Chem C, 115, 8945(2011).
[66] S B Zhang, S H Wei, A Zunger. Defect physics of the CuInSe2 chalcopyrite semiconductor. Phys Rev B, 57, 9642(1988).
[67] T Debnath, H N Ghosh. Ternary metal chalcogenides: Into the exciton and biexciton dynamics. J Phys Chem Lett, 10, 6227(2019).
[68] A D Leach, J E Macdonald. Optoelectronic properties of CuInS2 nanocrystals and their origin. J Phys Chem Lett, 7, 572(2016).
[69] W Y Liu, Y Zhang, J Zhao et al. Photoluminescence of indium-rich copper indium sulfide quantum dots. J Lumin, 162, 191(2015).
[70] A Fuhr, H J Yun, S A Crooker et al. Spectroscopic and magneto-optical signatures of Cu1+ and Cu2+ defects in copper indium sulfide quantum dots. ACS Nano, 14, 2212(2020).
[71] K E Knowles, K H Hartstein, T B Kilburn et al. Luminescent colloidal semiconductor nanocrystals containing copper: Synthesis, photophysics, and applications. Chem Rev, 116, 10820(2016).
[72] H Z Zhong, S S Lo, T Mirkovic et al. Noninjection gram-scale synthesis of monodisperse pyramidal CuInS2 nanocrystals and their size-dependent properties. ACS Nano, 4, 5253(2010).
[73] S N Rashkeev, W R L Lambrecht. Second-harmonic generation of I–III–VI2 chalcopyrite semiconductors: Effects of chemical substitutions. Phys Rev B, 63, 165212(2001).
[74] G Gong, Y H Liu, B D Mao et al. Ag doping of Zn–In–S quantum dots for photocatalytic hydrogen evolution: Simultaneous bandgap narrowing and carrier lifetime elongation. Appl Catal B, 216, 11(2017).
[75] Y Li, J Z Zhang. Hydrogen generation from photoelectrochemical water splitting based on nanomaterials. Laser Photonics Rev, 4, 517(2009).
[76] L Jing, W Zhou, G Tian et al. Surface tuning for oxide-based nanomaterials as efficient photocatalysts. Chem Soc Rev, 42, 9509(2013).
[77] Y Ye, Z G Zang, T W Zhou et al. Theoretical and experimental investigation of highly photocatalytic performance of CuInZnS nanoporous structure for removing the NO gas. J Catal, 357, 100(2018).
[78] X L Wang, M T Swihart. Controlling the size, shape, phase, band gap, and localized surface plasmon resonance of Cu2–
[79] O Yarema, M Yarema, A Moser et al. Composition- and size-controlled I–V–VI Semiconductor nanocrystals. Chem Mater, 32, 2078(2020).
[80] B D Mao, C H Chuang, C Mccleese et al. Near-infrared emitting AgInS2/ZnS nanocrystals. J Phys Chem C, 118, 13883(2014).
[81] O Yarema, M Yarema, D Bozyigit et al. Independent composition and size control for highly luminescent indium-rich silver indium selenide nanocrystals. ACS Nano, 9, 11134(2015).
[82] O Yarema, M Yarema, W M Lin et al. Cu–In–Te and Ag–In–Te colloidal nanocrystals with tunable composition and size. Chem Commun, 52, 10878(2016).
[83] C Liu, X Li, J Li et al. Carbon dots modifying sphere-flower CdIn2S4 on N-rGO sheet muti-dimensional photocatalyst for efficient visible degradation of 2,4-dichlorophenol. J Taiwan Inst Chem E, 99, 142(2019).
[84] D X Zhu, H H Ye, Z M Liu. Seed-mediated growth of heterostructured Cu1.94S–MS (M = Zn, Cd, Mn) and alloyed CuNS2 (N = In, Ga) nanocrystals for use in structure- and composition-dependent photocatalytic hydrogen evolution. Nanoscale, 12, 6111(2020).
[85] D K Sharma, S Hirata, L Bujak et al. Influence of Zn on the photoluminescence of colloidal (AgIn)
[86] M T Ng, C B Boothroyd, J J Vittal. One-pot synthesis of new-phase AgInSe2 nanorods. J Am Chem Soc, 128, 7118(2006).
[87] Z Liu, J Liu, Y Huang et al. From one-dimensional to two-dimensional wurtzite CuGaS2 nanocrystals: non-injection synthesis and photocatalytic evolution. Nanoscale, 11, 158(2018).
[88] Z M Liu, A W Tang, J Liu et al. Non-injection synthesis of L-shaped wurtzite Cu–Ga–Zn–S alloyed nanorods and their advantageous application in photocatalytic hydrogen evolution. J Mater Chem A, 6, 18649(2018).
[89] M D Regulacio, M Y Han. Multinary I–III–VI2 and I2–II–IV–VI4 semiconductor nanostructures for photocatalytic applications. Acc Chem Res, 49, 511(2016).
[90] D Chen, J H Ye. Photocatalytic H2 evolution under visible light irradiation on AgIn5S8 photocatalyst. J Phys Chem Solids, 68, 2317(2007).
[91] W Minoshima, R Ito, T Takiyama et al. Photoluminescence characterization of ZnS–AgInS2 (ZAIS) nanoparticles adsorbed on plasmonic chip studied with fluorescence microscopy. J Photoch Photobio A, 367, 347(2018).
[92] P S Vasekar, N G Dhere. Effect of sodium addition on Cu-deficient CuIn1−
[93] T A Kandiel, D H Anjum, K Takanabe. Nano-sized quaternary CuGa2In3S8 as an efficient photocatalyst for solar hydrogen production. ChemSusChem, 7, 3112(2014).
[94] X L Yu, X Q An, A Shavel et al. The effect of the Ga content on the photocatalytic hydrogen evolution of CuIn1–
[95] T A Kandiel, G A M Hutton, E Reisner. Visible light driven hydrogen evolution with a noble metal free CuGa2In3S8 nanoparticle system in water. Catal Sci Technol, 6, 6536(2016).
[96] A Hirase, Y Hamanaka, T Kuzuya. Ligand-induced luminescence transformation in AgInS2 nanoparticles: From defect emission to band-edge emission. J Phys Chem Lett, 11, 3969(2020).
[97] Y Peng, L Shang, Y Cao et al. Effects of surfactants on visible-light-driven photocatalytic hydrogen evolution activities of AgInZn7S9 nanorods. Appl Surf Sci, 358, 485(2015).
[98] S Jung, J H Cha, D Y Jung. Synthesis of oleic acid-capped CuInS2 nanocrystals from bimetallic hydroxide precursor. Thin Solid Films, 603, 243(2016).
[99] S Yu, X B Fan, X Wang et al. Efficient photocatalytic hydrogen evolution with ligand engineered all-inorganic InP and InP/ZnS colloidal quantum dots. Nat Commun, 9, 4009(2018).
[100] Y L Yang, Y H Liu, B D Mao et al. Facile surface engineering of Ag–In–Zn–S quantum dot photocatalysts by mixed-ligand passivation with improved charge carrier lifetime. Catal Lett, 149, 1800(2019).
[101] M Jagadeeswararao, S Dey, A Nag et al. Visible light-induced hydrogen generation using colloidal (ZnS)0.4(AgInS2)0.6 nanocrystals capped by S2− ions. J Mater Chem A, 3, 8276(2015).
[102] A S Kshirsagar, A Gautam, P K Khanna. Efficient photo-catalytic oxidative degradation of organic dyes using CuInSe2/TiO2 hybrid hetero-nanostructures. J Photoch Photobio A, 349, 73(2017).
[103] K Li, B Chai, T Y Peng et al. Preparation of AgIn5S8/TiO2 heterojunction nanocomposite and its enhanced photocatalytic H2 production property under visible light. Acs Catal, 3, 170(2013).
[104] S M Kobosko, D H Jara, P V Kamat. AgInS2–ZnS quantum dots: Excited state interactions with TiO2 and photovoltaic performance. ACS Appl Mater Inter, 9, 33379(2017).
[105] Y L Yang, B D Mao, G Gong et al. In-situ growth of Zn–AgIn5S8 quantum dots on g-C3N4 towards 0D/2D heterostructured photocatalysts with enhanced hydrogen production. Int J Hydrogen Energy, 44, 15882(2019).
[106] V Subramanian, E E Wolf, P V Kamat. Catalysis with TiO2/gold nanocomposites. Effect of metal particle size on the Fermi level equilibration. J Am Chem Soc, 126, 4943(2004).
[107] M Lan, R M Guo, Y B Dou et al. Fabrication of porous Pt-doping heterojunctions by using bimetallic MOF template for photocatalytic hydrogen generation. Nano Energy, 33, 238(2017).
[108] G Iervolino, V Vaiano, D Sannino et al. Enhanced photocatalytic hydrogen production from glucose aqueous matrices on Ru-doped LaFeO3. Appl Catal B, 207, 182(2017).
[109] F Xue, C Chen, W L Fu et al. Interfacial and dimensional effects of Pd Co-catalyst for efficient photocatalytic hydrogen generation. J Phys Chem C, 122, 25165(2018).
[110] J Q Huang, G J Li, Z F Zhou et al. Efficient photocatalytic hydrogen production over Rh and Nb codoped TiO2 nanorods. Chem Eng J, 337, 282(2018).
[111] G Gong, Y H Liu, B D Mao et al. Mechanism study on the photocatalytic efficiency enhancement of MoS2 modified Zn–AgIn5S8 quantum dots. Rsc Adv, 6, 99023(2016).
[112] T Huang, W Chen, T Y Liu et al. Hybrid of AgInZnS and MoS2 as efficient visible-light driven photocatalyst for hydrogen production. Int J Hydrogen Energ, 42, 12254(2017).
[113] W L Zhen, X F Ning, B J Yang et al. The enhancement of CdS photocatalytic activity for water splitting via anti-photocorrosion by coating Ni2P shell and removing nascent formed oxygen with artificial gill. Appl Catal B, 221, 243(2018).
[114] V M Daskalaki, M Antoniadou, G L Puma et al. Solar light-responsive Pt/CdS/TiO2 photocatalysts for hydrogen production and simultaneous degradation of inorganic or organic sacrificial agents in wastewater. Energ Environ Sci, 44, 7200(2010).
[115] M J Wang, S L Shen, L Li et al. Effects of sacrificial reagents on photocatalytic hydrogen evolution over different photocatalysts. J Mater Sci, 52, 5155(2017).
[116] J Zhang, J W Chen, Y Luo et al. Sandwich-like electrode with tungsten nitride nanosheets decorated with carbon dots as efficient electrocatalyst for oxygen reduction. Appl Surf Sci, 466, 911(2019).
[117] P Ding, J Di, X Chen et al. S, N codoped graphene quantum dots embedded in (BiO)2CO3: Incorporating enzymatic-like catalysis in photocatalysis. Acs Sustain Chem Eng, 6, 10229(2018).
[118] M Ji, Y Liu, J Di et al. N-CQDs accelerating surface charge transfer of Bi4O5I2 hollow nanotubes with broad spectrum photocatalytic activity. Appl Catal B, 237, 1033(2018).
[119] X Miao, X Yue, Z Ji et al. Nitrogen-doped carbon dots decorated on g-C3N4/Ag3PO4 photocatalyst with improved visible light photocatalytic activity and mechanism insight. Appl Catal B, 227, 459(2018).
[120] Y Q Zhou, E M Zahran, B A Quiroga et al. Size-dependent photocatalytic activity of carbon dots with surface-state determined photoluminescence. Appl Catal B, 248, 157(2019).
[121] A Vaneski, J Schneider, A S Susha et al. Colloidal hybrid heterostructures based on II–VI semiconductor nanocrystals for photocatalytic hydrogen generation. J Photoch Photobio C, 19, 52(2014).
[122] A J Houtepen, Z Hens, J S Owen et al. On the origin of surface traps in colloidal II–VI semiconductor nanocrystals. Chem Mater, 29, 752(2017).
[123] K L Ou, J C Fan, J K Chen et al. Hot-injection synthesis of monodispersed Cu2ZnSn(S
[124] H Ren, M Wang, Z Li et al. Synthesis and characterization of CuZnSe2 nanocrystals in wurtzite, zinc blende, and core–shell polytypes. Chem Mater, 31, 10085(2019).
[125] W Liu, C Li, X Sun et al. Highly crystalline carbon dots from fresh tomato: UV emission and quantum confinement. Nanotechnology, 28, 485705(2017).
[126] P Wu, X Y Wu, W Li et al. Ultra-small amorphous carbon dots: preparation, photoluminescence properties, and their application as TiO2 photosensitizers. J Mater Sci, 54, 5280(2018).
[127] P P Ingole. A consolidated account of electrochemical determination of band structure parameters in II–VI semiconductor quantum dots: a tutorial review. Phys Chem Chem Phys, 21, 4695(2019).
[128] X Miao, D Qu, D Yang et al. Synthesis of carbon dots with multiple color emission by controlled graphitization and surface functionalization. Adv Mater, 30, 1704740(2018).
[129] S Hu, A Trinchi, P Atkin et al. Tunable photoluminescence across the entire visible spectrum from carbon dots excited by white light. Angew Chem Int Ed Engl, 54, 2970(2015).
[130] H Qi, S J Wang, X H Jiang et al. Research progress and challenges of blue light-emitting diodes based on II–VI semiconductor quantum dots. J Mater Chem C(2020).
[131] J Du, R Singh, I Fedin et al. Spectroscopic insights into high defect tolerance of Zn:CuInSe2 quantum-dot-sensitized solar cells. Nat Energy, 5, 409(2020).
[132] X Y Xu, Z J Bao, W S Tang et al. Surface states engineering carbon dots as multi-band light active sensitizers for ZnO nanowire array photoanode to boost solar water splitting. Carbon, 121, 201(2017).
[133] F Yan, Z Sun, H Zhang et al. The fluorescence mechanism of carbon dots, and methods for tuning their emission color: A review. Mikrochim Acta, 186, 583(2019).
[134] Y S Lee, H B Bu, T Taniguchi et al. Hydrothermal synthesis of NAC-capped II–VI semiconductor ZnSe quantum dots in acidic condition. Chem Lett, 45, 878(2016).
[135] M Ghali, A Rezk, A M Eissa et al. Hot-injection synthesis of ultrasmall CuIn3Se5 quantum dots and production of ink-coated films. J Phys Chem Solids, 146, 109610(2020).
[136] L Shang, C Zhou, T Bian et al. Facile synthesis of hierarchical ZnIn2S4 submicrospheres composed of ultrathin mesoporous nanosheets as a highly efficient visible-light-driven photocatalyst for H2 production. J Mater Chem A, 1, 4552(2013).
[137] F A Delfin, S P Bruhl, C Forsich et al. Carbon based DLC films: Influence of the processing parameters on the structure and properties. Materia-Brazil, 23, 1517(2018).
[138] F Rigodanza, L Dordevic, F Arcudi et al. Customizing the electrochemical properties of carbon nanodots by using quinones in bottom-up synthesis. Angew Chem Int Ed, 57, 5062(2018).
[139] R Agarwal, N M Krook, M L Ren et al. Anion exchange in II–VI semiconducting nanostructures via atomic templating. Nano Lett, 18, 1620(2018).
[140] X Y Ren, W X Liang, P Wang et al. A new approach in functionalization of carbon nanoparticles for optoelectronically relevant carbon dots and beyond. Carbon, 141, 553(2019).
[141] S Kumar, T Nann. Shape control of II–VI semiconductor nanomaterials. Small, 2, 316(2006).
[142] R G Xie, M Rutherford, X G Peng. Formation of high-quality I−III−VI semiconductor nanocrystals by tuning relative reactivity of cationic precursors. J Am Chem Soc, 131, 5691(2009).
[143] I Du Fosse, S Ten Brinck, I Infante et al. Role of surface reduction in the formation of traps in n-doped II–VI semiconductor nanocrystals: How to charge without reducing the surface. Chem Mater, 31, 4575(2019).
[144] Y Y Zhan, Z B Shao, T H Jiang et al. Cation exchange synthesis of two-dimensional vertical Cu2S/CdS heterojunctions for photovoltaic device applications. J Mater Chem A, 8, 789(2020).
[145] S Jain, S Bharti, G K Bhullar et al. I–III–VI core/shell QDs: Synthesis, characterizations and applications. J Lumin, 219, 116912(2020).
[146] A Zhang, C Dong, L Li et al. Non-blinking (Zn)CuInS/ZnS quantum dots prepared by in situ interfacial alloying approach. Sci Rep, 5, 15227(2015).
[147] L B Li, T Dong. Photoluminescence tuning in carbon dots: surface passivation or/and functionalization, heteroatom doping. J Mater Chem C, 6, 7944(2018).
[148] S Hu, W Yang, N Li et al. Carbon-dot-based heterojunction for engineering band-edge position and photocatalytic performance. Small, 14, 1803447(2018).
[149] B W Yao, H Huang, Y Liu et al. Carbon dots: A small conundrum. Trends Chem, 1, 235(2019).
[150] R Ludmerczki, S Mura, C M Carbonaro et al. Carbon dots from citric acid and its intermediates formed by thermal decomposition. Chem-Eur J, 25, 11963(2019).
[151] H Asano, S Tsukuda, M Kita et al. Colloidal Zn(Te,Se)/ZnS core/shell quantum dots exhibiting narrow-band and green photoluminescence. ACS Omega, 3, 6703(2018).
[152] T Kameyama, K Sugiura, Y Ishigami et al. Rod-shaped Zn–Ag–In–Te nanocrystals with wavelength-tunable band-edge photoluminescence in the near-IR region. J Mater Chem C, 6, 2034(2018).
[153] H Y Huang, K B Cai, P W Chen et al. Engineering ligand–metal charge transfer states in cross-linked gold nanoclusters for greener luminescent solar concentrators with solid-state quantum yields exceeding 50% and low reabsorption losses. J Phys Chem C, 122, 20019(2018).
[154] C Wang, Y Chen, T Hu et al. Color tunable room temperature phosphorescent carbon dot based nanocomposites obtainable from multiple carbon sources via a molten salt method. Nanoscale, 11, 11967(2019).
[155] H Ding, J S Wei, P Zhang et al. Solvent-controlled synthesis of highly luminescent carbon dots with a wide color gamut and narrowed emission peak widths. Small, 14, 1800612(2018).
[156] A A P Mansur, H S Mansur, R L Mansur et al. Bioengineered II–VI semiconductor quantum dot-carboxymethylcellulose nanoconjugates as multifunctional fluorescent nanoprobes for bioimaging live cells. Spectrochim Acta A, 189, 393(2018).
[157] C Xia, W Wu, T Yu et al. Size-dependent band-gap and molar absorption coefficients of colloidal CuInS2 quantum dots. ACS Nano, 12, 8350(2018).
[158] J Z Li, J L Chen, Y M Shen et al. Extinction coefficient per CdE (E = Se or S) unit for zinc-blende CdE nanocrystals. Nano Research, 11, 3991(2018).
[159] J H Hu, J L Q Song, Z S Tang et al. Phospholipid-stabilized Cu
[160] J Lu, H Liu, X Zhang et al. One-dimensional nanostructures of II–VI ternary alloys: synthesis, optical properties, and applications. Nanoscale, 10, 17456(2018).
[161] B M Wieliczka, A L Kaledin, W E Buhro et al. Wavefunction engineering in CdSe/PbS core/shell quantum dots. ACS Nano, 12, 5539(2018).
[162] Y J Wu, Y Zhang, Y R Sui et al. Bandgap engineering of Cu2In
[163] J Sun, L An, G Xue et al. Wavefunction engineering for efficient photoinduced-electron transfer in CuInS2 quantum dot-sensitized solar cells. Nanotechnology, 31, 215408(2020).
[164] Y Z Chen, W H Li, L Li et al. Progress in organic photocatalysts. Rare Met, 37, 1(2017).
[165] X Liu, Y Yan, Z Da et al. Significantly enhanced photocatalytic performance of CdS coupled nanosheets and the mechanism study. Chem Eng J, 241, 243(2014).
[166] F Meinardi, H Mcdaniel, F Carulli et al. Highly efficient large-area colourless luminescent solar concentrators using heavy-metal-free colloidal quantum dots. Nat Nanotechnol, 10, 878(2015).
[167] J Li, K Liu, J Xue et al. CQDs preluded carbon-incorporated 3D burger-like hybrid ZnO enhanced visible-light-driven photocatalytic activity and mechanism implication. J Catal, 369, 450(2019).
[168] K A Mazzio, S K K Prasad, K Okamoto et al. End-functionalized semiconducting polymers as reagents in the synthesis of hybrid II–VI nanoparticles. Langmuir, 34, 9692(2018).
[169] X B Meng, J L Sheng, H L Tang et al. Metal-organic framework as nanoreactors to co-incorporate carbon nanodots and CdS quantum dots into the pores for improved H2 evolution without noble-metal cocatalyst. Appl Catal B, 244, 340(2019).
[170] J Di, J Xia, Y Huang et al. Constructing carbon quantum dots/Bi2SiO5 ultrathin nanosheets with enhanced photocatalytic activity and mechanism investigation. Chem Eng J, 302, 334(2016).
[171] M Zhukovskyi, H Yashan, M Kuno. Low-dimensional II–VI semiconductors for photocatalytic hydrogen generation. Res Chem Intermed, 45, 4249(2019).
[172] Y Qin, H Li, J Lu et al. Nitrogen-doped hydrogenated TiO2 modified with CdS nanorods with enhanced optical absorption, charge separation and photocatalytic hydrogen evolution. Chem Eng J, 384, 123275(2020).
[173] M Jiao, Y Li, Y Jia et al. Strongly emitting and long-lived silver indium sulfide quantum dots for bioimaging: Insight into co-ligand effect on enhanced photoluminescence. J Colloid Interface Sci, 565, 35(2020).
[174] F J Fan, L Wu, S H Yu. Energetic I–III–VI2 and I2–II–IV–VI4 nanocrystals: synthesis, photovoltaic and thermoelectric applications. Energ Environ Sci, 7, 190(2014).
[175] V A Ansi, K R Vijisha, K Muraleedharan et al. Fluorescent carbon nanodots as efficient nitro aromatic sensor- analysis based on computational perspectives. Sens Actuators A, 302, 111817(2020).
Set citation alerts for the article
Please enter your email address



