- Spectroscopy and Spectral Analysis
- Vol. 41, Issue 2, 658 (2021)
Abstract
Introduction
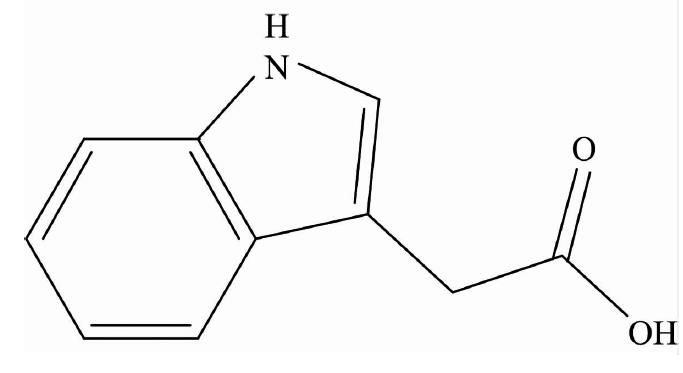
The indole-3-acetic acid (IAAH; Fig.1) is a dominant type of auxin found in plants and is involved in the growth responses of plants for the regulation of cell elongation, cell division, cell differentiation, root initiation and synthetic auxins are an important class of selective herbicides[
![]()
Figure 1.Structure of indole-3-acetic acid (IAAH)
Indole-3-acetic acid is a monocarboxylic acid that is acetic acid in which one of the methyl hydrogens has been replaced by a 1H-indol-3-yl group. It has a role as a plant hormone, a human metabolite, a plant metabolite, a mouse metabolite and an auxin. It is a monocarboxylic acid and a member of indole-3-acetic acids. Metal complexes have important applications in many aspects of life. In recent years, the successful development of metal-based pharmaceuticals has attracted more attention on metal complexes in microbial treatment[
1 Experimental
1.1 Chemicals and instruments
The chemicals presented in this article are of analytical grade and were used without further purification. These chemicals (Indole-3-acetic acid, TeCl4, SeCl4, VCl3, NbCl5 and TaCl5) were received from Sigma-Aldrich Chemical Company, USA. The analyses and the corresponding models are summarized as follows:
1.2 Synthesis
The five IAA solid complexes were prepared by mixing 1.0 mmol of TeCl4, SeCl4, VCl3, NbCl5 or TaCl5 in 25 mL of methanol solvent with 2.0 mmol IAAH (0.350 g) in 25 mL of methanol. The reaction mixtures were neutralized between pH=7~8 by 5% ammonia solution, then refluxed in a water bath for about 2 h to give the precipitate. After cooling to room temperature, the solid complexes were filtered as fine precipitates. The precipitates were washed with hot methanol. Then they were dried and stored in a desiccators containing dry calcium chloride. The yield of the products was about 80%~87%. The solid products have a higher melting point >250 ℃. The micro analytical (calculated/Calc. and experimental/Found) and physical data of the five synthesized complexes are summarized in Table 1.
1.3 Biological tests
Based on disc diffusion method[
2 Results and discussions
2.1 Elemental analysis and conductance measurements
Elemental analysis was applied to further confirm the composition of these IAA complexes. The contents of carbon, hydrogen, nitrogen and chloride in each complex were consistent with the calculated values (Table 1), which supported the proposed structure of these complexes (Fig.2). The IAAH ligand and its complexes were stable in air. Furthermore, Te(Ⅳ), Se(Ⅳ), V(Ⅲ), Nb(Ⅴ) and Ta(Ⅴ) complexes were soluble in DMSO with gently heating and their molar conductivities (Λm) were 96, 105, 14, 10, and 12 ohm-1·cm2·mol-1, respectively. Consequently, the V(Ⅲ), Nb(Ⅴ) and Ta(Ⅴ) IAA complexes were considered as non-electrolytes while the Te(Ⅳ) and Se(Ⅳ) complexes have an electrolyte feature[
![]()
Figure 2.Suggested structures of IAA complexes
2.2 FTIR spectra
The IR spectra of IAA complexes are shown in Fig.3. The important IR spectral bands of IAA complexes are presented in Table 2. According to the IR spectra data of IAAH ligand and complexes (IAA-Te, IAA-Se, IAA-V, IAA-Nb and IAA-Ta), the strong absorption peaks at ~3 400 cm-1 belong to the —NH group. No significant shifts were observed on the peaks among this ligand, suggesting that nitrogen atom in the indole ring didn’t coordinate with metal ions. Compared with the IR spectra of the IAA ligand, two new peaks at 1 549 and 1 403 cm-1 were assigned to νas(COO-) and νs(COO-), respectively[
![]()
Figure 3.FTIR spectra of IAA complexes Ⅰ—Ⅴ
Table Infomation Is Not Enable2.3 Electronic spectra and magnetic susceptibility
The UV-Vis spectrum of indol-3-acetic acid dissolved in DMSO solvent has displayed a two distinguish peaks at 219 and 280 nm that assigned to π—π* electronic transitions[
2.4 1HNMR spectra
The 1HNMR spectrum of free IAAH ligand (400 MHz, DMSO-d6): δ 12.16 [1H, COOH], 10.91 [1H, NH], 7.51~6.97 [5H, —C6H4, -C=CH] 3.64 [2H, CH2]. The 1H NMR spectra of IAA-Te, IAA-Se, IAA-Nb, and IAA-Ta complexes (Table 3) showed that there were upfield or downfield shifts in the signals among IAA ligand, suggesting the interaction between IAA ligand and metal ion. In addition, the signal for NH proton in the spectra of complexes Ⅰ, Ⅱ, Ⅳ, and Ⅴ are 10.909, 11.100, 10.909, and 10.948 ppm, respectively were still present, while the signal for the COOH proton of the IAA ligand was absent in these complexes, indicating the hydrogen atom of carboxyl group were displaced by metal ion.
2.5 Thermo gravimetric analysis
The TG analyses of IAA complexes Ⅰ—Ⅴ were performed in the temperature range of 30~700 ℃. The TG curves of the five IAA complexes are difference, further indicating that the five complexes possess a difference metal ions and structures. For clarity’s sake, only the TG curve of complex Ⅲ has been shown in Fig.4. From 30 to 700 ℃, the weight loss of complex Ⅲ is 71%, corresponding to the decomposition of IAA, and release of NH3 and HCl molecules, in a good accordance with the calculated value (71.02%). It was found that the five complexes (Ⅰ—Ⅴ) are thermally decomposed with DrTGA at 260, 307, 257, 450, and 254 ℃, respectively. Thermal analysis (TG) of IAA complexes IAA-Te, IAA-Se, IAA-V, IAA-Nb, and IAA-Ta were recorded in the range from 30-to-700 ℃. These complexes were all decomposed actually in one or two steps and the residue rates of them were 21.97%, 0.0%, 28.98%, 16.97%, and 28.47%, respectively, which were assigned to tellurium metal, sublimated selenium, VO2 polluted with few carbon atoms, niobium metal, and tantalum metal.
![]()
Figure 4.TG-DrTGA curve of [V(IAA)2(NH3)(Cl)] complex
2.6 Morphological analysis
The X-ray powder diffraction patterns of IAA complexes (Ⅰ—Ⅴ) at the range of 2θ=5°~90° are shown in Fig.5, these patterns have three types of structures as semi-crystalline for tellurium(Ⅳ) complex (Ⅰ), crystalline nature for selenium(Ⅳ) and vanadium(Ⅲ) complexes while both niobium (Ⅴ) and tantalum(Ⅴ) complexes have an amorphous structure (Fig.5). The particle size was calculated dependent on Scherrer relationship[
![]()
Figure 5.XRD pattern of IAA complexes Ⅰ—Ⅴ
TEM photos of the [Te(IAA)2(NH3)2]·2Cl (Ⅰ) and [Se(IAA)2(NH3)2]·2Cl (Ⅳ) complexes are displayed in Fig.6, the complexes refer to a black spots with spherical shape nanoparticles within 7~12 and 50~90 nm, respectively. SEM images of all IAA complexes (Fig.7) were scanned at energy 30 kV with magnification ×5 000~40 000. Figure 7 show the aggregation of tremendous micro-size fine powders and homogeneous uniform.
![]()
Figure 6.TEM images of (Ⅰ): tellurium and (Ⅳ): niobium complexes
![]()
Figure 7.SEM images of IAA complexes Ⅰ—Ⅴ
2.7 Biological results
To test the antibacterial property of the five IAA complexes in this study, four bacterial strains Klebsiella (g-), Escherichia coli (g-), Staphylococcus aureus (g+) and Staphylococcus epidermidis (g+) were used in the investigation. Disc diffusion method was used to evaluate the antibacterial activity for the selected strains. Gentamicin and Ceftriaxone were used as positive controls and DMSO, the solvent, was used as negative control in this test. As shown in Table 4 marginal antibacterial activity was observed in all complexes against Klebsiella strain only, whereas no antibacterial effect was seen in all complexes against the rest of the tested bacterial strains.
The effects of the five IAA complexes in the cytotoxicity of Caco-2 and Mcf-7 human cancer cell lines were studied by NR assay method. The percentage of cellular viability (IC50) for the five IAA complexes against two tumour cell lines Caco-2 and Mcf-7 deduced that only [V(IAA)2(NH3)Cl)] complex can be considered as a potential anticancer agent against colorectal adenocarcinoma cell line with IC50 value of 21.4 μg·mL-1. The IC50 values for the rest of IAA complexes against the two tumour cell lines were higher than >100 μg·mL-1.
References
[1] W M Gray, M Quint. Curr. Opin. Plant Biol, 9, 448(2006).
[2] Y Zhao. Annu. Rev. Plant. Biol, 61, 49(2010).
[3] N Hu, Y Jiang, Y Wu et al. Appl. Soil Ecology, 147, 103447(2020).
[4] J Ran, W Zheng, H Wang et al. Ecotoxicology and Environmental Safety, 19115, 110213(2020).
[5] Y Shu H, R Lin H, H Lin G. Microbiological Research, 216, 30(2018).
[6] S Srivastava, A Upadhyay, M Kochar. Research in Microbiology, 162, 426(2011).
[7] A Al-Saif F, A Alfurhood J, A Alibrahim K et al. J. Mol. Liq, 249, 438(2018).
[8] A Alibrahim K, F A Al-Saif, M T Alghamdi et al. RSC Advances, 8, 22515(2018).
[9] A Alibrahim K, A Al-Saif F, H Alosaimi E et al. J. Mol. Liq, 266, 242(2018).
[10] G Shchelochkov A, A Kamnev A, D Perfiliev Y et al. J. Mol. Struct, 563-564, 565(2001).
[11] L Blair V, A Pathak, L Ferrero R et al. J. Inorg. Biochem, 177, 266(2017).
[12] Y Bai F, N Xing, T Xu L et al. Inorg. Chim. Acta, Part B, 360.
[13] A Kirby W, W Bauer A, C Sherris et al. Am. J. Clin. Pathology, 45, 493(1996).
[14] L Zurita J, A del Peso, G Repetto. Neutral Red Uptake Assay for the Estimation of Cell Viability/Cytotoxicity, Nature Protocols(2008).
[15] A El-Habeeb A, S Refat M. J. Mol. Struct, 1175, 65(2019).
[16] K Nakamoto. Infrared and Raman Spectra of Inorganic and Coordination Compounds, fourth ed(1986).
[17] R J Philips, B Deacon G. Coord. Chem. Rev, 33, 227(1980).
[18] P Lever A B. Electronic Spectra of dn Ions Inorganic Electronic Spectroscopy(1984).
[19] E Knox J, J Heeg M, R Gust K et al. Eur. J. Inorg. Chem, 9, 2327(2002).
[20] A Syed, S Kumar, S Andotra et al. J. Mol. Struct, 1154, 165(2018).
[21] R Stock S, D Cullity B. Elements of X-ray Diffraction, 389(2001).
Set citation alerts for the article
Please enter your email address



