Hayoung Jeong, Paulson Bjorn, Seongjin Hong, Seunguk Cheon, Kyunghwan Oh, "Irreversible denaturation of DNA: a method to precisely control the optical and thermo-optic properties of DNA thin solid films," Photonics Res. 6, 918 (2018)
Search by keywords or author
- Photonics Research
- Vol. 6, Issue 9, 918 (2018)
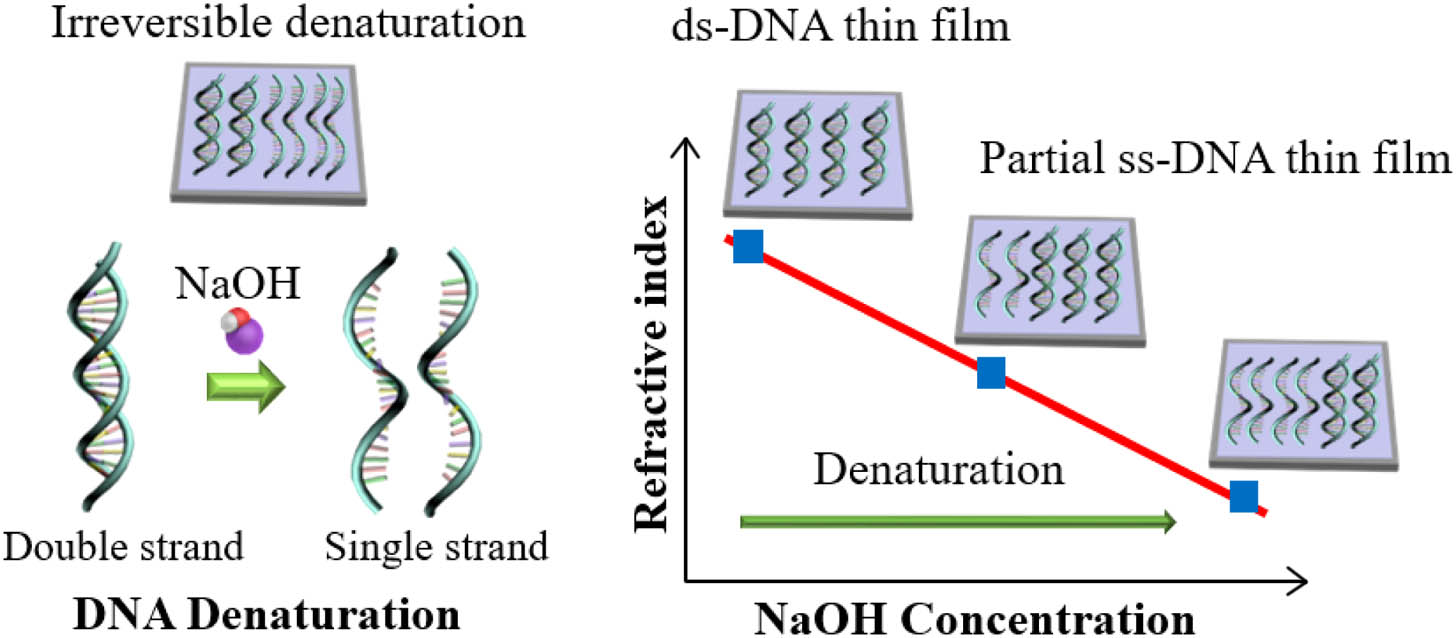
Fig. 1. Schematic diagram to control the refractive index of DNA thin solid film by denaturation. Denaturation is activated by adding NaOH in DNA aqueous solution precursors, which is irreversibly immobilized in thin solid film to change the refractive index (ds, double stranded; ss, single stranded).
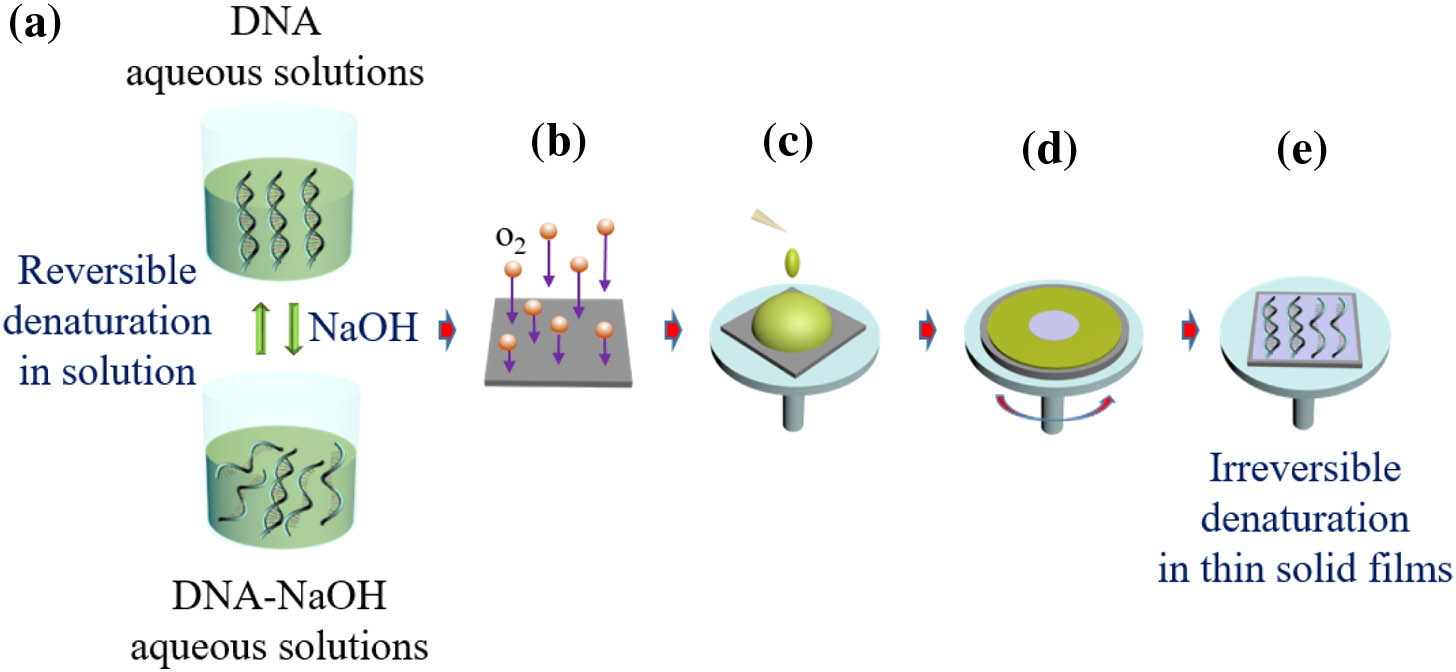
Fig. 2. DNA thin solid film fabrication process. (a) DNA aqueous solution and DNA-NaOH aqueous solution where the denaturation is reversible. (b) O 2 Si / SiO 2
Fig. 3. (a) UV/visible spectra of DNA aqueous solution with various NaOH concentrations in the precursor solutions. Here we used 0.15 wt. % DNA. (b) Hyperchromicity near λ = 260 nm
Fig. 4. (a) FTIR absorption spectra of solid freestanding DNA films made from DNA aqueous solutions with various NaOH concentrations. (b) Spectral shift of the cytosine vibration peak as a function of NaOH concentration in the DNA precursor solutions.
Fig. 5. (a) The refractive indices of DNA thin solid film in the spectral range from 380 to 900 nm for various NaOH concentrations in precursor solutions. (b) The refractive indices of DNA thin solid film as a function of NaOH concentration in the precursor solutions.
Fig. 6. Thermally induced changes in the refractive index and the film thickness of DNA thin solid film with various NaOH concentrations in precursor solutions. (a) Refractive index at λ = 633 nm λ = 633 nm
|
Table 1. Average Thickness of DNA Thin Solid Films Made from Precursor Solutions with Various NaOH Concentrations
|
Table 2. Thermo-Optic Coefficient of DNA Thin Solid Film at a Wavelength of 633 nm for Various NaOH Concentrations in the Precursor Solutions

Set citation alerts for the article
Please enter your email address



