- Journal of Semiconductors
- Vol. 41, Issue 4, 041607 (2020)
Abstract
1. Introduction
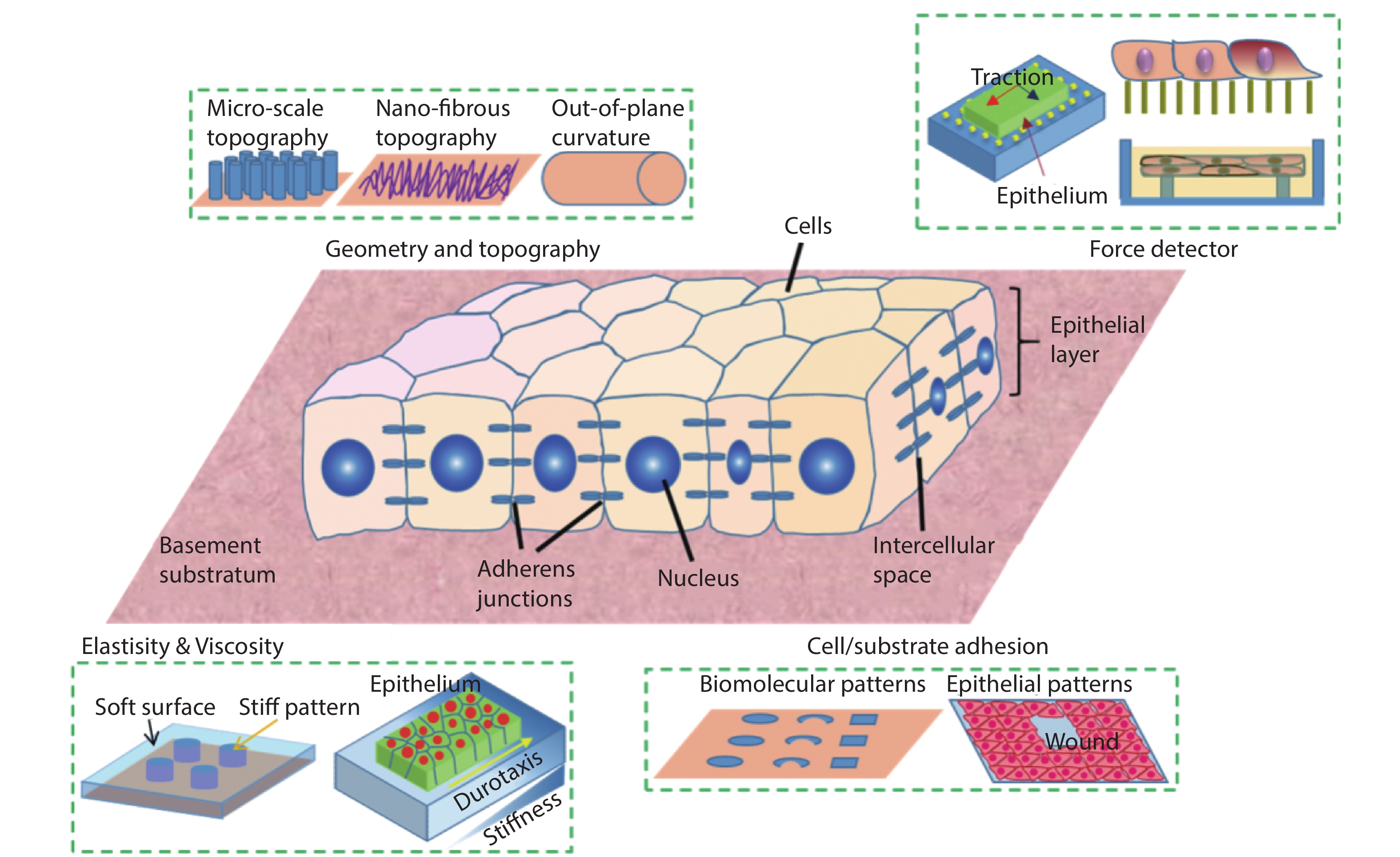
Designer substrates and devices have long been the heart of biomaterials, bioengineering, and a powerful tool for developmental biology, cell biology, biomedical studies. Especially, the high-cost of animal models and their concomitant long experimental cycle, ethical issues and poor reproducibility have cried out for in vitro substrates that mimic various in vivo conditions and tissue functions. The past decade has witnessed a surge in the interdisciplinary efforts in soft lithography, bio-inspired microfabrication, biochemistry and cell biology, allowing the design and production of sophisticated platforms that are capable to recapitulate natural contexts and elicit physiologically relevant events outside the body (Fig. 1). This emerging field that studies cell and tissue mechanics with biological and engineering approaches is coined as mechanobiology. Progress in this field has shed light on important biological processes of live systems at the interface of biology and artificial substrates, which are directly related to the design and advance of next generation biomaterials, biomedical devices, and large scale tissue engineering[
![]()
Figure 1.(Color online) Microengineered synthetic substrates for cell/tissue mechanics studies. The properties of a substratum can be modified to adjust the cell/material interactions, such as surface topographies, stiffness, and adhesiveness. In addition, mechanical probes can be integrated into the substrate to detect the force in tissue. These include microbeads in the traction force microscopy and elastomeric micro-pillars.
Live systems are made of cells that can support tissue development and homeostasis through processes such as self-replication, renewal, selective destruction and sensing of the microenvironment. These processes require the cells to actively react to the environmental inputs, while having sufficient mechanical stability to sustain shape and function, and at the same time adequate fluidity for remodeling. Supported by increasing evidences from reconstituted molecular systems and single cell studies, it is now known that these active properties can be attributed at the molecular level through ATP consumption and in particular, by the activity of cell cytoskeleton and molecular motors[
Such cell/tissue mechanosensing behaviors are influenced by a plethora of biochemical and biophysical cues[
In this review, we discuss how specific properties and functions of designer substrates/devices are achievable by various microfabrication methods and explain how underlying biological mechanisms due to cell-microenvironment interactions can be disclosed by these capabilities. In brief, we emphasize on semiconductor-based techniques that advance the study of cell/tissue mechanical responses to substrate adhesiveness, stiffness, topography, and shear flow. Moreover, we comment on the new concepts of measurement and paradigms for investigations of biological mechanotransduction that are yet to emerge due to on-going interdisciplinary efforts in the fields of mechanobiology and microengineering.
2. Engineering substrate adhesion
One important aspect of biologists’ concerns is the cell/substrate interface and their interaction. Attachment of cells to a surface via adhesion complexes (ACs, Fig. 2) provides important feedbacks that trigger a variety of signaling cascades and cellular behaviors. In vivo, cells take advantage of the heterogeneous distribution of extracellular matrix (ECM) cues to define many vital processes, such as wound healing[
![]()
Figure 2.(Color online) Molecular dynamics at adhesion complexes. The actin network as a mechanosensitive machine connecting the cell to its substrate and neighbors. The building of a stable focal adhesion (FA) complex for cell–substrate adhesion. Actomyosin forces apply on the FA at a fixed speed and the rate of force increase in the complex increases proportionally with the ECM stiffness. To avoid the destabilization and detachment of the FA, the binding-unbinding dynamics of the transmembrane protein, integrin, that connects the cells to the substrate needs to be equal to the force loading rate in the complex. Another force buffer and mechanosensor in the complex is Talin. Its unfolding at ~ 10 pN at the normal rate of force loading in cells lead to vinculin binding to recruit more actin fibers, thus reinforcing the FA.
![]()
Figure 3.(Color online) Methods for patterning adhesive surfaces. Semiconductor-based technologies has allowed the development of micro-contact printing and micro-stenciling for patterning biomolecules with define shapes. Later, researchers developed other techniques for this purpose, including Dip-pen lithography and UV-based patterning.
Patterning proteins is now made possible with multiple methods (Fig. 3) and has become a routine technique in many biological studies. As early as late 1990s, the Ingber group used μCP to constraint cells within arbitrary adhesive shapes containing fibronectin, a binding partner of integrin, and found that different areas of printing as well as its protein constituent results in dramatically modulation of cell growth and death[
3. Engineering substrate stiffness
The fact that FA growth is coupled to the cell–substrate interaction leads to the postulation that cells tune their contractility according to the substrate rigidity. In this context, a series of methods have been developed to adjust substrate stiffness. Conventional approaches involvethe modification of the cross-linking degree of the gels (Fig. 4), including polyacrylamide (PAA) and polydimethylsiloxane (PDMS). Typically, their Yong’s modulus can be changed over at least two orders of magnitudes, i.e., from 1 to 100 kPa, which is overlapping with the rigidity range of in vivo tissues[
![]()
Figure 4.(Color online) Methods for engineering substrate elasticity and viscosity. Conventionally, by controlling the cross-linking degree in elastomers, one could adjust the viscoelasticity of a gel. Another approach to change substrate rigidity involving photolithography is to pattern pillars of different shapes.
As discussed above, most types of cells response to the resistance via their attachment to a surface. Light resistance on a soft, flexible substrate will not stretch the force-bearing proteins, e.g., talin and vinculin, and leads to diffuse and dynamic ACs[
4. Engineering substrate topography
Many biological studies are still carried out on planar and featureless substrates, while perfectly flat surfaces do not usually exist in vivo. Instead, cells in physiological environment often experience complex 3D architectures and out-of-plane curvatures. To mimic in vivo situations and to study cell interactions with these features, cells are seeded on synthetic substrate harboring precise surface topographies. In general, photolithography is combined with etching techniques to create prescribed 3D topographic structures and later, functional substrates are produced using replica molding from masters[
Previous studies revealed that cellular FAs have a broad range of size from about 10 nm to 10 μm, suggesting cell may employ dynamic FA mechanism to sense the topographical cues. For instance, cell migratory dynamics changes on an array of 10 μm size micropillars, where the cells move slower with higher persistence in comparison with that on a flat surface[
![]()
Figure 5.(Color online) Topography cues influences cell adhesion and migration. (a) Scanning electron micrographs (SEMs) showing cells aligned to the nano-lines (Reproduced from Ref. [
5. Microfluidic chip
One major goal of bioengineering is to establish in vitro models that emulate the architecture, function, microenvironment, and physiological processes of living tissues. The aforementioned techniques show various advantages in understanding various mechano-properties of cells/tissues, but they also face limitations. Recently, with advanced microfabrication methods there is a surge in the integration of multiple moduli that provide controls to various biochemical/physical cues into a single microfluidic chip[
The fluid shear stress is due to the flow imposed on the tissues such as epithelial sheets that line ducts and endothelia that line the vessels. Epithelial cells sense fluid flow via their primary cilia – as the primary cilia are bended, the Ca2+-signaling pathway is elicited[
![]()
Figure 6.(Color online) Shear stress influences cell adherens junction (AJ) and filopodia protrusion. (a) At AJs, a higher force transmitted from F-actin caused by other factors (such as shear) leads to
Such active behaviors have been attributed to active cellular force distribution changes under static or flow conditions measured using traction force microscopy (TFM), micropillars and/or FRET bio-sensor techniques[
Moreover, cancer cell migration and invasion can also be stimulated by hydrodynamic currents in the form of lymphatic flows experienced by cancers cells that have spread into the lymphatic system, and interstitial flows between the ECM of tissues. It was found that wall shear stress few orders of magnitude lower than that experienced by ECs (~ 1 Pa for ECs) induced by flow on a dense but non-confluent layer of PC3 prostate cancer cells on the inner collagen-coated surfaces of a cylindrical PDMS tube, could stimulate Yes-associated protein (YAP) activation and increase filopodia protrusion and migration[
6. Outlook
The recent advancement of mechanobiology has been largely based on the rapid development of material microfabrication and engineering methods to recapitulate aspects of in vivo cell/tissue milieus. Microenvironmental factors, including the surface affinity to cells, substrate rigidity and topography, as well as shear flow have been found to contribute significantly for cell adhesion, migration, polarization, and differentiation. Our ability to independently control a range of parameters has offered unprecedented insight into cell/tissue mechanical properties. This is further combined with conventional cell biology approaches, which results in many discoveries in novel molecular pathways that are related to various mechanosensing mechanisms. The latest efforts in this field begin to focus on even more complex microenvironmental controls[
Acknowledgements
The authors thank T. B. Saw and C. T. Lim from the Mechanobiology Institute, Singapore and the members of CAML group at Institut Jacques Monod, Paris for fruitful discussions. Financial supports from the European Research Council (ERC) under the European Union’s Seventh Framework Programme (FP7/2007-2013) / ERC grant agreement numbers 617233 (B.L.), Marie Skłodowska-Curie Actions (W.X., Individual Fellowship, Project: 846449), the Groupama Foundation – Research Prize for Rare Diseases 2017 (to D.D), the Fondation pour la Recherche Médicale (FRM) (to D.D.), the LabEx “Who Am I?” #ANR-11-LABX-0071 and the Université de Paris IdEx #ANR-18-IDEX-0001 funded by the French Government through its “Investments for the Future” program (to D.D), and the Human Frontier Science Program (RGP0038/2018) (to D.D) are gratefully acknowledged.
References
[1] W Xi, T B Saw, D Delacour et al. Material approaches to active tissue mechanics. Nat Rev Mater, 4, 23(2019).
[2] J Prost, F Jülicher, J F Joanny. Active gel physics. Nat Phys, 11, 111(2015).
[3] M C Marchetti, J F Joanny, S Ramaswamy et al. Hydrodynamics of soft active matter. Rev Mod Phys, 85, 1143(2013).
[4] D Needleman, Z Dogic. Active matter at the interface between materials science and cell biology. Nat Rev Mater, 2, 17048(2017).
[5] C Grashoff, B D Hoffman, M D Brenner et al. Measuring mechanical tension across vinculin reveals regulation of focal adhesion dynamics. Nature, 466, 263(2010).
[6] G Giannone, B J Dubin-Thaler, H G Döbereiner et al. Periodic lamellipodial contractions correlate with rearward actin waves. Cell, 116, 431(2004).
[7] A Ray, O Lee, Z Win et al. Anisotropic forces from spatially constrained focal adhesions mediate contact guidance directed cell migration. Nat Commun, 8, 14923(2017).
[8] X Jiang, D A Bruzewicz, A P Wong et al. Directing cell migration with asymmetric micropatterns. Proc Natl Acad Sci USA, 102, 975(2005).
[9] B Chen, G Kumar, C C Co et al. Geometric control of cell migration. Sci Rep, 3, 1(2013).
[10] H E Johnson, S J King, S B Asokan et al. F-actin bundles direct the initiation and orientation of lamellipodia through adhesion-based signaling. J Cell Biol, 208, 443(2015).
[11] A J Engler, S Sen, H L Sweeney et al. Matrix elasticity directs stem cell lineage specification. Cell, 126, 677(2006).
[12] S R Naganathan, T C Middelkoop, S Fürthauer et al. Actomyosin-driven left-right asymmetry: from molecular torques to chiral self organization. Curr Opin Cell Biol, 38, 24(2016).
[13] Y H Tee, T Shemesh, V Thiagarajan et al. Cellular chirality arising from the self-organization of the actin cytoskeleton. Nat Cell Biol, 17, 445(2015).
[14] M Gupta, B R Sarangi, J Deschamps et al. Adaptive rheology and ordering of cell cytoskeleton govern matrix rigidity sensing. Nat Commun, 6, 1(2015).
[15] L Trichet, J Le Digabel, R J Hawkins et al. Evidence of a large-scale mechanosensing mechanism for cellular adaptation to substrate stiffness. Proc Natl Acad Sci USA, 109, 6933(2012).
[16] A Zemel, F Rehfeldt, A E Brown et al. Optimal matrix rigidity for stress-fibre polarization in stem cells. Nat Phys, 6, 468(2010).
[17] E M Levina, L V Domnina, Y A Rovensky et al. Cylindrical substratum induces different patterns of actin microfilament bundles in nontransformed and in ras-transformed epitheliocytes. Exp Cell Res, 229, 159(1996).
[18] T M Svitkina, Y A Rovensky, A D Bershadsky et al. Transverse pattern of microfilament bundles induced in epitheliocytes by cylindrical substrata. J Cell Sci, 108, 735(1995).
[19] B Sun, K Xie, T H Chen et al. Preferred cell alignment along concave microgrooves. RSC Adv, 7, 6788(2017).
[20] Y Y Biton, S A Safran. The cellular response to curvature-induced stress. Phys Biol, 6, 046010(2009).
[21] N D Bade, R D Kamien, R K Assoian et al. Curvature and Rho activation differentially control the alignment of cells and stress fibers. Sci Adv, 3, e1700150(2017).
[22] R De, A Zemel, S A Safran. Dynamics of cell orientation. Nat Phys, 3, 655(2007).
[23] A Livne, E Bouchbinder, B Geiger. Cell reorientation under cyclic stretching. Nat Commun, 5, 3938(2014).
[24] V K Sidhaye, K S Schweitzer, M J Caterina et al. Shear stress regulates aquaporin-5 and airway epithelial barrier function. Proc Natl Acad Sci USA, 105, 3345(2008).
[25] B Ladoux, R M Mège. Mechanobiology of collective cell behaviours. Nat Rev Molecul Cell Biol, 18, 743(2017).
[26] S Wong, W H Guo, Y L Wang. Fibroblasts probe substrate rigidity with filopodia extensions before occupying an area. Proc Natl Acad Sci USA, 111, 17176(2014).
[27] A Saez, M Ghibaudo, A Buguin et al. Rigidity-driven growth and migration of epithelial cells on microstructured anisotropic substrates. Proc Natl Acad Sci USA, 104, 8281(2007).
[28] X Wang, S Li, C Yan et al. Fabrication of RGD micro/nanopattern and corresponding study of stem cell differentiation. Nano Lett, 15, 1457(2015).
[29] T B Saw, A Doostmohammadi, V Nier et al. Topological defects in epithelia govern cell death and extrusion. Nature, 544, 212(2017).
[30] J Salomon, C Gaston, J Magescas et al. Contractile forces at tricellular contacts modulate epithelial organization and monolayer integrity. Nat Commun, 8, 13998(2017).
[31] Y Wang, D B Gunasekara, M I Reed et al. A microengineered collagen scaffold for generating a polarized crypt-villus architecture of human small intestinal epithelium. Biomaterials, 128, 44(2017).
[32] K M Stroka, H Jiang, S H Chen et al. Water permeation drives tumor cell migration in confined microenvironments. Cell, 157, 611(2014).
[33] M Raab, M Gentili, H de Belly et al. ESCRT III repairs nuclear envelope ruptures during cell migration to limit DNA damage and cell death. Science, 352, 359(2016).
[34] C M Denais, R M Gilbert, P Isermann et al. Nuclear envelope rupture and repair during cancer cell migration. Science, 352, 353(2016).
[35] H D Moreau, C Blanch-Mercader, R Attia et al. Macropinocytosis overcomes directional bias in dendritic cells due to hydraulic resistance and facilitates space exploration. Develop Cell, 49, 171(2019).
[36] W Xi, C K Schmidt, S Sanchez et al. Rolled-up functionalized nanomembranes as three-dimensional cavities for single cell studies. Nano Lett, 14, 4197(2014).
[37] W Xi, C K Schmidt, S Sanchez et al. Molecular insights into division of single human cancer cells in on-chip transparent microtubes. ACS Nano, 10, 5835(2016).
[38] B Koch, A K Meyer, L Helbig et al. Dimensionality of rolled-up nanomembranes controls neural stem cell migration mechanism. Nano Lett, 15, 5530(2015).
[39] F X Maquart, J C Monboisse. Extracellular matrix and wound healing. Pathol Biol, 62, 91(2014).
[40] H Kobayashi, A Enomoto, S L Woods et al. Cancer-associated fibroblasts in gastrointestinal cancer. Nat Rev Gastroenterol Hepatol, 16, 282(2019).
[41] H K Kleinman, D Philp, M P Hoffman. Role of the extracellular matrix in morphogenesis. Curr Opin Biotechnol, 14, 526(2003).
[42] Y Xia, G M Whitesides. Soft lithography. Ann Rev Mater Sci, 28, 153(1998).
[43] D Qin, Y Xia, G M Whitesides. Soft lithography for micro-and nanoscale patterning. Nat Protoc, 5, 491(2010).
[44] Y Xia, E Kim, X M Zhao et al. Complex optical surfaces formed by replica molding against elastomeric masters. Science, 273, 347(1996).
[45] P O Strale, A Azioune, G Bugnicourt et al. Multiprotein printing by light-induced molecular adsorption. Adv Mater, 28, 2024(2016).
[46] C S Chen, M Mrksich, S Huang et al. Geometric control of cell life and death. Science, 276, 1425(1997).
[47] J T Parsons, A R Horwitz, M A Schwartz. Cell adhesion: integrating cytoskeletal dynamics and cellular tension. Nat Rev Mol Cell Biol, 11, 633(2010).
[48] P Kanchanawong, G Shtengel, A M Pasapera et al. Nanoscale architecture of integrin-based cell adhesions. Nature, 468, 580(2010).
[49] N Wang, J P Butler, D E Ingber. Mechanotransduction across the cell surface and through the cytoskeleton. Science, 260, 1124(1993).
[50] N Q Balaban, U S Schwarz, D Riveline et al. Force and focal adhesion assembly: a close relationship studied using elastic micropatterned substrates. Nat Cell Biol, 3, 466(2001).
[51] X Wang, T Ha. Defining single molecular forces required to activate integrin and notch signaling. Science, 340, 991(2013).
[52] J Fink, N Carpi, T Betz et al. External forces control mitotic spindle positioning. Nat Cell Biol, 13, 771(2011).
[53] D E Discher, P Janmey, Y L Wang. Tissue cells feel and respond to the stiffness of their substrate. Science, 310, 1139(2005).
[54] O Du Roure, A Saez, A Buguin et al. Force mapping in epithelial cell migration. Proc Natl Acad Sci USA, 102, 2390(2005).
[55] Y Shao, J Fu. Integrated micro/nanoengineered functional biomaterials for cell mechanics and mechanobiology: a materials perspective. Adv Mater, 26, 1494(2014).
[56] R J Pelham, Y L Wang. Cell locomotion and focal adhesions are regulated by substrate flexibility. Proc Natl Acad Sci USA, 94, 13661(1997).
[57] B R Sarangi, M Gupta, B L Doss et al. Coordination between intra-and extracellular forces regulates focal adhesion dynamics. Nano Lett, 17, 399(2017).
[58] D Mitrossilis, J Fouchard, A Guiroy et al. Single-cell response to stiffness exhibits muscle-like behavior. Proc Natl Acad Sci USA, 106, 18243(2009).
[59] R Sunyer, V Conte, J Escribano et al. Collective cell durotaxis emerges from long-range intercellular force transmission. Science, 353, 1157(2016).
[60] M J Paszek, N Zahir, K R Johnson et al. Tensional homeostasis and the malignant phenotype. Cancer Cell, 8, 241(2005).
[61] K R Levental, H Yu, L Kass et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell, 139, 891(2009).
[62] S M Weiz, M Medina-Sánchez, O G Schmidt. Microsystems for single-cell analysis. Adv Biosyst, 2, 1700193(2018).
[63] M Ghibaudo, L Trichet, J Le Digabel et al. Substrate topography induces a crossover from 2D to 3D behavior in fibroblast migration. Biophys J, 97, 357(2009).
[64] P Clark, P Connolly, A S Curtis et al. Topographical control of cell behaviour. I. Simple step cues. Development, 99, 439(1987).
[65] E K Yim, R M Reano, S W Pang et al. Nanopattern-induced changes in morphology and motility of smooth muscle cells. Biomaterials, 26, 5405(2005).
[66] G M Whitesides. The origins and the future of microfluidics. Nature, 442, 368(2006).
[67] N J Douville, P Zamankhan, Y C Tung et al. Combination of fluid and solid mechanical stresses contribute to cell death and detachment in a microfluidic alveolar model. Lab Chip, 11, 609(2011).
[68] J Rahimzadeh, F Meng, F Sachs et al. Real-time observation of flow-induced cytoskeletal stress in living cells. Am J Physiol-Cell Physiol, 301, C646(2011).
[69] Y Imura, K Sato, E Yoshimura. Micro total bioassay system for ingested substances: assessment of intestinal absorption, hepatic metabolism, and bioactivity. Anal Chem, 82, 9983(2010).
[70] H A Praetorius, K R Spring. Bending the MDCK cell primary cilium increases intracellular calcium. J Membr Biol, 184, 71(2001).
[71] H A Praetorius, K R Spring. Removal of the MDCK cell primary cilium abolishes flow sensing. J Membr Biol, 191, 69(2003).
[72] K J Jang, K Y Suh. A multi-layer microfluidic device for efficient culture and analysis of renal tubular cells. Lab Chip, 10, 36(2010).
[73] K J Jang, H S Cho, D H Kang et al. Fluid-shear-stress-induced translocation of aquaporin-2 and reorganization of actin cytoskeleton in renal tubular epithelial cells. Integr Biol, 3, 134(2011).
[74] K J Jang, A P Mehr, G A Hamilton et al. Human kidney proximal tubule-on-a-chip for drug transport and nephrotoxicity assessment. Integr Biol, 5, 1119(2013).
[75] N Baeyens, C Bandyopadhyay, B G Coon et al. Endothelial fluid shear stress sensing in vascular health and disease. J Clin Invest, 126, 821(2016).
[76] S Kawashima, M Yokoyama. Dysfunction of endothelial nitric oxide synthase and atherosclerosis. Arterioscler, Thromb, Vasc Biol, 24, 998(2004).
[77] N Baeyens, M J Mulligan-Kehoe, F Corti et al. Syndecan 4 is required for endothelial alignment in flow and atheroprotective signaling. Proc Natl Acad Sci USA, 111, 17308(2014).
[78] D E Conway, M T Breckenridge, E Hinde et al. Fluid shear stress on endothelial cells modulates mechanical tension across VE-cadherin and PECAM-1. Curr Biol, 23, 1024(2013).
[79] R Jr Steward, D Tambe, C C Hardin et al. Fluid shear, intercellular stress, and endothelial cell alignment. Am J Physiol-Cell Physiol, 308, C657(2015).
[80] S S Hur, J C Del Alamo, J S Park et al. Roles of cell confluency and fluid shear in 3-dimensional intracellular forces in endothelial cells. Proc Natl Acad Sci USA, 109, 11110(2012).
[81] S Ohta, S Inasawa, Y Yamaguchi. Alignment of vascular endothelial cells as a collective response to shear flow. J Phys D, 48, 245401(2015).
[82] H J Lee, M F Diaz, K M Price et al. Fluid shear stress activates YAP1 to promote cancer cell motility. Nat Commun, 8, 14122(2017).
[83] W J Polacheck, J L Charest, R D Kamm. Interstitial flow influences direction of tumor cell migration through competing mechanisms. Proc Natl Acad Sci USA, 108, 11115(2011).
[84] A S Piotrowski-Daspit, J Tien, C M Nelson. Interstitial fluid pressure regulates collective invasion in engineered human breast tumors via Snail, vimentin, and E-cadherin. Integr Biol, 8, 319(2016).
[85] S N Bhatia, D E Ingber. Microfluidic organs-on-chips. Nat Biotechnol, 32, 760(2014).
[86] L Li, J Eyckmans, C S Chen. Designer biomaterials for mechanobiology. Nat Mater, 16, 1164(2017).
Set citation alerts for the article
Please enter your email address



