- Journal of Semiconductors
- Vol. 41, Issue 9, 091705 (2020)
Abstract
1. Introduction
In the rapid development and advancement of industrial society, the issues of excessive exploitation and over-reliance on fossil fuel accelerate the depletion earth's resources, which tremendously threatens our future. Meanwhile, the over-usage of fossil fuels leads to severe environment pollution and extreme climate change jeopardizing the whole ecosystem on the earth. Hence, the concerns about sustainability and security of energy on our planet have been raised widely, which urges researchers to address these serious issues by developing versatile energy conversion technologies to store and recycle clean and renewable sources, thus, reducing the dependency on traditional fossil fuel[
Water splitting was the most wide-studied electrocatalysis process owing to convenience, negligible environment pollution, and huge water feedstocks. By far, however, the overall efficiency of water splitting is still obstructed, primarily, by the sluggish kinetics of the half reaction of oxygen evolution[
In terms of these conventional pure metal electrocatalysts, no matter which reactions they are employed to work for, most of them endure either limited source, high cost, poor stability, or low Faradaic efficiency and selectivity[
2. Fundamentals of water splitting
The overall efficiency of water splitting is undoubtedly a vital factor that determines the future of the large-scale applications. To this end, a good and in-depth understanding of reaction mechanism and rate/potential-determining steps is very necessary to gain insight into questions like how to rationally design and fabricate an ideal electrocatalysts and how to improve the design of an electrolyzer device for the reducing energy lost during energy conversion process. Therefore, in this section, we will briefly introduce some fundamental knowledge background regarding the well-accepted reaction mechanism and some necessary concepts for understanding the water splitting reaction. The related reaction pathways of HER and OER are presented firstly followed by the representative theoretically results obtained from density functional theory (DFT) calculations.
2.1. The overall reactions in water splitting
In an electrolyzer coupling with a stable power supply, water could be constantly split to hydrogen and oxygen (O2) via two crucial multi-proton/electron coupled half-cell reactions, cathodic HER and anodic OER. According to the Nernst equation under standard conditions (25 °C, 1 atm), the thermodynamic voltage of this electrolyzer is estimated to be 1.23 V related to a reversible hydrogen electrode (RHE), regardless of the type of electrolyte[
Overall reaction:
In acidic electrolyte:
In neutral and alkaline electrolyte:
In practice, to initiate water splitting, high activation energy, sluggish kinetics, and poor energy efficiency need to be overcame, which demands a greater applied potential rather than 1.23 V decided by Nernst equation[
where iR designates the ohmic potential drop owing to the resistance of ionic electrolyte, which cannot be avoided but can minimized by improving the electrolyzer setup.
2.2. Reaction pathways of HER
Regarding the pH of the electrolyte, the HER could take places based on either the Volmer-Heyrovsky or the Volmer-Tafel mechanisms. The specific reaction pathways are described below:
(1) Volmer steps: A reaction species adopts an electron at an active site to form an adsorbed hydrogen.
(2) Heyrovsky steps: A reaction species (a proton or a molecular water) and an electron are adopted by adsorbed hydrogen to form a hydrogen molecule and then desorb from electrode surface.
(3) Tafel step: Two adsorbed hydrogen atoms are coupled to produce a hydrogen molecule and the desorb from surface.
where * represents an active site on electrode surface and H* designates the adsorbed hydrogen atom. The initiation of HER always starts with Volmer steps (Eqs. (7) and (8)) to capture and fix a proton on electrode surface for the subsequently steps that may proceed through two pathways, either the electrochemical desorption via Heyrovsky steps (Eqs. (9) and (10)) or the chemical desorption via Tafel step (Eq. (11)).
From experimental results, the HER mechanism can be inferred by Tafel plot derived from the polarization curves[
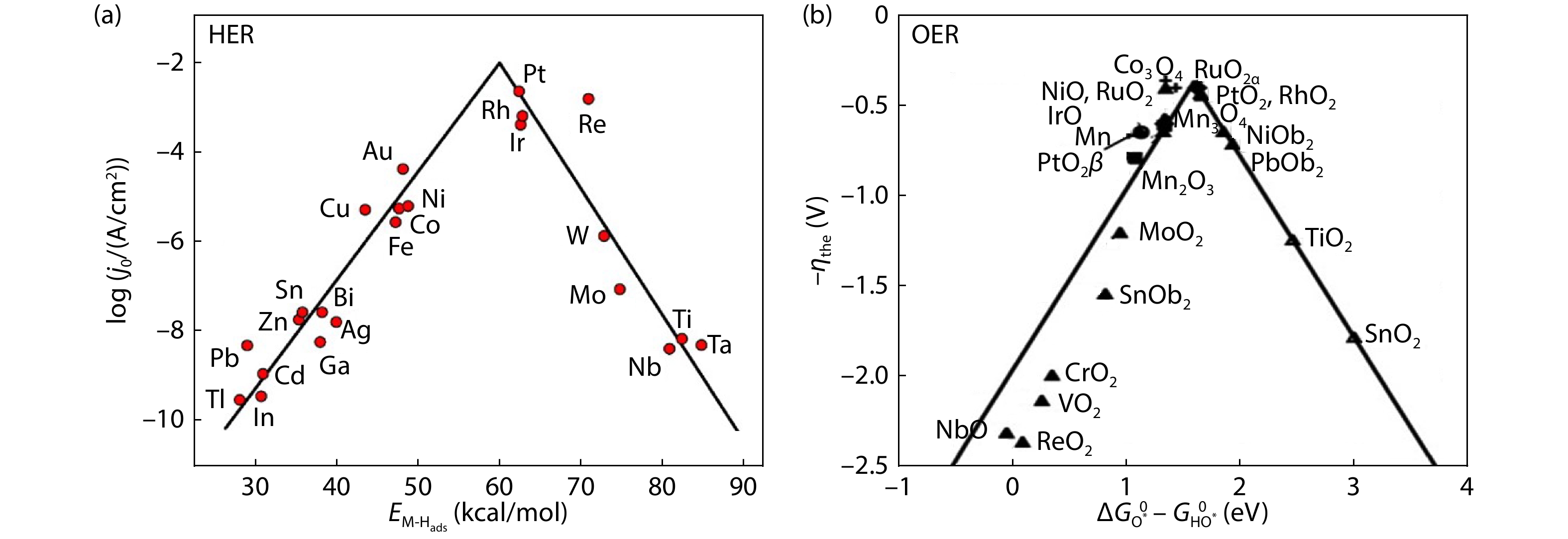
Furthermore, as indicated in Volmer steps and Heyrovsky/Tafel steps, in HER, the chemical adsorption and desorption of H atoms are competing processes. According to the Sabatier principle, an excellent electrocatalyst should possess a suitable binding energy neither too strong nor too weak to not only facilitate the proton-electron-transfer process by adsorbing H* tightly, but also accelerate the release of gaseous H2 via facile bond breaking[
where
![]()
Figure 1.(a) Volcano plot for the HER on metal electrodes in acidic media[
2.3. Reaction pathways of OER
In terms of the OER, the proton coupling four-electron transfer involved process results in its sluggish kinetics and more possible reaction pathways compared with HER. Meanwhile, a well-accepted common sense indicates, in an alkaline electrolyte, a generated O2 molecule inclines to form on a metal oxide/oxyhydroxide, rather than a metal surface or metal compound like phosphide, sulfide, etc[
In acidic electrolyte:
In neutral and alkaline electrolyte:
where * represents the active sites on the electrocatalysts surface, and OH*, O*, and OOH* designate the intermediates adsorbed at the active sites, respectively. Apart from theoretical results mentioned before, some practical parameters are also worth highlighting here for comprehensive evaluation of the ability of a nanostructured electrocatalyst towards overall water splitting[
3. Recent progress of nanostructured bimetallic electrocatalysts towards water splitting
Over the last few decades, the noble metal-based (e.g. Pt, Ir, Ru, Pd, and Rh) electrode have been recognized as the most trusted and reliable catalysts for water splitting which can be readily explained by the “volcano plot”. In terms of HER, the Pt locates at the summit of the HER activity volcano curve, so it can easily be understood why the Pt family materials were the commonly chosen electrode in an acid medium in early practice and research. As for OER, Ir/Ru based metal alloys and compounds due to advantageous electronic structure and suitable redox potential, have been proven as state-of-the-art electrocatalysts with low overpotential and Tafel slope. However, these noble metal family, even occupying the summit of the “volcano curve” naturally, always suffer from extreme scarcity of resource and/or severe corrosion, which disqualifies them as ideal electrodes in scalable applications that required extend working period. Meanwhile, the electrolyzer design for alkaline media offers opportunities for a wide range of bimetallic and/or metal-based multicomponent electrocatalysts serving as high-efficiency, low-cost, and long-term stable electrode. Since then, extensive studies have been devoted to developing strategies for design, fabrication, and improving the performance of nanostructure bimetallic-based electrocatalysts.
Up to now, most of the efforts have been aimed at either tuning the electronic structure, consequently, enhancing the intrinsic activity (higher average turnover frequency), or boosting the accessible active sites on the electrode surface. Commonly utilized strategies for metal-based catalysts, typically, can be classified into three main methods: 1) alloy involving multiple metallic elements with various composition ratios, which increase the intrinsic activity per active site of the electrocatalysts by tuning the electronic structure of the atoms on electrode surface[
3.1. Bimetallic alloying effect
The excellent intrinsic activity due to the inherent electronic structure of platinum group metals (PGMs), excludes the utilization of other non-precious transition metal as candidates for water splitting. However, the comparatively low activity of non-precious metal could be synergistically boosted via alloying materials[
Although the noble metal involved alloying electrode could not only maintain superior activity and stability, but also significantly reduce the cost of electrocatalysts, the researchers still want to realize the noble metal-free electrode with competitive performance[
3.2. Interface effect
If the development of nanostructured bimetallic alloys is recognized as a promising strategy, the formation of interfaces between two distinct components would bring more possibilities for the performance-enhancement of electrocatalysts. Typically, the categories of interfaces in the field of electrocatalysts for water splitting include metal/metal interfaces[
![]()
Figure 2.(Color online) (a) Microstructure and interfaces of the molybdenum sulfide@NPG. (b) HER polarization curves of NPG, MoS2@GCE, and MoS2@NPG[
Apart from localized interfaces between nanoparticles, the interface formed on the current collector surface have also been investigated to explore the possible contribution to the performance of electrocatalysts. For instance, Desmond Ng and co-workers prepared thin film of NiCeOx–Au onto Au-coated GC disk and bare GC disk, respectively to demonstrate the Au support effect[
3.3. Nanostructured effect
Although tuning the electronic structure and thereby enhancing the intrinsic activity is a promising strategy towards improving the catalytic efficiency of electrocatalysts, the catalytic reaction only occurring on the electrocatalyst surface inspires the development of an electrode with nanostructured modification. To date, many template-assisted[
![]()
Figure 3.(Color online) (a) SEM image of an
As an increasing number of research works have starting to concentrate on the role of nanostructuring on mass transport and electrode durability, recently, Song and coworkers revealed more detailed information about that by using assembled striped-pattern superlattices[
![]()
Figure 4.(Color online) (a) SEM image of the large area SP superlattices and inserted photograph of the SP superlattices with dimensions 0.7 × 0.7 cm2. (b) High-magnification SEM image of the long-rang-ordered Pt nanocrystals and TEM image of the nanocrystal superlattice. (c) Polarization curves of Pt SP5, Pt SP10, Pt SP20, Pt SP50, Pt NP film, Pt/C film and Pt foil electrocatalysts, the current density was normalized by geometry area. (d) Stability testing on Pt SP5 for 11 h. (e) Stability testing on Pt NP films. (f) Schematic illustration of the growth of gas bubbles on a flat film electrode. (g) Schematic illustration of the stability difference between flat film and SP electrode. (h–j) Snapshots of digital videos taken during electrolysis at 10 mA/cm2, magnified observations and schematic illustration of single bubble behavior on (h) Pt SP5, (i) Pt SP20 and (j) Pt NP film. (k)
4. Fundamentals of CO2 reduction
Due solely to the final product (H2 or O2) resulting from water splitting, the reaction pathways and the number of intermediates are not that elusive and relatively easy to track down and confirm. Nevertheless, in electroreduction of CO2, the reaction mechanism is far more intricate and are not fully understood. In 2012, the very first observation of a total of 16 different products from CO2 reduction by Jaramillo’s group unveiled the complex and intertwining nature of the CO2 reduction pathways[
The reduction of CO2 on the surface of heterogeneous catalysts involves the transformation of electrons and protons from/to the adsorbed CO2 molecule. As an increasing number of electrons are transferred (from 2 electrons to 12 electrons), numerous possible reaction pathways and increasing intermediates are brought up. Yet, the initiating of CO2RR always accompany the activation of CO2 molecule that is widely recognized as a difficult step requiring large energy input due to the large kinetic barrier. Additionally, the equilibrium potentials of CO2RR are located around 0 V (mostly in the range of –0.2 to 0.2 V vs. RHE), making the HER a competing reaction during activation of CO2 to impair the selectivity of CO2RR. The redox reactions related to the activation of CO2 is considered as the following Eqs. (22)–(25)[
where * represents the active sites on the catalytic surface, while Eq. (25) is the competing hydrogen evolution reaction process. Eqs. (22) and (23) are well-accepted concerted proton-electron transfer reactions (CPET) involving a proton and an electron during the capture of the CO2 molecule. As the 2e– transferred reduced products, the formation of either carbon monoxide (*CO) or formate (HCOO*) is dependent on the form of adsorbed active CO2. Specifically, the bonding strength between the metal and the adsorbed species determines the selectivity in the first step. Metals, like Pb and In, that do not offer a strong M–C bond with CO2 prefer to capture CO2 via O atom and hence favor the formation of formate; while metals like Au and Ag, preferred to bind CO2 via C, are more likely to generate CO. Consequently, *COOH and *OCHO are recognized as the key intermediates for producing CO and HCOOH, respectively[
Additionally, other form of intermediates, like formaldehyde (CH2O), have been reported to produce methanol[
Up to now, 5 possible terminal products in the C1 group, including formate, CO, methanol, and methane, and other typical C2+ group products, such as acetaldehyde, acetic acid, ethylene, ethanol, and glycol, have been proven, and the comprehensive reaction pathways are also elaborated in Fig. 5[
![]()
Figure 5.(Color online) Possible mechanistic pathways of CO2 reduction to C1 and C2 products[
5. Recent progress for nanostructured bimetallic electrocatalysts towards high-performance electrocatalysts for CO2RR
As we discussed in a previous section, the reaction selectively produces formate or CO in the first step depending on how the electrocatalysts combine with CO2 molecules. Only the formation of *COOH intermediate can be further reduced via combining with another proton and electron, yielding CO and H2O. The generated CO paves the way for the formation of hydrocarbons and oxygenates from C1 group products. According to the Gibbs free energy of adsorption towards various intermediates species obtained by DFT, a long-time thought classified the current active metal electrocatalysts for CO2RR into 3 groups based on their selectivity[
5.1. Bimetallic alloy effect
Early theoretical and experimental studies have widely employed pure metal[
![]()
Figure 6.(Color online) (a) Microstructural analysis and bulk compositions of the catalysts. (b) Calculated reaction energy profiles for CO2 electroreduction to form CO (top) and HCOOH (bottom) on the PdSnO2 surface[
In terms of producing CO, Cu is frequently alloyed with other transition metalsin order to achieve high performance[
The direct conversion of CO2 to hydrocarbons or multi-carbon oxygenates presents a striking route for the high energy density chemical fuels. Various Cu-based alloys, such as CuAu[
5.2. Bi-component effect
In addition to utilizing alloying electrocatalysts with varying composition and mixing patterns to directly adjust surface electronic/structure environments, bi-component catalysts, that are able to combine distinct catalytic/electronic properties of each single-component counterpart, have emerged as another pronounced electrocatalyst. Some experiments demonstrated that the enhanced performance towards different hydrocarbons and oxygenates was due to the generated various interfaces. Luc and co-workers reported Ag–Sn bimetallic core-shell structure for selective conversion of CO2 to formic acid (Fig. 7(a))[
![]()
Figure 7.(Color online) (a) A schematic illustration of the Ag3Sn core-shell structure. (b) CO2 reduction Faradaic efficiencies of AgSn/SnO
Besides bi-component electrocatalysts consisting of metal/metal interfaces, other form of combination are also promising for CO2RR. For instance, Guntern and co-workers reported novel nanocrystal/metal-organic framework hybrids, Ag@Al-PMOF, as the electrocatalysts (Fig. 7(h)) to realize the CO2 conversion with relatively high Faradaic efficiency[
Copper involved bi-component tandem electrocatalysts that could conduct two and/or more sequential steps of CO2RR is also a promising strategy to realize targeted C2+ products. For instance, Carlos and co-workers reported the discovery of gold nanoparticles deposited on a polycrystalline copper foil (Au/Cu) as a novel electrocatalyst that is highly active towards formation of alcohols[
5.3. Nanostructured effect
Under certain conditions, the distinct activity and improved selectivity regarding bimetallic electrocatalysts might be attributed to the nanostructuring effect[
![]()
Figure 8.(Color online) (a) Schematic illustration of the structure of Cu nanofoams and the terminal products. (b) Product distribution as a function of applied potential during the electrochemical reduction of CO2[
Apart from directly mediating the morphology of catalysts for higher activity and selectivity, another merit of nanostructured electrocatalysts is its ability to affect the hydrophobicity of the electrode as well as the local pH around the catalyst surface, which results in the inhibition of competitive reaction (HER) and the promotion of CO2RR. For instances, Nam and co-workers reported a superhydrophobic surface of hierarchically structured Cu dendrites that could surprisingly achieve 56% FE for ethylene and 17% for ethanol production, comparing to poor FE of 9% and 4% on a hydrophilic and wettable equivalent[
![]()
Figure 9.(Color online) (a) This is illustrated on a diving bell spider for subaquatic breathing. (b) A hydrophobic dendritic Cu surface for aqueous CO2 reduction. (c) The contact angle measurements of the wettable and hydrophobic dendrite. (d) SEM image of the hydrophobic dendrite. (e) Illustration of the hydrophobic dendrite gaining a solid–liquid interface on the application of a negative potential. (f) CPE product FEs from the wettable and hydrophobic dendrite at various potentials. (g) The proposed role of hydrophobicity in promoting CO2 reduction over proton reduction[
6. Summary and perspectives
The development of highly efficient and durable electrocatalysts is essential for the wide-spread application of various energy conversion systems. To date, bimetallic electrocatalysts with morphology modification in the nano scale have been recognized as a promising options. In this review, fundamental knowledge background and vital concepts in theoretical studies for water splitting and CO2RR are covered firstly to understand the exact process happening on the surface of nanostructured bimetallic electrocatalysts. After that, the dominant advantages of these electrocatalysts was summarized to bimetallic alloying, bi-component interfacing, and nanostructuring, which could result in electronic environment alterations of surface atoms, coupled benefits of two distinct materials regarding different electronic properties, and fine-design structure modifications in the nano scale for fast mass/proton/electron transformation. Despite these significant progresses, there are still many challenges and questions, which must be addressed before the nanostructured bimetallic catalysts can find a wide range of practical uses.
(1) In the current stage of nanostructured bimetallic electrocatalysts developed for water splitting, the in-situ spectroscopic study is extremely lacking. Although the development of DFT calculation already provides a useful theoretical tool to identify and predict the active sites and electronic structure change, the further design and optimization of performance of electrocatalysts is highly reliant on the exact catalytic mechanism on these structure/electronic tuned catalysts where a straightforward observation of catalytic reaction process under various in-situ spectroscopic techniques could be very promising for that goals. Especially for these nanostructured bimetallic electrocatalysts, the fine-tuning structure in nano scale significantly alter the bubbles evolution process for fast kinetics and mass transferring, but, lots of unknown questions, like have these structure modification affected strain or resistance in bulk, is left behind, which need to be unraveled by directly investigation of the structure/bubbles evolution behavior. On the other hand, the precise identification of catalytic sites is essential for unveiling how the interaction between two metals or interfaces between two components boosts the activity of reactions, which can be experimentally verified through in-situ observation in atomic-level.
(2) As for CO2RR, advanced in situ characterization techniques are also necessary for an in-depth understanding of the reaction pathways and to improve the activity and/or selectivity for further rational design of nanostructured bimetallic electrocatalysts. In fact, the in situ FT-IR is already used for unravelling the reaction pathways and intermediates by providing the information about adsorbed carbon species on catalysts[
References
[1] G Fu, J M Lee. Ternary metal sulfides for electrocatalytic energy conversion. J Mater Chem A, 7, 9386(2019).
[2] M Tahir, L Pan, F Idrees et al. Electrocatalytic oxygen evolution reaction for energy conversion and storage: A comprehensive review. Nano Energy, 37, 136(2017).
[3] X Li, X Hao, A Abudula et al. Nanostructured catalysts for electrochemical water splitting: Current state and prospects. J Mater Chem A, 4, 11973(2016).
[4] D Gao, R M Arán-Ais, H S Jeon et al. Rational catalyst and electrolyte design for CO2 electroreduction towards multicarbon products. Nat Catal, 2, 198(2019).
[5] Y Y Birdja, E Pérez-Gallent, M C Figueiredo et al. Advances and challenges in understanding the electrocatalytic conversion of carbon dioxide to fuels. Nat Energy, 4, 732(2019).
[6] Q Shao, P Wang, X Huang. Opportunities and challenges of interface engineering in bimetallic nanostructure for enhanced electrocatalysis. Adv Funct Mater, 29, 1806419(2019).
[7] C Niether, S Faure, A Bordet et al. Improved water electrolysis using magnetic heating of FeC-Ni core-shell nanoparticles. Nat Energy, 3, 476(2018).
[8] Y Wang, D Yan, S El Hankari et al. Recent progress on layered double hydroxides and their derivatives for electrocatalytic water splitting. Adv Sci, 5, 1800064(2018).
[9] M Gong, H Dai. A mini review of NiFe-based materials as highly active oxygen evolution reaction electrocatalysts. Nano Res, 8, 23(2014).
[10] Y Yan, B Y Xia, B Zhao et al. A review on noble-metal-free bifunctional heterogeneous catalysts for overall electrochemical water splitting. J Mater Chem A, 4, 17587(2016).
[11] L Zhang, Z J Zhao, J Gong. Nanostructured materials for heterogeneous electrocatalytic CO2 reduction and their related reaction mechanisms. Angew Chem Int Ed, 56, 11326(2017).
[12] S Nitopi, E Bertheussen, S B Scott et al. Progress and perspectives of electrochemical CO2 reduction on copper in aqueous electrolyte. Chem Rev, 119, 7610(2019).
[13] M Y Gao, C Yang, Q B Zhang et al. Facile electrochemical preparation of self-supported porous Ni-Mo alloy microsphere films as efficient bifunctional electrocatalysts for water splitting. J Mater Chem A, 5, 5797(2017).
[14] C Cheng, F Zheng, C Zhang et al. High-efficiency bifunctional electrocatalyst based on 3D freestanding Cu foam in situ armored CoNi alloy nanosheet arrays for overall water splitting. J Power Sources, 427, 184(2019).
[15] S Ghosh, R N Basu. Multifunctional nanostructured electrocatalysts for energy conversion and storage: current status and perspectives. Nanoscale, 10, 11241(2018).
[16] P Xiao, W Chen, X Wang. A review of phosphide-based materials for electrocatalytic hydrogen evolution. Adv Energy Mater, 5, 1500985(2015).
[17] Y Jiao, Y Zheng, M Jaroniec et al. Design of electrocatalysts for oxygen- and hydrogen-involving energy conversion reactions. Chem Soc Rev, 44, 2060(2015).
[18] Y Zheng, Y Jiao, M Jaroniec et al. Advancing the electrochemistry of the hydrogen-evolution reaction through combining experiment. Angew Chem Int Ed, 54, 52(2015).
[19] J K Nørskov, T Bligaard, A Logadottir et al. Trends in the exchange current for hydrogen evolution. J Electrochem Soc, 152, 23(2005).
[20] T R Cook, D K Dogutan, S Y Reece et al. Solar energy supply and storage for the legacy and nonlegacy worlds. Chem Rev, 110, 6474(2010).
[21] H Dau, C Limberg, T Reier et al. The mechanism of water oxidation: from electrolysis via homogeneous to biological catalysis. ChemCatChem, 2, 724(2010).
[22] L K Wu, W Y Wu, J Xia et al. A nanostructured nickel-cobalt alloy with an oxide layer for an efficient oxygen evolution reaction. J Mater Chem A, 5, 10669(2017).
[23] K Liu, C Zhang, Y Sun et al. High-performance transition metal phosphide alloy catalyst for oxygen evolution reaction. ACS Nano, 12, 158(2018).
[24] C Liang, P Zou, A Nairan et al. Exceptional performance of hierarchical Ni-Fe oxyhydroxide@NiFe alloy nanowire array electrocatalyst for large current density water splitting. Energy Environ Sci, 13, 86(2020).
[25] C C L Mccrory, S Jung, J C Peters et al. Benchmarking heterogeneous electrocatalysts for the oxygen evolution reaction. J Am Chem Soc, 135, 16977(2013).
[26] C Tang, H F Wang, X L Zhu et al. Advances in hybrid electrocatalysts for oxygen evolution reactions: Rational integration of NiFe layered double hydroxides and nanocarbon. Part Part Syst Charact, 33, 473(2016).
[27] I C Man, H Y Su, F Calle-Vallejo et al. Universality in oxygen evolution electrocatalysis on oxide surfaces. ChemCatChem, 3, 1159(2011).
[28] U Y Qazi, C Z Yuan, N Ullah et al. One-step growth of iron-nickel bimetallic nanoparticles on FeNi alloy foils: Highly efficient advanced electrodes for the oxygen evolution reaction. ACS Appl Mater Interfaces, 9, 28627(2017).
[29] Q Hu, X Liu, B Zhu et al. Crafting MoC2-doped bimetallic alloy nanoparticles encapsulated within N-doped graphene as roust bifunctional electrocatalysts for overall water splitting. Nano Energy, 50, 212(2018).
[30] J Fan, Z Chen, H Shi et al. In situ grown, self-supported iron-cobalt-nickel alloy amorphous oxide nanosheets with low overpotential toward water oxidation. Chem Commun, 52, 4290(2016).
[31] Y Jin, X Yue, C Shu et al. Three-dimensional porous MoNi4 networks constructed by nanosheets as bifunctional electrocatalysts for overall water splitting. J Mater Chem A, 5, 2508(2017).
[32] Y Ma, X Dai, M Liu et al. Strongly coupled feni alloys/NiFe2O4@carbonitride layers-assembled microboxes for enhanced oxygen evolution reaction. ACS Appl Mater Interfaces, 8, 34396(2016).
[33] D Y Chung, P P Lopes, P F Martins et al. Dynamic stability of active sites in hydr(oxy)oxides for the oxygen evolution reaction. Nat Energy, 5, 222(2020).
[34] S Saha, A K Ganguli. FeCoNi alloy as noble metal-free electrocatalyst for oxygen evolution reaction (OER). ChemistrySelect, 2, 1630(2017).
[35] P Zhang, L Li, D Nordlund et al. Dendritic core-shell nickel-iron-copper metal/metal oxide electrode for efficient electrocatalytic water oxidation. Nat Commun, 9, 381(2018).
[36] J Zhang, Q Shao, P Wang et al. Catalytic hydrogen production by janus CuAg nanostructures. ChemNanoMat, 4, 477(2018).
[37] Q Song, Z Xue, C Liu et al. A general strategy to optimize gas evolution reaction via assembled striped-pattern superlattices. J Am Chem Soc, 142, 1857(2019).
[38] F A Garcés-Pineda, M Blasco-Ahicart, D Nieto-Castro et al. Direct magnetic enhancement of electrocatalytic water oxidation in alkaline media. Nat Energy, 4, 519(2019).
[39] P Ganesan, A Sivanantham, S Shanmugam. Nanostructured nickel-cobalt-titanium alloy grown on titanium substrate as efficient electrocatalyst for alkaline water electrolysis. ACS Appl Mater Interfaces, 9, 12416(2017).
[40] X Zhu, T Jin, C Tian et al. In situ coupling strategy for the preparation of FeCo alloys and Co4N hybrid for highly efficient oxygen evolution. Adv Mater, 29, 1704091(2017).
[41] R Subbaraman, D Tripkovic, K C Chang et al. Trends in activity for the water electrolyser reactions on 3d M(Ni,Co,Fe,Mn) hydr(oxy)oxide catalysts. Nat Mater, 11, 550(2012).
[42] F Wang, K Kusada, D Wu et al. Solid-solution alloy nanoparticles of the immiscible iridium–copper system with a wide composition range for enhanced electrocatalytic applications. Angew Chem Int Ed, 57, 4505(2018).
[43] Y Zhao, M Luo, S Chu et al. 3D nanoporous iridium-based alloy microwires for efficient oxygen evolution in acidic media. Nano Energy, 59, 146(2019).
[44] S Gupta, L Qiao, S Zhao et al. Highly active and stable graphene tubes decorated with FeCoNi alloy nanoparticles via a template-free graphitization for bifunctional oxygen reduction and evolution. Adv Energy Mater, 6, 1601198(2016).
[45] X Cui, P Ren, D Deng et al. Single layer graphene encapsulating non-precious metals as high-performance electrocatalysts for water oxidation. Energy Environ Sci, 9, 123(2016).
[46] Y Yang, Z Lin, S Gao et al. Tuning electronic structures of nonprecious ternary alloys encapsulated in graphene layers for optimizing overall water splitting activity. ACS Catal, 7, 469(2017).
[47] C Wang, H Yang, Y Zhang et al. NiFe alloy nanoparticles with hcp crystal structure stimulate superior oxygen evolution reaction electrocatalytic activity. Angew Chem Int Ed, 58, 6099(2019).
[48] S Anantharaj, K Karthick, M Venkatesh et al. Enhancing electrocatalytic total water splitting at few layer Pt-NiFe layered double hydroxide interfaces. Nano Energy, 39, 30(2017).
[49] Q Xiang, F Li, W Chen et al. In situ vertical growth of Fe-Ni layered double-hydroxide arrays on Fe-Ni alloy foil: Interfacial layer enhanced electrocatalyst with small overpotential for oxygen evolution reaction. ACS Energy Lett, 3, 2357(2018).
[50] X Ge, L Chen, L Zhang et al. Nanoporous metal enhanced catalytic activities of amorphous molybdenum sulfide for high-efficiency hydrogen production. Adv Mater, 26, 3100(2014).
[51] J Fester, A Makoveev, D Grumelli et al. The structure of the cobalt oxide/au catalyst interface in electrochemical water splitting. Angew Chem, 130, 12069(2018).
[52] J W D Ng, M García-Melchor, M Bajdich et al. Gold-supported cerium-doped NiO
[53] P Chakthranont, J Kibsgaard, A Gallo et al. Effects of gold substrates on the intrinsic and extrinsic activity of high-loading nickel-based oxyhydroxide oxygen evolution catalysts. ACS Catal, 7, 5399(2017).
[54] S Zhao, R Jin, H Abroshan et al. Gold nanoclusters promote electrocatalytic water oxidation at the nanocluster/CoSe2 Interface. J Am Chem Soc, 139, 1077(2017).
[55] Y Gorlin, C J Chung, J D Benck et al. Understanding interactions between manganese oxide and gold that lead to enhanced activity for electrocatalytic water oxidation. J Am Chem Soc, 136, 4920(2014).
[56] S Ci, S Mao, Y Hou et al. Rational design of mesoporous NiFe-alloy-based hybrids for oxygen conversion electrocatalysis. J Mater Chem A, 3, 7986(2015).
[57] L Wen, Z Wang, Y Mi et al. Designing heterogeneous 1D nanostructure arrays based on AAO templates for energy applications. Small, 11, 3408(2015).
[58] L Wen, R Xu, C Cui et al. Template-guided programmable janus heteronanostructure arrays for efficient plasmonic photocatalysis. Nano Lett, 18, 4914(2018).
[59] J Kang, A Hirata, H J Qiu et al. Self-grown oxy-hydroxide@nanoporous metal electrode for high-performance supercapacitors. Adv Mater, 26, 269(2014).
[60] L Yu, H Zhou, J Sun et al. Cu nanowires shelled with NiFe layered double hydroxide nanosheets as bifunctional electrocatalysts for overall water splitting. Energy Environ Sci, 10, 1820(2017).
[61] X Zhu, R Amal, X Lu. N,P co-coordinated manganese atoms in mesoporous carbon for electrochemical oxygen reduction. Small, 15, 1804524(2019).
[62] X Zhu, X Tan, K H Wu et al. N,P co-coordinated Fe species embedded in carbon hollow spheres for oxygen electrocatalysis. J Mater Chem A, 7, 14732(2019).
[63] Y Dang, J He, T Wu et al. Constructing bifunctional 3D holey and ultrathin CoP nanosheets for efficient overall water splitting. ACS Appl Mater Interfaces, 11, 29879(2019).
[64] M Li, T Liu, X Bo et al. A novel flower-like architecture of FeCo@NC-functionalized ultra-thin carbon nanosheets as a highly efficient 3D bifunctional electrocatalyst for full water splitting. J Mater Chem A, 5, 5413(2017).
[65] X Zhu, D Zhang, C J Chen et al. Harnessing the interplay of Fe–Ni atom pairs embedded in nitrogen-doped carbon for bifunctional oxygen electrocatalysis. Nano Energy, 71, 104597(2020).
[66] J Jin, J Yu, D Guo et al. A hierarchical Z-scheme CdS-WO3 photocatalyst with enhanced CO2 reduction activity. Small, 11, 5262(2015).
[67] A S Bandarenka, AS Varela, M Karamad et al. Design of an active site towards optimal electrocatalysis: Overlayers, surface alloys and near-surface alloys of Cu/Pt(111). Angew Chem Int Ed, 51, 11845(2012).
[68] M S Faber, R Dziedzic, M A Lukowski et al. High-performance electrocatalysis using metallic cobalt pyrite (CoS2) micro- and nanostructures. J Am Chem Soc, 136, 10053(2014).
[69] C L Huang, X F Chuah, C T Hsieh et al. NiFe alloy nanotube arrays as highly efficient bifunctional electrocatalysts for overall water splitting at high current densities. ACS Appl Mater Interfaces, 11, 24096(2019).
[70] H Xu, Z X Shi, Y X Tong et al. Porous microrod arrays constructed by carbon-confined NiCo@NiCoO2 core@shell nanoparticles as efficient electrocatalysts for oxygen evolution. Adv Mater, 30, 1705442(2018).
[71] Y Hou, S Cui, Z Wen et al. Strongly coupled 3D hybrids of n-doped porous carbon nanosheet/CoNi alloy-encapsulated carbon nanotubes for enhanced electrocatalysis. Small, 11, 5940(2015).
[72] X Zhang, o Y F Zhao, o Y X Zhao et al. A simple synthetic strategy toward defect-rich porous monolayer NiFe-layered double hydroxide nanosheets for efficient electrocatalytic water oxidation. Adv Energy Mater, 9, 1900881(2019).
[73] E J Popczun, J R McKone, C G Read et al. Nanostructured nickel phosphide as an electrocatalyst for the hydrogen evolution reaction. J Am Chem Soc, 135, 9267(2013).
[74] M Gao, W Sheng, Z Zhuang et al. Efficient water oxidation using nanostructured α-nickel-hydroxide as an electrocatalyst. J Am Chem Soc, 136, 7077(2014).
[75] S Kim, C Ahn, Y Cho et al. Suppressing buoyant force: New avenue for long-term durability of oxygen evolution catalysts. Nano Energy, 54, 184(2018).
[76] K P Kuhl, E R Cave, D N Abram et al. New insights into the electrochemical reduction of carbon dioxide on metallic copper surfaces. Energy Environ Sci, 5, 7050(2012).
[77] H Z Yang, L Shang, Q H Zhang et al. A universal ligand mediated method for large scale synthesis of transition metal single atom catalysts. Nat Commun, 10, 4585(2019).
[78] W Wang, L Shang, G J Chang et al. Intrinsic carbon-defect-driven electrocatalytic reduction of carbon dioxide. Adv Mater, 31, 1808276(2019).
[79] J S Yoo, R Christensen, T Vegge et al. Theoretical Insight into the trends that guide the electrochemical reduction of carbon dioxide to formic acid. ChemSusChem, 9, 358(2016).
[80] A Bagger, W Ju, A S Varela et al. Electrochemical CO2 reduction: A classification problem. ChemPhysChem, 18, 3266(2017).
[81] A A Peterson, J K Nørskov. Activity descriptors for CO2 electroreduction to methane on transition-metal catalysts. J Phys Chem Lett, 3, 251(2012).
[82] K P Kuhl, T Hatsukade, E R Cave et al. Electrocatalytic conversion of carbon dioxide to methane and methanol on transition metal surfaces. J Am Chem Soc, 136, 14107(2014).
[83] A A Peterson, F Abild-Pedersen, F Studt et al. How copper catalyzes the electroreduction of carbon dioxide into hydrocarbon fuels. Energy Environ Sci, 3, 1311(2010).
[84] H Ooka, M C Figueiredo, M T M Koper. Competition between hydrogen evolution and carbon dioxide reduction on copper electrodes in mildly acidic media. Langmuir, 33, 9307(2017).
[85] J He, N J J Johnson, A Huang et al. Electrocatalytic alloys for CO2 reduction. ChemSusChem, 11, 48(2018).
[86] D Chen, g Y L Wang, u D Y Liu et al. Surface composition dominates the electrocatalytic reduction of CO2 on ultrafine CuPd nanoalloys. Carbon Energy, 2, 443(2020).
[87] K J P Schouten, Y Kwon, C J M Van Der Ham et al. A new mechanism for the selectivity to C1 and C2 species in the electrochemical reduction of carbon dioxide on copper electrodes. Chem Sci, 2, 1902(2011).
[88] Y Hori, R Takahashi, Y Yoshinami et al. Electrochemical reduction of CO at a copper electrode. J Phys Chem B, 101, 7075(1997).
[89] R L Cook, R C Macduff, A F Sammells. Evidence for formaldehyde, formic acid, and acetaldehyde as possible intermediates during electrochemical carbon dioxide reduction at copper. J Electrochem Soc, 136, 1982(1989).
[90] J H Montoya, C Shi, K Chan et al. Theoretical insights into a CO dimerization mechanism in CO2 electroreduction. J Phys Chem Lett, 6, 2032(2015).
[91] K J P Schouten, Z Qin, E P Gallent et al. Two pathways for the formation of ethylene in CO reduction on single-crystal copper electrodes. J Am Chem Soc, 134, 9864(2012).
[92] Q Fan, M Zhang, M Jia et al. Electrochemical CO2 reduction to C2+ species: Heterogeneous electrocatalysts, reaction pathways, and optimization strategies. Mater Today Energy, 10, 280(2018).
[93] K U D Calvinho, A B Laursen, K M K Yap et al. Selective CO2 reduction to C3 and C4 oxyhydrocarbons on nickel phosphides at overpotentials as low as 10 mV. Energy Environ Sci, 11, 2550(2018).
[94] R Kortlever, I Peters, C Balemans et al. Palladium-gold catalyst for the electrochemical reduction of CO2 to C1-C5 hydrocarbons. Chem Commun, 52, 10229(2016).
[95] D A Torelli, S A Francis, J C Crompton et al. Nickel-gallium-catalyzed electrochemical reduction of CO2 to highly reduced products at low overpotentials. ACS Catal, 6, 2100(2016).
[96] A J Garza, A T Bell, M Head-Gordon. Mechanism of CO2 reduction at copper surfaces: Pathways to C2 products. ACS Catal, 8, 1490(2018).
[97] J Resasco, L D Chen, E Clark et al. Promoter effects of alkali metal cations on the electrochemical reduction of carbon dioxide. J Am Chem Soc, 139, 11277(2017).
[98] I Ledezma-Yanez, E P Gallent, M T M Koper et al. Structure-sensitive electroreduction of acetaldehyde to ethanol on copper and its mechanistic implications for CO and CO2 reduction. Catal Today, 262, 90(2016).
[99] E L Clark, A T Bell. Direct observation of the local reaction environment during the electrochemical reduction of CO2. J Am Chem Soc, 140, 7012(2018).
[100] R Kortlever, J Shen, K J P Schouten et al. Catalysts and reaction pathways for the electrochemical reduction of carbon dioxide. J Phys Chem Lett, 6, 4073(2015).
[101] W Tang, A A Peterson, A S Varela et al. The importance of surface morphology in controlling the selectivity of polycrystalline copper for CO2 electroreduction. Phys Chem Chem Phys, 14, 76(2012).
[102] A Loiudice, P Lobaccaro, E A Kamali et al. Tailoring copper nanocrystals towards C2 products in electrochemical CO2 reduction. Angew Chem, 128, 5883(2016).
[103] M Ma, B J Trześniewski, J Xie et al. Selective and efficient reduction of carbon dioxide to carbon monoxide on oxide-derived nanostructured silver electrocatalysts. Angew Chem, 128, 9900(2016).
[104] B A Rosen, A Salehi-khojin, M R Thorson et al. Ionic liquid-mediated selective conversion of CO2 to CO at low overpotentials. Science, 334, 643(2011).
[105] M Liu, Y Pang, B Zhang et al. Enhanced electrocatalytic CO2 reduction via field-induced reagent concentration. Nature, 537, 382(2016).
[106] M Asadi, K Kim, C Liu et al. Nanostructured transition metal dichalcogenide electrocatalysts for CO2 reduction in ionic liquid. Science, 353, 467(2016).
[107] S Gao, Y Lin, X Jiao et al. Partially oxidized atomic cobalt layers for carbon dioxide electroreduction to liquid fuel. Nature, 529, 68(2016).
[108] J L White, M F Baruch, J E Pander et al. Light-driven heterogeneous reduction of carbon dioxide: Photocatalysts and photoelectrodes. Chem Rev, 115, 12888(2015).
[109] S Y Choi, S K Jeong, H J Kim et al. Electrochemical reduction of carbon dioxide to formate on tin-lead alloys. ACS Sustain Chem Eng, 4, 1311(2016).
[110] C H Lee, M W Kanan. Controlling H+ vs CO2 reduction selectivity on Pb electrodes. ACS Catal, 5, 465(2015).
[111] R L MacHunda, H Ju, J Lee. Electrocatalytic reduction of CO2 gas at Sn based gas diffusion electrode. Curr Appl Phys, 11, 986(2011).
[112] W Luc, C Collins, S Wang et al. Ag-Sn bimetallic catalyst with a core-shell structure for CO2 reduction. J Am Chem Soc, 139, 1885(2017).
[113] X Bai, W Chen, C Zhao et al. Exclusive formation of formic acid from CO2 electroreduction by a tunable Pd-Sn alloy. Angew Chem Int Ed, 56, 12219(2017).
[114] R Kortlever, I Peters, S Koper et al. Electrochemical CO2 reduction to formic acid at low overpotential and with high faradaic efficiency on carbon-supported bimetallic Pd-Pt nanoparticles. ACS Catal, 5, 3916(2015).
[115] C Hahn, D N Abram, H A Hansen et al. Synthesis of thin film AuPd alloys and their investigation for electrocatalytic CO2 reduction. J Mater Chem A, 3, 20185(2015).
[116] X Min, M W Kanan. Pd-catalyzed electrohydrogenation of carbon dioxide to formate: High mass activity at low overpotential and identification of the deactivation pathway. J Am Chem Soc, 137, 4701(2015).
[117] S G da Silva, J C M Silva, G S Buzzo et al. PdAu/C electrocatalysts as anodes for direct formate fuel cell. Electrocatalysis, 6, 442(2015).
[118] Z Xu, E Lai, Y Shao-Horn et al. Compositional dependence of the stability of AuCu alloy nanoparticles. Chem Commun, 48, 5626(2012).
[119] P Hirunsit. Electroreduction of carbon dioxide to methane on copper, copper-silver, and copper-gold catalysts: A DFT study. J Phys Chem C, 117, 8262(2013).
[120] D Kim, J Resasco, Y Yu et al. Synergistic geometric and electronic effects for electrochemical reduction of carbon dioxide using gold-copper bimetallic nanoparticles. Nat Commun, 5, 4948(2014).
[121] S Rasul, D H Anjum, A Jedidi et al. A highly selective copper-indium bimetallic electrocatalyst for the electrochemical reduction of aqueous CO2 to CO. Angew Chem, 127, 2174(2015).
[122] S Sarfraz, AT Garcia-Esparza, A Jedidi et al. Cu-Sn bimetallic catalyst for selective aqueous electroreduction of CO2 to CO. ACS Catal, 6, 2842(2016).
[123] M Li, J Wang, P Li et al. Mesoporous palladium-copper bimetallic electrodes for selective electrocatalytic reduction of aqueous CO2 to CO. J Mater Chem A, 4, 4776(2016).
[124] Z Yin, D Gao, S Yao et al. Highly selective palladium-copper bimetallic electrocatalysts for the electrochemical reduction of CO2 to CO. Nano Energy, 27, 35(2016).
[125] D Kim, C Xie, N Becknell et al. Electrochemical activation of CO2 through atomic ordering transformations of AuCu nanoparticles. J Am Chem Soc, 139, 8329(2017).
[126] M Bernal, A Bagger, F Scholten et al. CO2 electroreduction on copper-cobalt nanoparticles: Size and composition effect. Nano Energy, 53, 27(2018).
[127] D Chen, Q Yao, P Cui et al. Tailoring the selectivity of bimetallic copper-palladium nanoalloys for electrocatalytic reduction of CO2 to CO. ACS Appl. Energy Mater, 1, 883(2018).
[128] Y Hori, I Takahashi, O Koga et al. Selective formation of C2 compounds from electrochemical reduction of CO2 at a series of copper single crystal electrodes. J Phys Chem B, 106, 15(2002).
[129] F Jia, X Yu, L Zhang. Enhanced selectivity for the electrochemical reduction of CO2 to alcohols in aqueous solution with nanostructured Cu-Au alloy as catalyst. J Power Sources, 252, 85(2014).
[130] X Guo, Y Zhang, C Deng et al. Composition dependent activity of Cu-Pt nanocrystals for electrochemical reduction of CO2. Chem Commun, 51, 1345(2015).
[131] D Ren, B S H Ang, B S Yeo. Tuning the selectivity of carbon dioxide electroreduction toward ethanol on oxide-derived Cu
[132] E L Clark, C Hahn, T F Jaramillo et al. Electrochemical CO2 reduction over compressively strained CuAg surface alloys with enhanced multi-carbon oxygenate selectivity. J Am Chem Soc, 139, 15848(2017).
[133] S Ma, M Sadakiyo, M Heim et al. Electroreduction of carbon dioxide to hydrocarbons using bimetallic Cu-Pd catalysts with different mixing patterns. J Am Chem Soc, 139, 47(2017).
[134] D Gao, Y Zhang, Z Zhou et al. Enhancing CO2 electroreduction with the metal-oxide interface. J Am Chem Soc, 139, 5652(2017).
[135] C Rogers, W S Perkins, G Veber et al. Synergistic enhancement of electrocatalytic CO2 reduction with gold nanoparticles embedded in functional graphene nanoribbon composite electrodes. J Am Chem Soc, 139, 4052(2017).
[136] S Lee, G Park, J Lee. Importance of Ag-Cu biphasic boundaries for selective electrochemical reduction of CO2 to ethanol. ACS Catal, 7, 8594(2017).
[137] J Huang, M Mensi, E Oveisi et al. Structural sensitivities in bimetallic catalysts for electrochemical CO2 reduction revealed by Ag-Cu nanodimers. J Am Chem Soc, 141, 2490(2019).
[138] Y T Guntern, J R Pankhurst, J Vávra et al. Nanocrystal/metal–organic framework hybrids as electrocatalytic platforms for CO2 conversion. Angew Chem Int Ed, 58, 12632(2019).
[139] J Yuan, M P Yang, W Y Zhi et al. Efficient electrochemical reduction of CO2 to ethanol on Cu nanoparticles decorated on N-doped graphene oxide catalysts. J CO2 Util, 33, 452(2019).
[140] G M Carlos, R C Etosha, A N Stephanie et al. Improved CO2 reduction activity towards C2+ alcohols on a tandem gold on copper electrocatalyst. Nat Catal, 1, 764(2018).
[141] B O Peter, W Patrick, M B Tania et al. Cascade reaction in nanozymes: spatially separated active sites inside Ag-core-porous-Cu-shell nanoparticles for multistep carbon dioxide reduction to higher organic molecules. J Am Chem Soc, 141, 36(2019).
[142] X L Wang, Araújo J F de, W Ju et al. Mechanistic reaction pathways of enhanced ethylene yields during electroreduction of CO2-CO co-feeds on Cu and Cu-tandem electrocatalysts. Nat Nanotechnol, 14, 1063(2019).
[143] Z Haochen, C Xiaoxia, G C Jingguang et al. Computational and experimental demonstrations of one-pot tandem catalysis for electrochemical carbon dioxide reduction to methane. Nat Commun, 10, 3340(2019).
[144] A S Varela, C Schlaup, Z P Jovanov et al. CO2 electroreduction on well-defined bimetallic surfaces: Cu overlayers on Pt(111) and Pt(211). J Phys Chem C, 117, 20500(2013).
[145] S Sen, D Liu, G T R Palmore. Electrochemical reduction of CO2 at copper nanofoams. ACS Catal, 4, 3091(2014).
[146] F S Roberts, K P Kuhl, A Nilsson. High selectivity for ethylene from carbon dioxide reduction over copper nanocube electrocatalysts. Angew Chem, 127, 5268(2015).
[147] R Reske, M Duca, M Oezaslan et al. Controlling catalytic selectivities during CO2 electroreduction on thin Cu metal overlayers. J Phys Chem Lett, 4, 2410(2013).
[148] D Wakerley, S Lamaison, F Ozanam et al. Bio-inspired hydrophobicity promotes CO2 reduction on a Cu surface. Nat Mater, 18, 1222(2019).
[149] J Liu, J Fu, Y Zhou et al. Controlled synthesis of EDTA modified porous hollow copper microspheres for high-efficiency conversion of CO2 to multi-carbon products. Nano Lett, 20, 7(2020).
[150] M Dunwell, Q Lu, J M Heyes et al. The central role of bicarbonate in the electrochemical reduction of carbon dioxide on gold. J Am Chem Soc, 139, 3774(2017).
[151] S Zhu, B Jiang, W B Cai et al. Direct observation on reaction intermediates and the role of bicarbonate anions in CO2 electrochemical reduction reaction on Cu surfaces. J Am Chem Soc, 139, 15664(2017).
[152] A Wuttig, Y Yoon, J Ryu et al. Bicarbonate is not a general acid in Au-catalyzed CO2 electroreduction. J Am Chem Soc, 139, 17109(2017).
Set citation alerts for the article
Please enter your email address



