- Spectroscopy and Spectral Analysis
- Vol. 40, Issue 11, 3644 (2020)
Abstract
Introduction
Nanotechnology is able to monitor, measure, process and manufacture objects on a nanometer scale. The application of nanoparticles (NPs) can be related to many fields such as medical, food, environmental studies, electronics production, power generation and agriculture. Recently, many nanomaterials are produced with the help of this emerging technology that occupies an important place in scientific research[
Selenium in metallic forms plays a vital functions in the human body by improving the action of enzymes such as glutathione peroxidase and selenium-enzymes that defend the body against diseases associated with immunity[
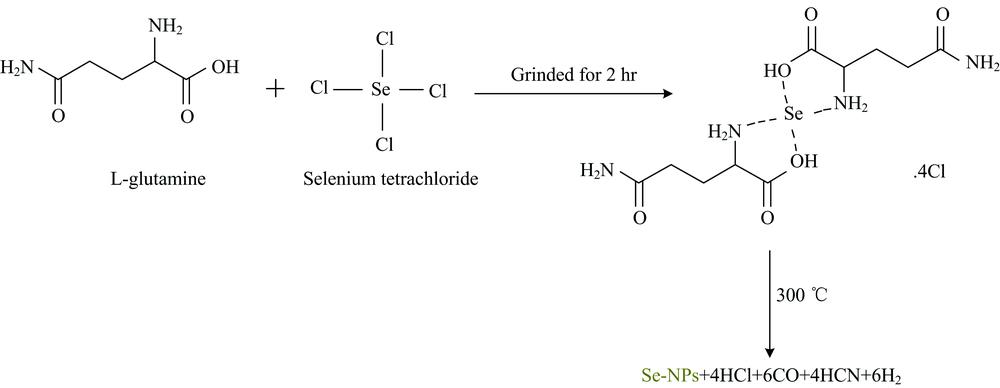
Herein, this present study demonstrate that preparation of Se-NPs. It was synthesized through thermal decomposition Se(Ⅳ) L-glutamine precursor at 300 ℃. The synthesized Se-NPs was characterized using FTIR, Raman, UV-Vis, XRD, SEM, and TEM. It exhibits a spherical shape with an average diameter between 5-10 nm, which was confirmed by TEM analysis.
1 Experimental
1.1 Materials and spectroscopic measurements
All materials for synthesis were commercially available and used as received without further purifications.
The micro analytical were measured using a Perkin Elmer CHN 2400. Infrared spectra were recorded on Bruker FTIR Spectrophotometer (4 000~400 cm-1). Raman laser spectra were measured on the Bruker FT Raman with laser 50 mW. The electronic spectra was scanned using UV2 Unicam UV/Vis Spectrophotometer. Scanning electron microscopy images were taken in Quanta FEG 250 equipment. X-ray diffraction patterns were recorded on X ’Pert PRO PANanalytical X-ray powder diffraction, target copper with secondary monochromate. Transmission electron microscopy images (TEM) were performed using JEOL 100s microscopy.
1.2 Synthesis of Se-NPs
SeCl4 (1 mmole) and L-glutamine(2 mmole) were grinded separately for 10 min in an agate mortar. The fine powder reactants were mixed and grinded for 2 hr. The brown solid product was annealed in a muffle furnace at 300 ℃ for 3 hr in oxygen atmosphere. Se NPs were kept in a closed glass bottle until the beginning of the spectroscopic and physical analyses.
2 Results and discussions
Eco-friendly synthesis of Se-NPs was associated by a thermal decomposition method using the Se(Ⅳ) L-glutamine complexity as a green precursor. The nano-selenium was well characterized by FT-IR, UV-Vis spectroscopy, XRD, SEM and TEM techniques.
FTIR of Se(Ⅳ) L-glutamine precursor and Se-NPs spectra with frequency range from 4 000~4 000 cm-1were displayed in (Fig.1). Figure 1(a) refer to the FT-IR spectrum of Se(Ⅳ) L-glutamine precursor. This spectrum has three distinguish bands at 3 174, 1 589, and 1 110 cm-1 attributed to —NH2 frequencies[
![]()
Figure 1.Chemical synthesized of Se-NPs by thermal decomposition of Se(Ⅳ) L-glutamine precursor
![]()
Figure 2.Raman spectrum of as prepared Se-NPs
The most noticeable characteristic of nanoparticles is their color change in different sizes. As the size changes, the color of the formed particles will also change; absorption in the visible area of the spectrum. Consequently, the UV-Vis spectrum is the most important method for identifying and characterizing nanoparticles[
![]()
Figure 3.UV-Vis spectra of: (a) Se(Ⅳ) L-glutamine precursor and (b) as prepared Se-NPs
Both XRD diffraction patterns of Se(Ⅳ) L-glutamine precursor and prepared Se-NPsare shown in Fig.4. The X-ray spectrum of Se(Ⅳ) L-glutamine precursor [Fig.4(a)] includes a number of peaks corresponding to both L-glutamine and also the characteristic peak of nano-selenium at 2θ=29° due to 101 crystal plane. The crystallinity nature of synthetic Se-NPs (Fig.4) was checked using X-ray powder diffraction patterns. The Se-NPs resulted by thermal decomposition of Se(Ⅳ) L-glutamine precursor has a crystalline shape and the distinguish diffraction patterns have been indexed as 23.36°, 29.58°, 41.18°, 43.47°, 45.28°, 47.88°, 51.33°, 55.89°, 57.05°, 61.29°, 65.06°, 67.67° and 70.94°, which assigned to 100, 101, 110, 012, 111, 200, 201, 003, 112, 013, 120, 211 and 113 crystal planes, respectively, accordingly with JCPDS 86-2246[
(1)
![]()
Figure 4.XRD spectra of: (a) Se(Ⅳ) L-glutamine precursor and (b) as prepared Se-NPs
Scanning electron microscopy technique is used for examining the surface morphology of prepared Se-NPs. SEM photo of synthesized nanoselenium show that it is a spherical shape, well distributed and homogeneous [Fig.5(a)]. Figure 5(b) show TEM image of chemically synthesized Se-NPs. This image describe that the shape of nano-selenium is spherical or semi-spherical with sizes presence between 5~10 nm, which is in agreement with the SEM analysis. Comparing this outcome data with those generated by other published articles[
![]()
Figure 5.Micrographs (a) SEM and (b) TEM of Se-NPs obtained by thermal decomposition of Se(Ⅳ) L-glutamine precursor
Image Infomation Is Not Enable3 Conclusions
Herein, the pure and homogeneous spherical-like Se-NPs with an average size between 5~10 nm were successfully prepared by thermal decomposition of Se(Ⅳ) L-glutamine precursor at 300 ℃. This method is green, friendly, simple and conform for the preparation of nanoselenium.
References
[1] L Ma, J Liu. iScience, 23, 100815(2020).
[2] G Gumbs, N Do T, H Shih P et al. Physica E: Low-dimensional Systems and Nanostructures, 118, 113894(2020).
[3] J L García-Fierro, A Solana, D López-Díaz et al. J. Luminescence, 219, 116954(2020).
[4] C Okumah V, A Odeniyi M, B C Adebayo-Tayo et al. Sustainable Chemistry and Pharmacy, 15, 100197(2020).
[5] M Do, R George, K Stinson. Biochemistry and Biophysics Reports, 22, 100745(2020).
[6] H Adineh, M Harsij, G Kanani H. Aquaculture, 52115, 734942(2020).
[7] N Manjunathan K, S Paul, S Alotaibi. Applied Surface Science, 424, 330(2017).
[8] R Mo, J Li, Y Tang et al. Appl. Surface Sci, 47615, 552(2019).
[9] V Goia D, A Kumar, I Sevonkaev. Journal of Colloid and Interface Science, 41615, 119(2014).
[10] P You, M Song, B Yu et al. New J. Chem, 40, 1118(2016).
[11] J Zhang, Y Zhang S, Y Wang H et al. Materials Letters, 58, 2590(2004).
[12] H Yu, H Wang, J Zhang. Free Radic. Biol. Med, 42, 1524(2007).
[13] A Husen, S Siddiqi K. J. Nanobiotechnology, 12, 28(2014).
[14] J Fesharaki P, P Nazari, M Shakibaie et al. Braz. J. Microbiol, 41, 461(2010).
[15] S Rajan, L Lortie, D Gould W et al. Appl. Environ. Microbiol, 58, 4042(1992).
[16] K S Siddiqi, A Husen. Nano Res. Lett, 9, 229(2014).
[17] N Sharma, R Prakash, T Prakash N et al. Biotechnol. Lett, 31, 1857(2009).
[18] Q Jiang, L Yue, W Chen et al. Int. J. Biol. Macromolecules, 11415, 751(2018).
[19] X Yang, H Zhu, M Sun. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 320, 74(2008).
[20] K Nakamoto. Infrared and Raman Spectra of Inorganic and Coordination Compounds, Fourth ed., Wiley, New York(1986).
[21] G Guisbiers, V Overschelde O. Opt. Laser Technol, 73, 156(2015).
[22] Y Golkari, H Forootanfar, M Shakibaie et al. Journal of Trace Elements in Medicine and Biology, 29, 235(2015).
[23] G Krithiga, S Kumar B, T Hemalatha et al. Acta Metallurgica Sinica (English Letters), 27, 1152(2014).
[24] T Pradeep, B Nair. Crystal Growth & Design, 2, 293(2002).
[25] R Stock S, D Cullity B. Elements of X-Ray Diffraction, 3rd ed., New York: Prentice Hall, 389(2001).
Set citation alerts for the article
Please enter your email address



