[1] Shackley D C, Whitehurst C, Clarke N W et al. Photodynamic therapy[J]. Journal of the Royal Society of Medicine, 92, 562-565(1999).
[3] Kessel D. Photodynamic therapy: a brief history[J]. Journal of Clinical Medicine, 8, 1581(2019).
[4] Babilas P, Schreml S, Landthaler M et al. Photodynamic therapy in dermatology: state-of-the-art[J]. Photodermatology, Photoimmunology & Photomedicine, 26, 118-132(2010).
[5] Majiya H, Adeyemi O O, Herod M et al. Photodynamic inactivation of non-enveloped RNA viruses[J]. Journal of Photochemistry and Photobiology B: Biology, 189, 87-94(2018).
[7] Fan W P, Huang P, Chen X Y. Overcoming the Achilles' heel of photodynamic therapy[J]. Chemical Society Reviews, 45, 6488-6519(2016).
[8] Wei C, Jun Z. Using nanoparticles to enable simultaneous radiation and photodynamic therapies for cancer treatment[J]. Journal of Nanoscience and Nanotechnology, 6, 1159-1166(2006).
[9] Haase M, Schäfer H. Upconverting nanoparticles[J]. Angewandte Chemie International Edition, 50, 5808-5829(2011).
[11] Laptev R, Nisnevitch M, Siboni G et al. Intracellular chemiluminescence activates targeted photodynamic destruction of leukaemic cells[J]. British Journal of Cancer, 95, 189-196(2006).
[12] Robertson R, Germanos M S, Li C et al. Optical imaging of Cerenkov light generation from positron-emitting radiotracers[J]. Physics in Medicine and Biology, 54, N355-N365(2009).
[13] Spinelli A E, Boschi F. Novel biomedical applications of Cerenkov radiation and radioluminescence imaging[J]. Physica Medica, 31, 120-129(2015).
[14] Kotagiri N, Niedzwiedzki D M, Ohara K et al. Activatable probes based on distance‐dependent luminescence associated with cerenkov radiation[J]. Angewandte Chemie, 52, 7756-7760(2013).
[17] Guo W S, Sun X L, Jacobson O et al. Intrinsically radioactive [ 64Cu]CuInS/ZnS quantum dots for PET and optical imaging: improved radiochemical stability and controllable cerenkov luminescence[J]. ACS Nano, 9, 488-495(2015).
[18] Li Y, Ma J L, Han S X. Advances in Cerenkov luminescence imaging for radiation oncology[J]. Chinese Journal of Medical Physics, 35, 60-69(2018).
[19] Fu Q F, Chen J Y, Liu Z B. Cherenkov light: a new light source for theranostic nanomedicine[J]. Journal of Nuclear and Radiochemistry, 41, 201-207(2019).
[20] Cline B, Delahunty I, Xie J. Nanoparticles to mediate X-ray-induced photodynamic therapy and Cherenkov radiation photodynamic therapy[J]. Wiley Interdisciplinary Reviews: Nanomedicine and Nanobiotechnology, 11, e1541(2019).
[21] Cao X, Chen X L, Kang F et al. Intensity enhanced cerenkov luminescence imaging using terbium-doped Gd2O2S microparticles[J]. ACS Applied Materials & Interfaces, 7, 11775-11782(2015).
[22] Kotagiri N, Sudlow G, Akers W J et al. Breaking the depth dependency of phototherapy with Cerenkov radiation and low-radiance-responsive nanophotosensitizers[J]. Nature Nanotechnology, 10, 370-379(2015).
[23] Shaffer T M, Pratt E C, Grimm J. Utilizing the power of Cerenkov light with nanotechnology[J]. Nature Nanotechnology, 12, 106-117(2017).
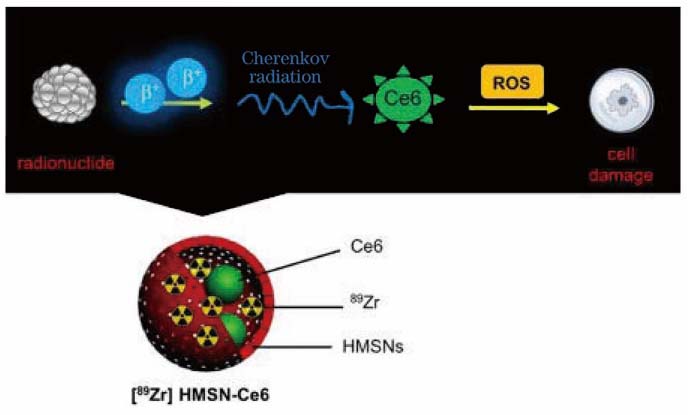
[24] Kamkaew A, Cheng L, Goel S et al. Cerenkov radiation induced photodynamic therapy using chlorin e6-loaded hollow mesoporous silica nanoparticles[J]. ACS Applied Materials & Interfaces, 8, 26630-26637(2016).
[25] Hartl B A, Hirschberg H, Marcu L et al. Activating photodynamic therapy in vitro with cerenkov radiation generated from yttrium-90[J]. Journal of Environmental Pathology, Toxicology and Oncology, 35, 185-192(2016).
[26] Kim K, Zhang H W, La Rosa S et al. Bombesin antagonist-based radiotherapy of prostate cancer combined with WST-11 vascular targeted photodynamic therapy[J]. Clinical Cancer Research, 23, 3343-3351(2017).
[27] Duan D B, Liu H, Xu Y et al. Activating TiO2 nanoparticles: gallium-68 serves as a high-yield photon emitter for Cerenkov-induced photodynamic therapy[J]. ACS Applied Materials & Interfaces, 10, 5278-5286(2018).
[28] Ni D L, Ferreira C A, Barnhart T E et al. Magnetic targeting of nanotheranostics enhances cerenkov radiation-induced photodynamic therapy[J]. Journal of the American Chemical Society, 140, 14971-14979(2018).
[29] Yoon S W, Tsvankin V, Shrock Z et al. Enhancing radiation therapy through Cherenkov light-activated phototherapy[J]. International Journal of Radiation Oncology, Biology, Physics, 100, 794-801(2018).
[30] Alexander D A, Tendler I I, Bruza P et al. Assessment of imaging Cherenkov and scintillation signals in head and neck radiotherapy[J]. Physics in Medicine & Biology, 64, 145021(2019).
[31] Jia M J, Bruza P, Andreozzi J M et al. Cherenkov-excited luminescence scanned imaging using scanned beam differencing and iterative deconvolution in dynamic plan radiation delivery in a human breast phantom geometry[J]. Medical Physics, 46, 3067-3077(2019).
[33] Henderson B W, Busch T M, Snyder J W. Fluence rate as a modulator of PDT mechanisms[J]. Lasers in Surgery and Medicine, 38, 489-493(2006).
[34] Glaser A K, Zhang R X, Andreozzi J M et al. Cherenkov radiation fluence estimates in tissue for molecular imaging and therapy applications[J]. Physics in Medicine and Biology, 60, 6701-6718(2015).
[35] Pratx G, Kapp D S. Is Cherenkov luminescence bright enough for photodynamic therapy[J]. Nature Nanotechnology, 13, 354(2018).
[36] Gill R K, Mitchell G S, Cherry S R. Computed Cerenkov luminescence yields for radionuclides used in biology and medicine[J]. Physics in Medicine and Biology, 60, 4263-4280(2015).
[37] Rajh T, Dimitrijevic N M, Rozhkova E A. Titanium dioxide nanoparticles in advanced imaging and nanotherapeutics[J]. Methods of Molecular Biology, 726, 63-75(2011).
[38] Kotagiri N, Laforest R, Achilefu S. Reply to ‘Is Cherenkov luminescence bright enough for photodynamic therapy?’[J]. Nature Nanotechnology, 13, 354-355(2018).
[39] Liu F, Xiao L, Ye Y et al. Integrated Cherenkov radiation emitter eliminating the electron velocity threshold[J]. Nature Photonics, 11, 289-292(2017).