Qinghua Wang, Huixin Wang, Zhandong Wang, Guifang Sun. Highly Efficient Nanosecond Laser-Based Multifunctional Surface Fabrication and Corrosion Resistance Performance[J]. Chinese Journal of Lasers, 2021, 48(14): 1402018
Search by keywords or author
- Chinese Journal of Lasers
- Vol. 48, Issue 14, 1402018 (2021)
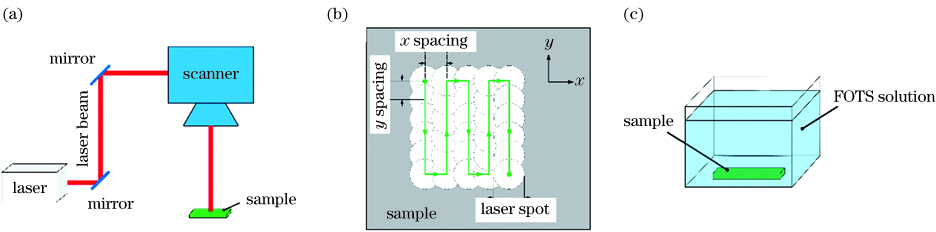
Fig. 1. Process schematic of the highly efficient laser-based micro/nanostructuring superhydrophobic surface. (a) Laser surface treatment; (b) scanning trajectory; (c) chemical immersion treatment
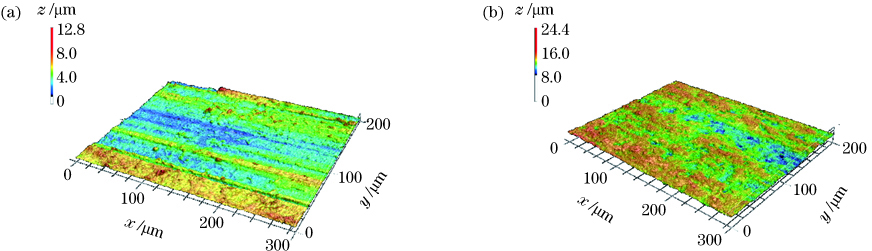
Fig. 2. 3D surface profiles. (a) Untreated surface; (b) laser micro/nanostructured surface
Fig. 3. SEM micrographs of surface structures fabricated by the existing ultra-fast laser-based surface texturing methods. (a) LIPSS structure fabricated by Cunha et al.[39]; (b) hierarchical structures fabricated by Martínez-Calderon et al.[40]; (c)(d) untreated surface; (e)(f) laser micro/nanostructured surface fabricated by the proposed method using the power intensity of 0.6 GW/cm2; (g)(h) laser micro/nanostructured surface fabricated by the proposed method using the power intensity of 8.4 GW/cm2
Fig. 4. XPS survey spectra analysis. (a) Untreated surface; (b) laser micro/nanostructured surface, the inset shows the core elemental spectrum of C element on laser micro/nanostructured surface
Fig. 5. Contact angle and roll-off angle measurement results for laser micro/nanostructured material with different laser power intensities. (a) AISI4130 low alloy steel; (b) AA6061 aluminium alloy
Fig. 6. Microhardness measurement results of laser micro/nanostructured AISI4130 low alloy steel and AA6061 aluminium alloy treated by different laser power intensities
Fig. 7. Corrosion resistance of the laser micro/nanostructured surface. (a) Comparison of the polarization curves for the untreated surface and laser micro/nanostructured surface; (b) surface structures and contact angles for the laser micro/nanostructured surface before and after the electrochemical test
Fig. 8. Deliquescence of NaCl on horizontal untreated surface and laser micro/nanostructured surface with relative humidity of 90%. (a)--(c) Untreated surface; (d)--(f) laser micro/nanostructured surface
Fig. 9. Deliquescence of NaCl on laser micro/nanostructured surface inclined at an angle of ~20° with relative humidity of 90%
Fig. 10. Comparison of the processing efficiency between proposed method and traditional laser processing methods
| ||||||||||||||||||||||||||||||||
Table 1. Main chemical compositions of AISI4130 low alloy steel and AA6061 aluminum alloy
|
Table 2. Ecorr, Icorr and corrosion rate of the untreated surface and laser micro/nanostructured surface

Set citation alerts for the article
Please enter your email address



