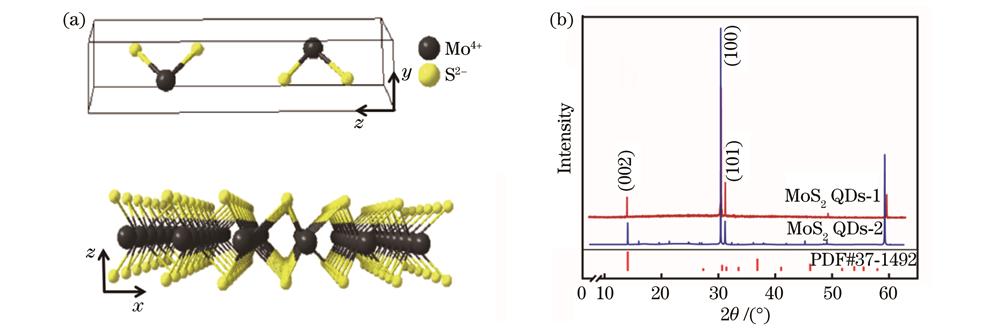
Molybdenum disulfide quantum dots (MoS2 QDs) have potential applications in the fields of sensing, fluorescence detection, and photocatalysis due to their excellent physicochemical properties such as controllable size and strong quantum confinement effect. The performance of MoS2 QDs is closely related to their size and number of layers. How to obtain MoS2 QDs with controllable size and number of layers is still a difficult problem. In this study, the MoS2 QDs with a small average grain size and few layers are synthesized by a facile and energy-intensive hydrothermal method. The effects of different sulfur sources (glutathione and L-cysteine) on the photoluminescence properties of MoS2 QDs are systematically studied. The MoS2 QDs prepared with glutathione as the sulfur source have a smaller average grain size, fewer layers, and better photoluminescence in comparison to L-cysteine-based MoS2 QDs. We hope that our basic strategy and findings can be helpful on the design of high-quality MoS2 QDs.
Firstly, 0.0468 g of (NH4)6Mo7O24·4H2O is dissolved in 2.5 mL of deionized water, and its pH value is adjusted to 6.5 with 10% mass fraction of ammonia water. Then, 0.254 g of glutathione and the above solution are added to 10 mL of ionized water (molar ratio of Mo∶S=1∶3) and stirred for 8 min until complete dissolution. Next, the mixed solution is transferred to a polytetrafluoroethylene stainless steel autoclave with a size of 50 mL and placed in an oven at 200 ℃ for 24 h. Then, the solution obtained from the reaction is placed in a sand core filter (0.22 μm) to filter out suspended particles, and the solution supernatant is collected after centrifugation at 4 ℃ and 10000 r/min for 15 min. Finally, the supernatant is dialyzed in a dialysis bag (the interception molecular weight of the dialysis bag is 10000 u) for 24 h, and the solution is collected and stored in a refrigerator at 4 ℃ and labeled as MoS2 QDs-1. Similarly, we weighes 0.0983 g ammonium molybdate as molybdenum source and 0.200 g L-cysteine as sulfur source (molar ratio of Mo∶S=1∶3) to prepare MoS2 QDs-2.
In this study, homogeneous dispersed MoS2 QDs are successfully obtained by a one-step hydrothermal method using glutathione and L-cysteine as sulfur sources respectively. Among them, the MoS2 QDs-1 sample has a smaller average size (3.88 nm), a lower average height (4.75 nm), a smaller optical band gap (3.65 eV), and a higher fluorescence quantum yield (10.8%) in comparison to MoS2 QDs-2 sample. Therefore, the structural and optical properties of the MoS2 QDs-1 sample are better under these experimental conditions. The carbon chain of glutathione (C10H17N3O6S) is longer than that of L-cysteine (C3H7NO2S), which is beneficial to the nucleation of nanocrystals. In addition to providing a sulfur source, glutathione can also act as a surfactant to inhibit the growth of crystal nuclei. Consequently, compared with L-cysteine, MoS2 QDs with a smaller average size and lower average height are more easily obtained from glutathione as a sulfur source, and the optical properties and photoluminescence properties of MoS2 QDs are affected by their sizes and number of layers. The average size of MoS2 QDs-1 is smaller than that of MoS2 QDs-2. Meanwhile, MoS2 QDs-1 has fewer layers. Therefore, MoS2 QDs-1 has a better optical band gap and higher fluorescence quantum yield.