Hua Lu, Shouhao Shi, Dikun Li, Shuwen Bo, Jianxu Zhao, Dong Mao, Jianlin Zhao.
Search by keywords or author
- Advanced Photonics
- Vol. 6, Issue 3, 036001 (2024)
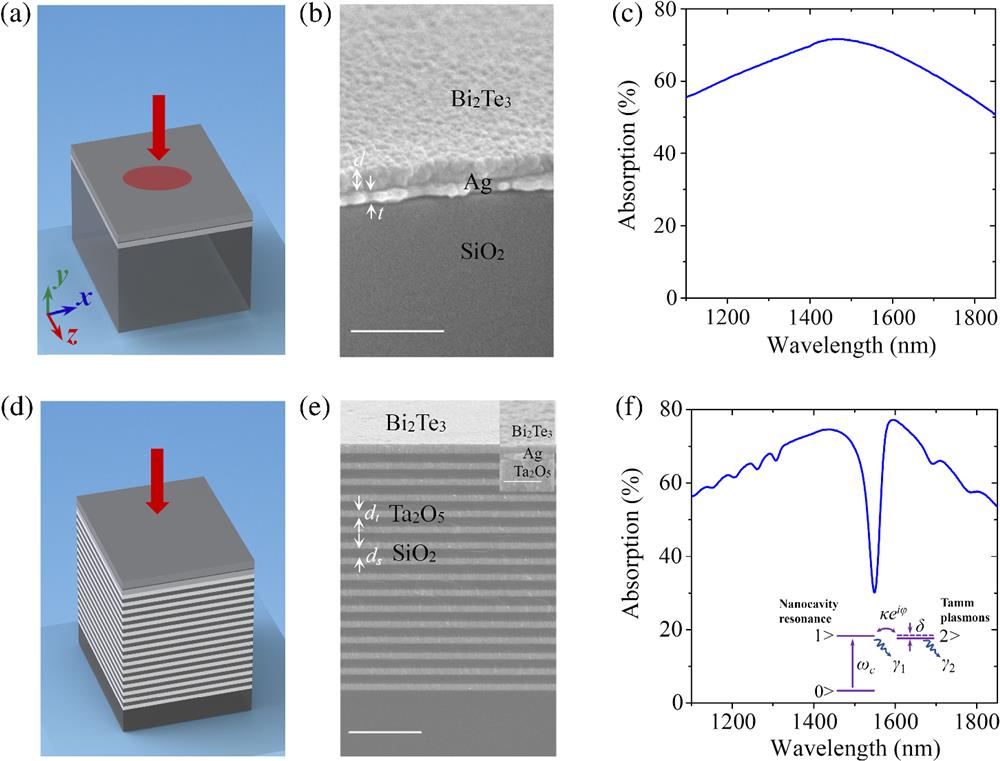
Fig. 1. TI structures and absorption spectra. (a) 3D diagram of the ultrathin nanocavity with a TI nanofilm coated on a metallic layer. (b) Angle-tilted cross-sectional SEM image of a
![Material characterization and optical constants of Bi2Te3 TI. (a) TEM image of a grown 42-nm Bi2Te3 TI film transferred on the supporter of a copper microgrid. (b) SAED pattern of the Bi2Te3 nanofilm. (c) HRTEM image of the Bi2Te3 nanofilm. (d) Ellipsometer-measured (circles) and fitted (curves) refractive indices and extinction coefficients of Bi2Te3 TI at the wavelengths from 230 to 1930 nm. (e), (f) Fitted refractive indices and extinction coefficients of Bi2Te3 TI surface and bulk states with the layer-on-bulk model [the inset in panel (d)].](/richHtml/ap/2024/6/3/036001/img_002.png)
Fig. 2. Material characterization and optical constants of
Fig. 3. TI thickness-dependent nanocavity resonance and induced transparency. (a) Experimentally measured absorption spectra of the TI nanocavities on the
Fig. 4. EIT-like response with different silver film thicknesses. (a) Theoretical evolution of absorption spectrum from the TI nanocavity/photonic crystal structures with different silver film thicknesses

Set citation alerts for the article
Please enter your email address



