| hydrogen peroxide
(H2O2)
| (1) titanium sulfate reagent colorimetry
(2) colorimetric or fluorometric analysis
| (1) less interference, good stability, but limited sensitivity(2) many interference factors (such as air, pH, solvent, etc.), but high sensitivity, lower detection limit~10×10−6 | OH + H2O·→ H2O2+ H2O2+2H++2e−→H2O2H2O2+H++e−→H2O+OH
| [17][18][19]
|
| hydrogen
atom (H)
| (1) EPR technology
(2) isotope labeling
| (1) high sensitivity, low detection limit, easy decomposition of the product, requiring excess capture agent(2) the source, distribution and chemical reaction of H atoms can be explored, but it is usually used in conjunction with the EPR method | H++e−→H
| [20][21][22]
|
| hydroxyl radicals
(OH·)
| (1) EPR technology(2) HPLC method
(3) spectrophotometry
| (1) high sensitivity and low detection limit, the most ideal method for the determination of hydroxyl radicals, the instrument is expensive, the choice of capture agent is critical(2) easy to implement, but its sensitivity and accuracy are still insufficient(3) easy to operate, has high selectivity, and is not easily affected by other ions at specific wavelengths; insufficient accuracy and sensitivity, requires the capture agent or the adduct product with the hydroxyl to have characteristic fluorescence | H2O→H+OH·
OH·+H++e−→H2O
| [23][24][25][26]
|
| superoxide anion
(O2−)
| (1) EPR technology
(2) fluorescent probe method
| (1) high sensitivity, low detection limit, expensive instrument(2) easy to operate, lower instrument price, but the sensitivity is low, and it is interfered by particles like H2O2, OH·
| 2O+H2O+e−→H2O+O2− | [27][28][29]
|
| ozone
(O3)
| (1) spectrophotometry
(2) fluorescent probe method
| (1) the operation is simple and fast, but the sensitivity is low, and it will be interfered by other oxidants(2) good selectivity, but it needs to be in a specific pH range; when pH ≥ 10, the probe will be destroyed in H2O2 | O3+2H++2e−→O2+H2O
| [30][31][32]
|
| nitric oxide
(NO)
| (1) EPR technology(2) spectrophotometry
(3) fluorescent probe method
| (1) high sensitivity, low detection limit, wide application, but need to avoid oxidation of Fe2+ particles in the reagent(2) easy to operate, instrument is relatively cheap, and the aqueous solution to be tested needs to be acidified in advance before the test(3) has good selectivity, but the pH of the aqueous solution to be tested needs to be higher than 5.5
| 4NO+2O2+2H2O→
4NO2−+4H+ | [33][34][35][36]
|
| nitrite (NO2−)
| (1) spectrophotometry
(2) HPLC method
| (1) the operation is simple and fast, but the sensitivity is low, and it will be interfered by other oxidants(2) easy to implement, but its sensitivity and accuracy are still insufficient | 2HNO2→NO+NO2+H2ONO2−+H+→HNO2 | [37]
[38]
|
| nitrate
(NO3−)
| (1) spectrophotometry
(2) HPLC method
| (1) the operation is simple and fast, but the sensitivity is low, and it will be interfered by other oxidants(2) easy to implement, but its sensitivity and accuracy are still insufficient | NO2−+ OH·→NO3− | [39][40][41]
|
| peroxynitrite
(ONOO−)
| (1) colorimetric or fluorometric analysis(2) chemical reaction(3) fluorescent probe method | (1) multiple interference factors, extremely sensitive to light-induced oxidation, and specificity problems(2) the existence of ONOO− can only be indirectly proved by the concentration decay of NO2− and H2O2, and the accuracy is insufficient (3) low selectivity, interfered by particles like H2O2 and HClO, in the absence ofHClO, the selectivity is enhanced
| NO2−+H2O2→ONOO−+H2OONOO−→NO3− | [42][43][44]
|
| peroxynitric
acid (O2NOOH)
| (1) HPLC method | (1) easy to detect, but the sensitivity is low, the detection environment requires low temperature and low pH value | NO3−+H2O2→
O2NOO−+H2O
| [45]
|
| singlet oxygen
(1O2)
| (1) EPR technology
(2) fluorescent probe method
| (1) good selectivity, high sensitivity, not affected by other free radicals, the detection reagent has a strong pungent odor and is easily oxidized in the air(2) the probe acts as a photosensitizer to generate singlet oxygen, and the probe is destroyed in hydrogen peroxide | O2+e−→1O2+e−21O2+2H+→H2O2+O21O2+H2O2→OH·+OH−+O2 | [46][47][48][49]
|
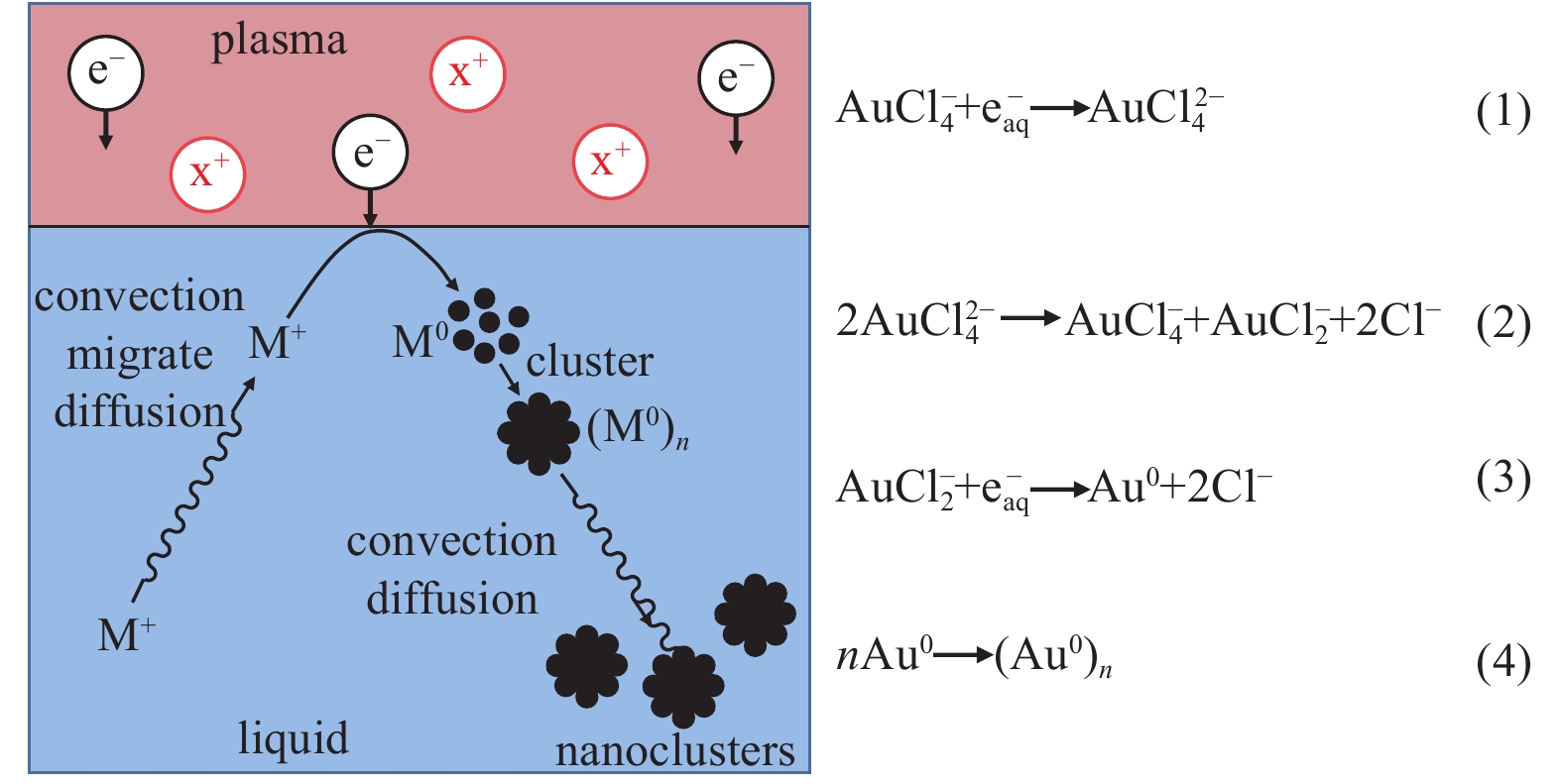
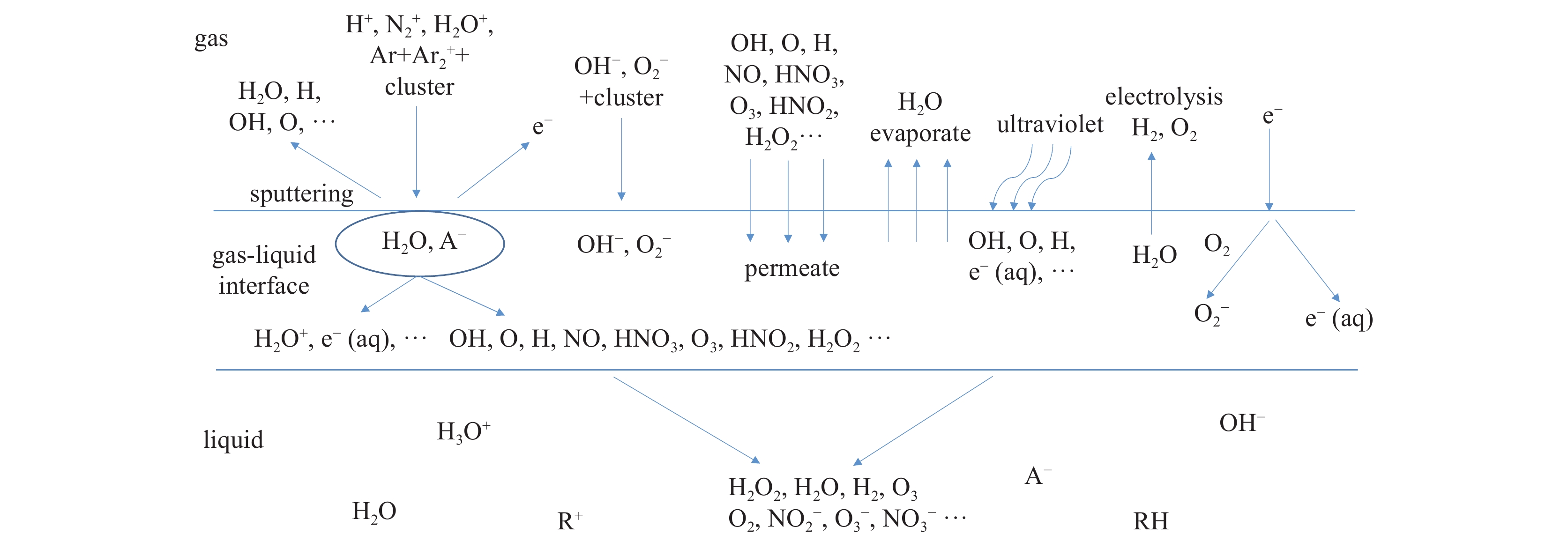
![Schematic diagram of mass transfer, reaction and active components that may exist in non-contact liquid plasma[10]](/richHtml/qjglzs/2022/34/6/069001/img_1.jpg)