Author Affiliations
1College of Physics and Electronic Engineering, Xinjiang Normal University, Urumqi , Xinjiang 830054, China2Xinjiang Institute of Metrology and Testing, Urumqi , Xinjiang 830011, Chinashow less
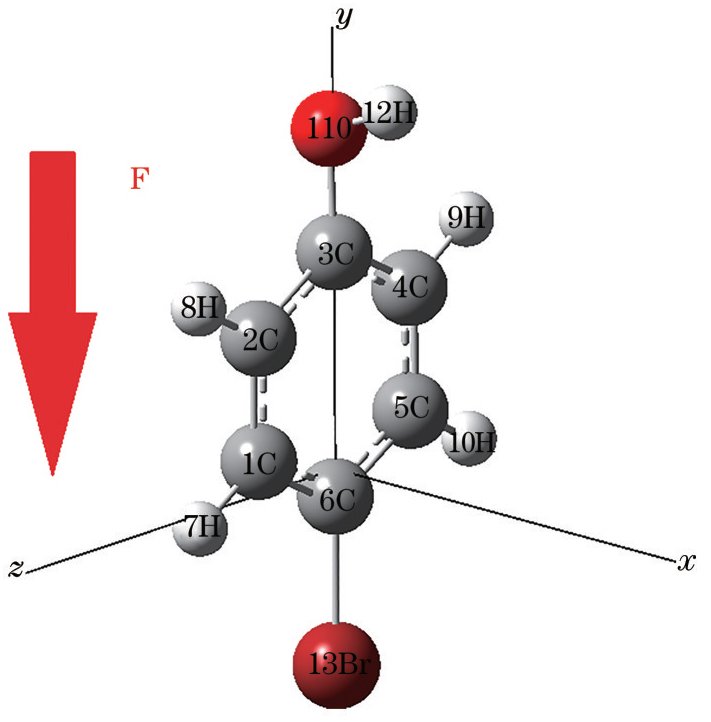
Fig. 1. Stable ground state configuration of 4-bromophenol molecule without external electric field
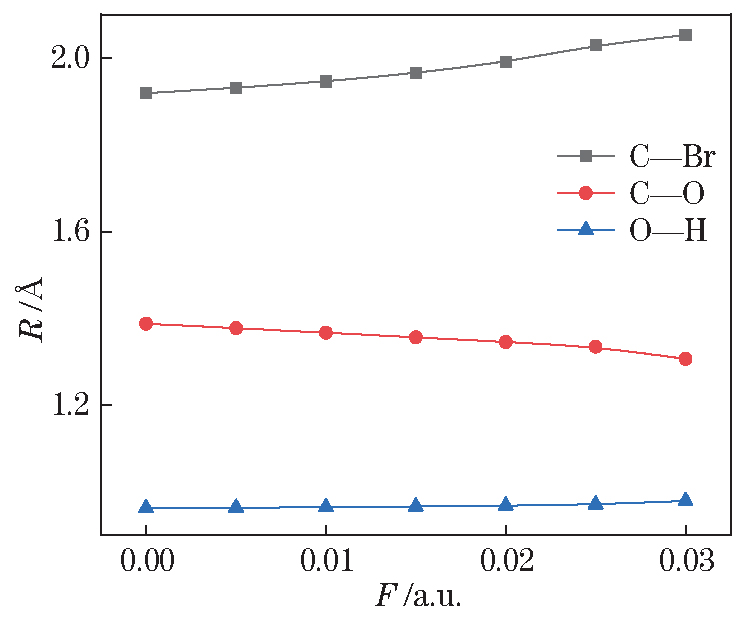
Fig. 2. Change of 4-bromophenol molecular bond length under different external electric fields
Fig. 3. Change of 4-bromophenol molecular total energy under different external electric fields
Fig. 4. Change of 4-bromophenol molecular electric dipole moment under different external electric fields
Fig. 5. Change of 4-bromophenol molecular the lowest empty orbital energy and the highest occupied orbital energy under different external electric fields
Fig. 6. Change of 4-bromophenol molecular energy gap under different external electric fields
Fig. 7. Changes of 4-bromophenol molecular vibration frequency and infrared spectrum under different external electric fields
Fig. 8. Change of potential energy surface of C-Br bond dissociation of 4-bromophenol molecules under different external electric fields
| Parameter | B3LYP/6-31+G | Semi-empirical/PM6 | B3LYP/6-311+G(d,p) | HF/6-31+G(d,p) | HF/6-311+G(d,p) | Experimental value[28] |
|---|
| R(6,13) /Å | 1.94796 | 1.90910 | 1.91881 | 1.89006 | 1.90146 | 1.91378 | | R(1,2) /Å | 1.39885 | 1.39628 | 1.39359 | 1.38598 | 1.38494 | 1.39546 | | R(3,11) /Å | 1.40993 | 1.38825 | 1.38745 | 1.36752 | 1.36640 | 1.36660 | | R(5,10) /Å | 1.08326 | 1.09193 | 1.08246 | 1.07364 | 1.07336 | 1.08473 | | R(11,12) /Å | 0.97827 | 0.98203 | 0.96282 | 0.94420 | 0.94186 | 0.98972 | | A(1,6,13) /(º) | 119.15890 | 119.07557 | 119.43041 | 119.67087 | 119.58250 | 119.61728 | | A(2,1,7) /(º) | 120.64629 | 119.93268 | 120.33960 | 120.12178 | 120.18052 | 120.20896 | | A(4,3,11) /(º) | 119.68248 | 118.74810 | 119.89736 | 119.87399 | 119.94049 | 117.30265 |
|
Table 1. Ground state structures of 4-bromophenol molecules optimized by different methods
| Parameter | F /a.u. |
|---|
| 0 | 0.005 | 0.010 | 0.015 | 0.020 | 0.025 | 0.030 |
|---|
| R(6,13) /Å | 1.91881 | 1.93098 | 1.94575 | 1.96486 | 1.99027 | 2.02914 | 2.05280 | | R(3,11) /Å | 1.38746 | 1.37756 | 1.36683 | 1.35639 | 1.34549 | 1.33455 | 1.30656 | | R(11,12) /Å | 0.96282 | 0.96385 | 0.96480 | 0.96639 | 0.96847 | 0.97136 | 0.97962 | | E /Hartree | -2881.09492 | -2881.09930 | -2881.10746 | -2881.11953 | -2881.13576 | -2881.15652 | -2881.18601 | | μ /Debye | 1.9717 | 3.5083 | 5.3254 | 7.2921 | 9.4298 | 11.8408 | 21.8223 |
|
Table 2. Bond length, total energy, and electric dipole moment of 4-bromophenol molecules under different external electric fields
| Atom | F /a.u. |
|---|
| 0 | 0.005 | 0.010 | 0.015 | 0.020 | 0.025 | 0.030 |
|---|
| 1C | -0.538 | -0.542 | -0.551 | -0.563 | -0.581 | -0.604 | -0.593 | | 2C | 0.278 | 0.280 | 0.282 | 0.281 | 0.279 | 0.274 | 0.304 | | 3C | -0.483 | -0.482 | -0.481 | -0.476 | -0.466 | -0.450 | -0.507 | | 6C | 0.634 | 0.671 | 0.719 | 0.778 | 0.851 | 0.941 | 0.941 | | 7H | 0.149 | 0.140 | 0.130 | 0.121 | 0.113 | 0.106 | 0.116 | | 8H | 0.137 | 0.149 | 0.162 | 0.174 | 0.187 | 0.200 | 0.224 | | 11O | -0.336 | -0.316 | -0.296 | -0.278 | -0.260 | -0.243 | -0.193 | | 12H | 0.275 | 0.290 | 0.307 | 0.325 | 0.347 | 0.369 | 0.422 | | 13Br | -0.143 | -0.216 | -0.294 | -0.377 | -0.467 | -0.570 | -0.765 |
|
Table 3. Charge distribution of 4-bromophenol molecules under different external electric fields
| Parameter | F /a.u. |
|---|
| 0 | 0.005 | 0.010 | 0.015 | 0.020 | 0.025 | 0.030 |
|---|
| EL /Hartree | -0.03655 | -0.03413 | -0.05751 | -0.08849 | -0.12361 | -0.16097 | -0.20128 | | EH /Hartree | -0.24983 | -0.24543 | -0.23883 | -0.23047 | -0.22078 | -0.2106 | -0.22105 | | EG /eV | 5.80122 | 5.74736 | 4.93190 | 3.86186 | 2.64302 | 1.349936 | 0.53774 |
|
Table 4. Lowest empty orbital energy, highest occupied orbital energy and energy gap of 4-bromophenol molecules under different external electric fields
| F /a.u. | 0 | 0.005 | 0.010 | 0.015 | 0.020 | 0.025 | 0.030 |
|---|
| 745.48 cm-1 | 826.81 | 823.10 | 815.79 | 803.84 | 785.85 | 757.16 | 745.48 | | 847.12 cm-1 | 947.93 | 945.51 | 941.47 | 935.36 | 926.94 | 914.40 | 847.12 | | 1552.53 cm-1 | 1626.30 | 1623.98 | 1618.88 | 1611.04 | 1600.96 | 1590.96 | 1552.53 | | 3606.77 cm-1 | 3825.33 | 3816.79 | 3808.78 | 3790.85 | 3765.33 | 3728.17 | 3606.77 |
|
Table 5. Frequency changes of 4-bromophenol molecules under different external electric fields
| State | F /a.u. |
|---|
| 0 | 0.005 | 0.010 | 0.015 | 0.020 | 0.025 | 0.030 |
|---|
| λ /nm | ƒ | λ /nm | ƒ | λ /nm | ƒ | λ /nm | ƒ | λ /nm | ƒ | λ /nm | ƒ | λ /nm | ƒ |
|---|
| n=1 | 248.57 | 0.0111 | 259.97 | 0.0004 | 299.69 | 0.0012 | 384.84 | 0.0014 | 593.34 | 0.0011 | 1470.97 | 0.0007 | 4145.03 | 0.0280 | | n=2 | 239.79 | 0 | 244.90 | 0.0098 | 254.60 | 0 | 321.97 | 0 | 437.80 | 0 | 880.99 | 0 | 1499.15 | 0 | | n=3 | 223.54 | 0.1415 | 223.21 | 0 | 244.58 | 0.0059 | 289.24 | 0.0001 | 422.16 | 0 | 700.06 | 0 | 1039.51 | 0.0004 | | n=4 | 211.78 | 0.0120 | 220.25 | 0.1367 | 243.40 | 0.0088 | 278.93 | 0.0030 | 324.65 | 0.002 | 449.03 | 0.0002 | 559.56 | 0.0913 | | n=5 | 204.45 | 0.0003 | 218.50 | 0.0150 | 223.00 | 0.1447 | 255.12 | 0 | 323.47 | 0 | 394.07 | 0.0032 | 432.88 | 0.0563 | | n=6 | 200.83 | 0.0081 | 199.18 | 0.0004 | 222.64 | 0.0001 | 243.77 | 0.0069 | 288.44 | 0.0393 | 374.21 | 0 | 420.25 | 0.0537 | | n=7 | 198.93 | 0.0003 | 196.95 | 0.0043 | 214.96 | 0 | 242.36 | 0.0866 | 272.30 | 0.0001 | 366.23 | 0.0219 | 401.93 | 0.0343 | | n=8 | 196.23 | 0.0252 | 196.19 | 0.0034 | 214.34 | 0.0001 | 240.55 | 0.0005 | 271.77 | 0 | 347.09 | 0.0071 | 380.67 | 0.0003 | | n=9 | 196.10 | 0.0479 | 193.34 | 0.0025 | 208.28 | 0.0051 | 228.05 | 0.0161 | 270.94 | 0.0083 | 336.09 | 0.0007 | 367.04 | 0.0133 | | n=10 | 194.38 | 0.0570 | 192.68 | 0.0017 | 207.22 | 0.0013 | 227.18 | 0.0091 | 265.42 | 0.0011 | 323.03 | 0.0001 | 351.79 | 0.0004 |
|
Table 6. Wavelength and oscillator intensity of singlet excited state of 4-bromophenol molecule under different external electric fields