Huan SONG, Lin WANG, Hong-Qing WANG, Wei-Qun SHI. Adsorption of Eu(III) on Alkalized Ti3C2Tx MXene Studied by Batch Experiment and Its Mechanism Investigation [J]. Journal of Inorganic Materials, 2020, 35(1): 65
Search by keywords or author
- Journal of Inorganic Materials
- Vol. 35, Issue 1, 65 (2020)
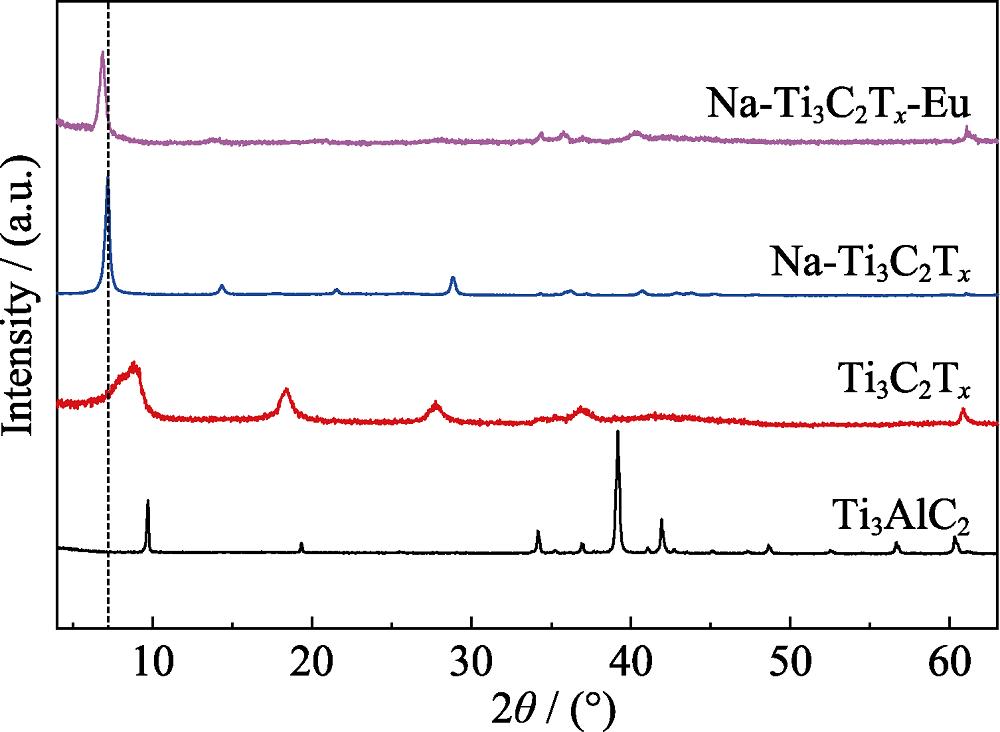
1. XRD patterns of the synthesized Ti3AlC2, Ti3C2Tx , Na-Ti3C2Tx and Na-Ti3C2Tx -Eu
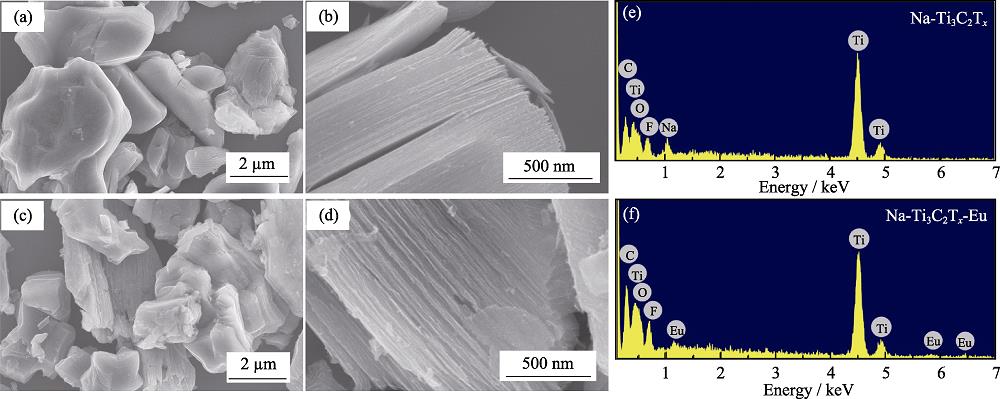
2. SEM images of Na-Ti3C2Tx before (a-b), and after (c-d) adsorption of Eu(III), with EDS analysis resulted of Na-Ti3C2Tx before (e) and after (f) adsorption of Eu(III)
3. Effect of sorbent dose on Eu(III) removal by Na-Ti3C2Tx (pH=(4.0±0.1), C 0=20 mg/L)
4. Effect of solution pH and ionic strength on removal of Eu(III) by Na-Ti3C2Tx (a), Zeta potential of Na-Ti3C2Tx (b), and distribution of Eu(III) species in aqueous solution as a function of pH calculated using the MEDUSA program (c)
5. Time-dependent adsorption of Eu(III) on Na-Ti3C2Tx (pH= (4.0±0.1), T = 293 K, m /V = 0.4 g/L) (a), and pseudo-second- order kinetic plots of Eu(III) on Na-Ti3C2Tx (b)
6. (a) Isotherms of Na-Ti3C2Tx towards Eu(III) under the conditions of pH (4.0±0.1) and m /V =0.40 g/L, and (b) plot of lnK dvs 1/T
7. (A) Eu L 3 edge k 3-weighted EXAFS spectra (solid lines) and the best theoretical fits (dots lines) of Eu-loaded Na-Ti3C2Tx samples under different solution pH, and (B) corresponding non-phase shift corrected Fourier transforms
| ||||||||||||||
Table 1. Optimized parameters of pseudo-first-order and pseudo-second-order kinetic models
| ||||||||||||||||||||||||||||||||||||||||||
Table 2. Fitting parameters of Langmuir and Freundlich models
|
Table 3. Thermodynamic parameters for removal of Eu(III) by Na-Ti3C2Tx
|
Table 4. Fitting parameters extracted from least-squares fitting analysis of EXAFS spectra

Set citation alerts for the article
Please enter your email address



