[1] G LI, R ZHU, Y YANG. Polymer solar cells. Nature Photonics, 6, 153-161(2012).
[2] Y SUN, G C WELCH, W L LEONG et al. Solution-processed small-molecule solar cells with 6.7% efficiency. Nature Materials, 11, 44-48(2012).
[3] J ZHAO, Y LI, G YANG et al. Efficient organic solar cells processed from hydrocarbon solvents. Nature Energy, 1, 15027(2016).
[4] C YAN, S BARLOW, Z WANG et al. Non-fullerene acceptors for organic solar cells. Nature Reviews Materials, 3, 18003(2018).
[5] Y CUI, H YAO, J ZHANG et al. Single-junction organic photovoltaic cells with approaching 18% efficiency. Advanced Materials, 32, 1908205(2020).
[6] Q LIU, Y JIANG, K JIN et al. 18% Efficiency organic solar cells. Science Bulletin, 65, 272-275(2020).
[7] J QIN, L ZHANG, C ZUO et al. A chlorinated copolymer donor demonstrates a 18.13% power conversion efficiency. Journal of Semiconductors, 42(2021).
[8] K JIN, Z XIAO, L DING. 69% PCE from organic solar cells. Journal of Semiconductors, 42(18).
[9] M B SALIM, R NEKOVEI, R JEYAKUMAR. Organic tandem solar cells with 18.6% efficiency. Solar Energy, 198, 160-166(2020).
[10] A MISHRA, P BAEUERLE. Small molecule organic semiconductors on the move: promises for future solar energy technology. Angewandte Chemie-International Edition, 51, 2020-2067(2012).
[11] P DUTTA, W YANG et al. Development of naphtho[1,2-b:5,6-b']dithiophene based novel small molecules for efficient bulk-heterojunction organic solar cells. Chemical Communications, 48, 573-575(2012).
[12] Y A DUAN, Y GENG, H B LI et al. Theoretical characterization and design of small molecule donor material containing naphthodithiophene central unit for efficient organic solar cells. Journal of Computational Chemistry, 34, 1611-1619(2013).
[13] D FERNANDEZ, A VITERISI, V CHALLURI et al. Understanding the limiting factors of solvent-annealed small-molecule bulk-heterojunction organic solar cells from a chemical perspective. ChemSusChem, 10, 3118-3134(2017).
[14] H LI, Y ZHAO, J FANG et al. Improve the performance of the all-small-molecule nonfullerene organic solar cells through enhancing the crystallinity of acceptors. Advanced Energy Materials, 8, 1702377(2018).
[15] Y SHI, C YANG, H LI et al. A-π-D-π-A small-molecule donors with different end alkyl chains obtain different morphologies in organic solar cells. Chinese Chemical Letters, 30, 906-910(2019).
[16] S J LOU, S LOSER, K A LUCK et al. Charge generation mechanism tuned via film morphology in small molecule bulk-heterojunction photovoltaic materials. Journal of Materials Chemistry C, 8, 15234-15252(2020).
[17] Z WEI, K LU, R ZHOU et al. Research progress of small molecule donors with high crystallinity in all small molecule organic solar cells. Acta Chimica Sinica, 79, 284-302(2021).
[18] Y CUI, H YAO, L HONG et al. Achieving over 15% efficiency in organic photovoltaic cells via copolymer design. Advanced Materials, 31, 1808356(2019).
[19] S Q ZHANG, Y P QIN, J ZHU et al. Over 14% efficiency in polymer solar cells enabled by a chlorinated polymer donor. Advanced Materials, 30, 1800868(2018).
[20] Y CUI, H YAO, J ZHANG et al. Over 16% efficiency organic photovoltaic cells enabled by a chlorinated acceptor with increased open-circuit voltages. Nature Communications, 10, 2515(2019).
[21] H X JIANG, X M LI, H WANG et al. Appropriate molecular interaction enabling perfect balance between induced crystallinity and phase separation for efficient photovoltaic blends. ACS Applied Materials & Interfaces, 12, 26286-26292(2020).
[22] H LI, Q WU, R ZHOU et al. Liquid-crystalline small molecules for nonfullerene solar cells with high fill factors and power conversion efficiencies. Advanced Energy Materials, 9, 1803175(2019).
[23] W NI, M M LI, B KAN et al. Fullerene-free small molecule organic solar cells with a high open circuit voltage of 1.15 V. Chemical Communications, 52, 465-468(2016).
[24] W WANG, B CHEN, X JIAO et al. A new small molecule donor for efficient and stable all small molecule organic solar cells. Organic Electronics, 70, 78-85(2019).
[25] H ZHANG, C WANG, X LI et al. The effect of processing conditions on performance of small-molecule organic solar cells. Solar Energy, 157, 71-80(2017).
[26] Y L WANG, Y WANG, L ZHU et al. A novel wide-bandgap small molecule donor for high efficiency all-small-molecule organic solar cells with small non-radiative energy losses. Energy & Environmental Science, 13, 1309-1317(2020).
[27] J GE, L HONG, W SONG et al. Solvent annealing enables 15.39% efficiency all-small-molecule solar cells through improved molecule interconnection and reduced non-radiative loss. Advanced Energy Materials, 2100800(2021).
[28] S LOSER, C J BRUNS, H MIYAUCHI et al. A naphthodithiophene-diketopyrrolopyrrole donor molecule for efficient solution-processed solar cells. Journal of the American Chemical Society, 133, 8142-8145(2011).
[29] S SHINAMURA, I OSAKA, E MIYAZAKI et al. Linear- and angular-shaped naphthodithiophenes: selective synthesis, properties, and application to organic field-effect transistors. Journal of the American Chemical Society, 133, 5024-5035(2011).
[30] K TAKIMIYA, I OSAKA. Naphthodithiophenes: emerging building blocks for organic electronics. Chemical Record, 15, 175-188(2015).
[31] X W ZHU, K LU, H LI et al. Naphthodithiophene-based donor materials for solution processed organic solar cells. Chinese Chemical Letters, 27, 1271-1276(2016).
[32] Y CHANG, Y CHANG, X ZHU et al. Constructing high-performance all-small-molecule ternary solar cells with the same third component but different mechanisms for fullerene and non-fullerene systems. Advanced Energy Materials, 9, 1900190(2019).
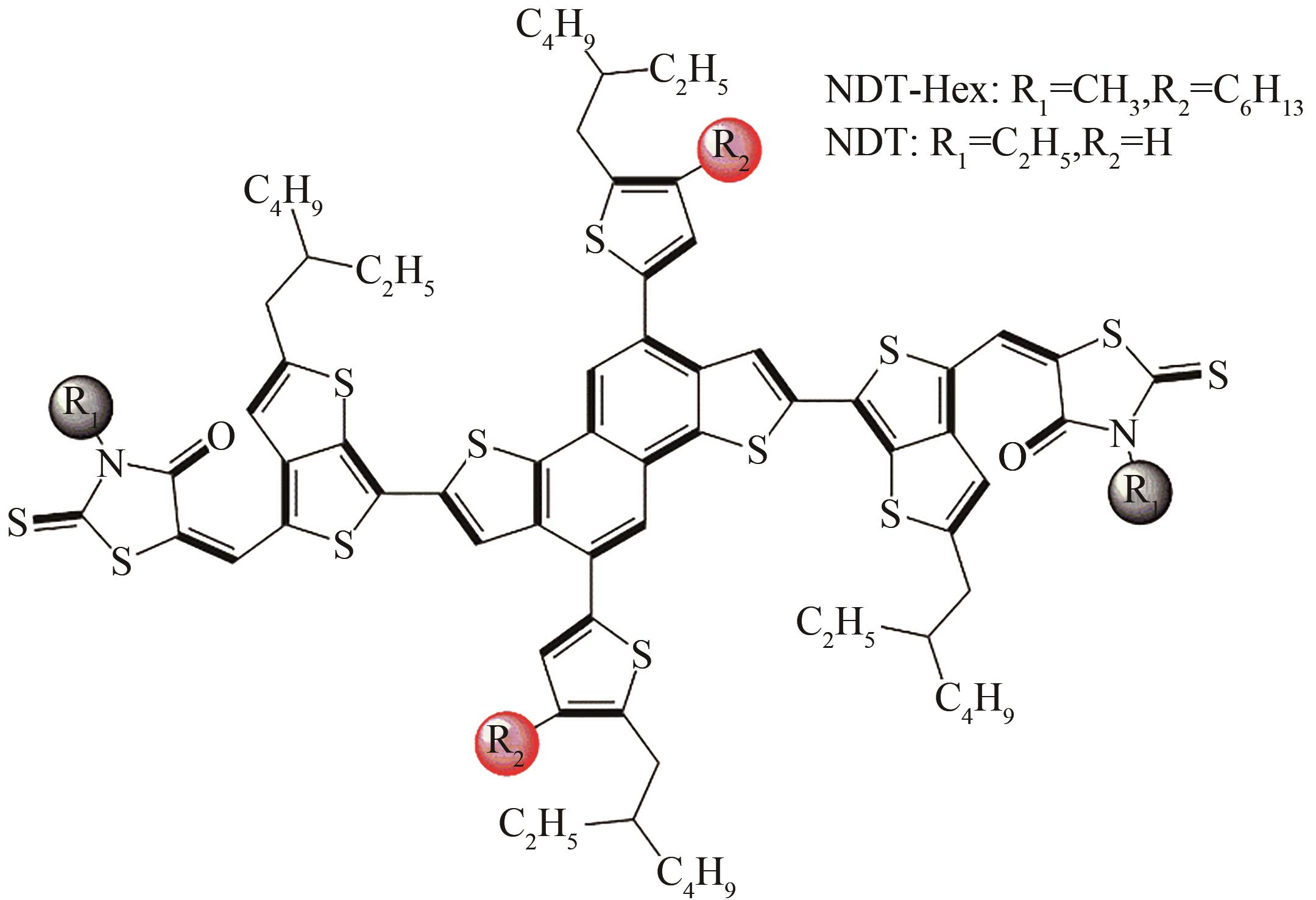
[33] C XIA, H WU, H FAN et al. Efficient NDT small molecule solar cells with high fill factor using pendant group engineering. Journal of Materials Chemistry C, 8, 7561-7566(2020).
[34] STOKKUM I H MVAN, D S LARSEN, GRONDELLE RVAN. Global and target analysis of time-resolved spectra. Biochimica Et Biophysica Acta-Bioenergetics, 1657, 82-104(2004).
[35] C HIPPIUS, STOKKUM I H MVAN, E ZANGRANDO et al. Excited state interactions in calix 4 arene-perylene bisimide dye conjugates: Global and target analysis of supramolecular building blocks. Journal of Physical Chemistry C, 111, 13988-13996(2007).
[36] X WANG, B KAN, Z KUANG et al. Unveiling the molecular symmetry dependence of exciton dissociation processes in small-molecular heterojunctions. Journal of Physical Chemistry C, 122, 26851-26856(2018).
[37] C RUCKEBUSCH, M SLIWA, P PERNOT et al. Comprehensive data analysis of femtosecond transient absorption spectra: A review. Journal of Photochemistry and Photobiology C-Photochemistry Reviews, 13, 1-27(2012).
[38] R BERERA, GRONDELLE RVAN, J T M KENNIS. Ultrafast transient absorption spectroscopy: principles and application to photosynthetic systems. Photosynthesis Research, 101, 105-118(2009).
[39] S GELINAS, A RAO, A KUMAR et al. Ultrafast long-range charge separation in organic semiconductor photovoltaic diodes. Science, 343, 512-516(2014).
[40] A C JAKOWETZ, M L BOEHM, A SADHANALA et al. Visualizing excitations at buried heterojunctions in organic semiconductor blends. Nature Materials, 16, 551-557(2017).
[41] M LI, F LIU, X WAN et al. Subtle balance between length scale of phase separation and domain purification in small-molecule bulk-heterojunction blends under solvent vapor treatment. Advanced Materials, 27, 6296-6302(2015).
[42] K SUN, Z XIAO, E HANSSEN et al. The role of solvent vapor annealing in highly efficient air-processed small molecule solar cells. Journal of Materials Chemistry A, 2, 9048-9054(2014).
[43] L XIE, C YANG, R ZHOU et al. Ternary organic solar cells based on two non-fullerene acceptors with complimentary absorption and balanced crystallinity. Chinese Journal of Chemistry, 38, 935-940(2020).
[44] B YANG, Y YUAN, J HUANG. Reduced bimolecular charge recombination loss in thermally annealed bilayer heterojunction photovoltaic devices with large external quantum efficiency and fill Factor. Journal of Physical Chemistry C, 118, 5196-5202(2014).