Chang-qing YE, Xue YU, Shuo-ran CHEN, Zuo-qin LIANG, Yu-yang ZHOU, Xiao-mei WANG. Study on the Structure/Energy-Level of Palladuim-Porphyrin Sensitizers on the Triplet-Triplet-Annihilation Upconversion Performance[J]. Spectroscopy and Spectral Analysis, 2021, 41(1): 71
Search by keywords or author
- Spectroscopy and Spectral Analysis
- Vol. 41, Issue 1, 71 (2021)
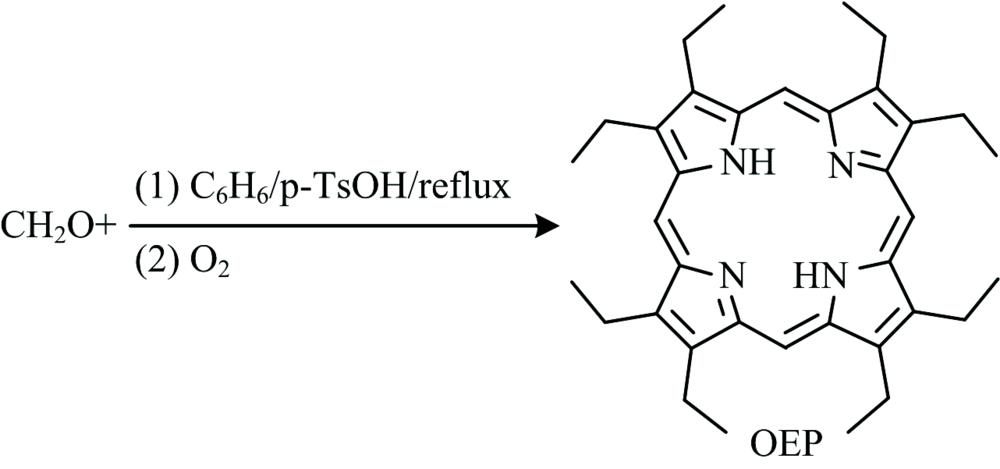
Fig. 1. The synthetic procedure of OEP
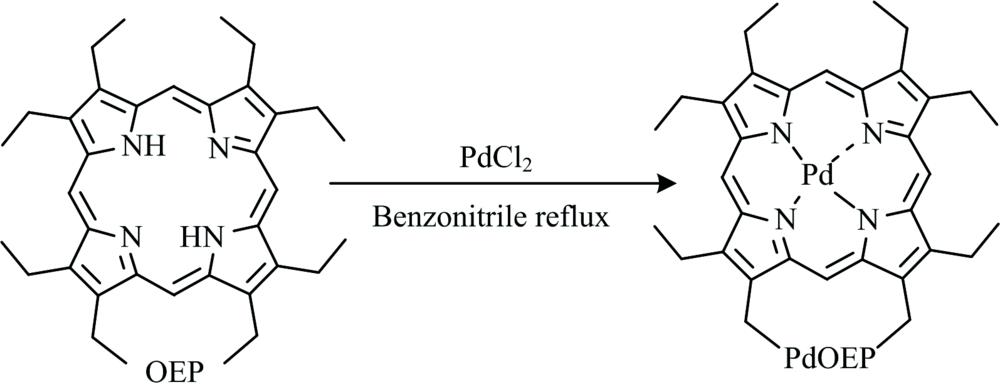
Fig. 2. The synthetic procedure of PdOEP
Fig. 3. The synthetic procedure of BrTPP
Fig. 4. The synthetic procedure of PdBrTPP
Fig. 5. The synthetic procedure of p -DHMPA
Fig. 6. Optical characterization of PdOEP in n -propanol
(a): Absorption spectrum; (b): Fluorescence spectrum; (c): The phosphorescence spectrum in N2 atmosphere; (excitation wavelength: 532 nm); (d): The phosphorescence decay curves associated with dual exponential fitting for PdOEP at room temperature in N2 atmosphere (Emission wavelength 665 nm)
(a): Absorption spectrum; (b): Fluorescence spectrum; (c): The phosphorescence spectrum in N2 atmosphere; (excitation wavelength: 532 nm); (d): The phosphorescence decay curves associated with dual exponential fitting for PdOEP at room temperature in N2 atmosphere (Emission wavelength 665 nm)
Fig. 7. Optical characterization of PdBrTPP in n -propanol
(a): Absorption spectrum; (b): Fluorescence spectrum; (c): The phosphorescence spectrum in N2 atmosphere; (excitation wavelength: 532 nm); (d): The phosphorescence decay curves associated with dual exponential fitting for PdBrTPP at room temperature in N2 atmosphere (Emission wavelength 694 nm)
(a): Absorption spectrum; (b): Fluorescence spectrum; (c): The phosphorescence spectrum in N2 atmosphere; (excitation wavelength: 532 nm); (d): The phosphorescence decay curves associated with dual exponential fitting for PdBrTPP at room temperature in N2 atmosphere (Emission wavelength 694 nm)
Fig. 8. Absorption and fluorescence spectra of p -DHMPA in n -propanol solution
Fig. 9. Jablonski diagram of the TTA-UC process between the sensitizers (PdOEP, PdBrTPP) and the emitter (p -DHMPA)
Fig. 10. Concentration-dependent upconversion spectra of emitter (p -DHMPA) doped with sensitizer (a)PdOEP, (b)PdBrTPP, (c)Stern-Volmer plots of sensitizers (PdOEP and PdBrTPP) quenched by emitter p -DHMPA
Fig. 11. The power-dependent upconversion spectra
(a): PdOEP/p -DHMPA; (b): PdBrTPP/p -DHMPA; Inset: Logarithmic plots of the integral of upconversion peak versus power density
(a): PdOEP/
Fig. 12. Under excitation of 532 nm laser (400 mW·cm-2), the concentration-dependent spectra and UC efficiency curves of emitter doped with different sensitizers. (a, b) PdOEP (10 μmol·L-1); (c, d) PdBrTPP(4 μmol·L-1), the fluorescence of Rh6G was set as the reference for the calculation of UC efficiency. The original data was shrank six times to fit the chart
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 1. Optical properitiesaod siglet/triplet energy levels of PdOEP, PdBrTPP and p-DHMPA in n-propanol
|
Table 2. Upconversion performance of PdOEP/p-DHMPAand PdBrTPP/p-DHMPA in n-propanol

Set citation alerts for the article
Please enter your email address



