Author Affiliations
1Faculty of Materials Science and Engineering, Kunming University of Science and Technology, Kunming 650093, Yunnan, China2Research Center for Analysis and Measurement, Kunming University of Science and Technology, Kunming 650093, Yunnan, China3Institute of Theoretical Chemistry, College of Chemistry, Jilin University, Changchun 130021, Jilin, Chinashow less
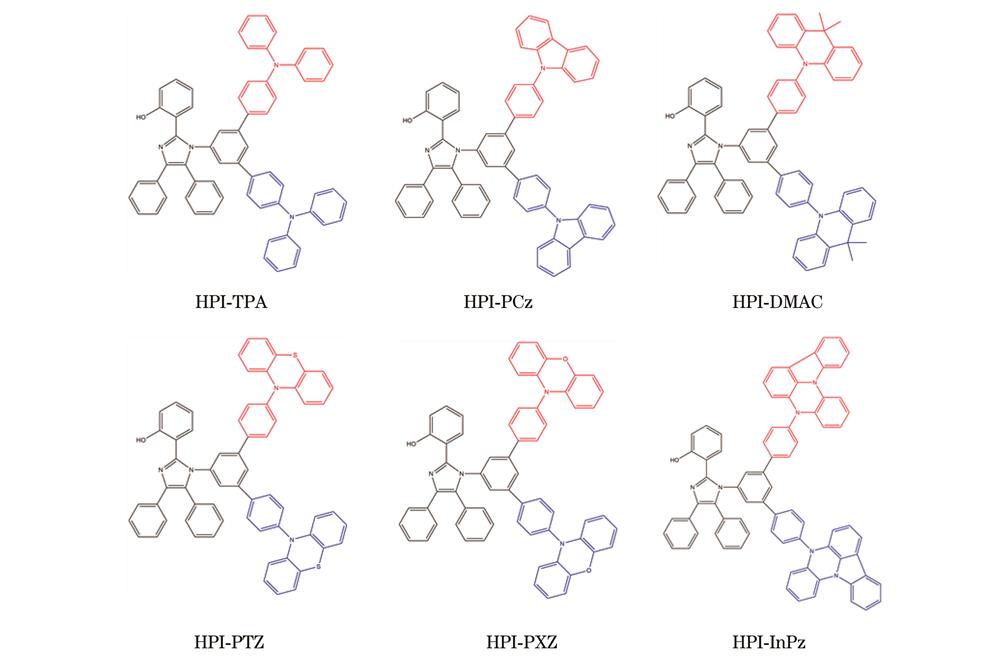
Fig. 1. Schematic diagram of designed molecular structures
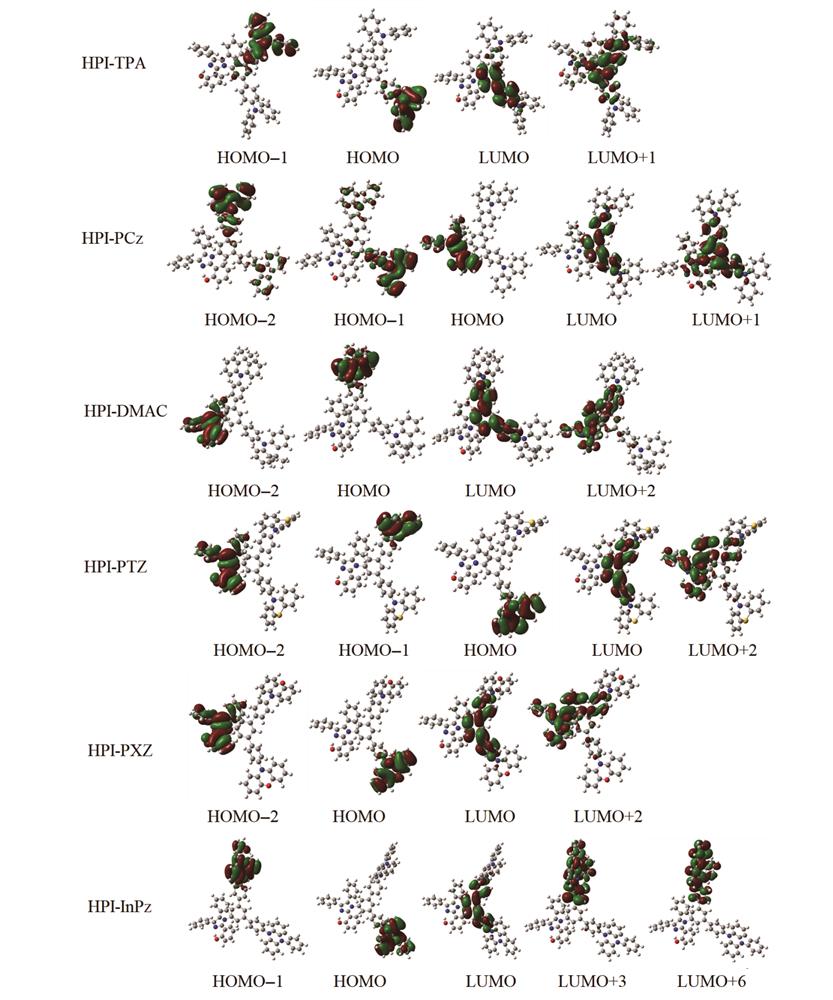
Fig. 2. Kohn-Sham frontier orbitals for HPI-TPA, HPI-PCz, HPI-DMAC, HPI-PTZ, HPI-PXZ and HPI-InPz predicted by PBE0/6-31G(d, p) method
Fig. 3. Absorption spectra simulated by Gaussian function
| Derivative | Absorption spectrum | Emission spectrum |
|---|
| Experiment[16] | B3LYP | PBE0 | CAM-B3LYP | Experiment[16] | B3LYP | PBE0 | CAM-B3LYP |
|---|
| HPI-TPA | 338 | 364 | 350 | 302 | 402 | 478 | 448 | 351 | | HPI-PCz | 339 | 381 | 358 | 285 | 490 | 540 | 498 | 358 |
|
Table 1. Absorption and emission spectra of HPI-TPA and HPI-PCz in experimental and theoretical calculation obtained by combining B3LYP, PBE0 and CAM-B3LYP with 6-31G(d, p) basis set
| Segment | Orbital | HPI-TPA | HPI-PCz | | HPI-DMAC | HPI-PTZ | HPI-PXZ | HPI-InPz |
|---|
| HPI | HOMO-2 | 97.0 | 2.5 | | 99.8 | 99.9 | 99.8 | 99.9 | | HOMO-1 | 7.0 | 3.6 | | 0 | 0 | 0 | 0 | | HOMO | 1.0 | 99.3 | | 0 | 0 | 0.1 | 0 | | LUMO | 49.4 | 53.4 | | 54.3 | 53.0 | 53.2 | 53.1 | | LUMO+1 | 56.2 | 58.0 | | 59.7 | 56.4 | 57.6 | 56.7 | | LUMO+2 | 92.5 | 86.7 | | 86.5 | 86.5 | 86.3 | 87.3 | | LUMO+3 | 18.4 | 0.1 | | 67.4 | 23.1 | 18.2 | 12.0 | | LUMO+6 | 0.4 | 20.4 | | 91.4 | 79.6 | 89.0 | 4.0 | | R-up | HOMO-2 | 3.0 | 85.3 | | 0.2 | 0.1 | 0.2 | 0 | | HOMO-1 | 92.8 | 11.5 | | 0 | 100.0 | 100.0 | 100.0 | | HOMO | 0.2 | 0.5 | | 100.0 | 0 | 0 | 0 | | LUMO | 7.9 | 23.0 | | 24.5 | 24.9 | 23.8 | 23.5 | | LUMO+1 | 33.3 | 18.6 | | 16.7 | 18.7 | 18.6 | 19.2 | | LUMO+2 | 6.7 | 10.6 | | 10.4 | 11.2 | 10.6 | 10.1 | | LUMO+3 | 75.5 | 99.9 | | 19.6 | 11.7 | 78.6 | 86.1 | | LUMO+6 | 99.6 | 47.5 | | 6.0 | 15.1 | 7.5 | 95.6 | | R-down | HOMO-2 | 0 | 12.2 | | 0 | 0 | 0 | 0 | | HOMO-1 | 0.2 | 84.9 | | 100.0 | 0 | 0 | 0 | | HOMO | 98.8 | 0.2 | | 0 | 100.0 | 99.9 | 100.0 | | LUMO | 42.7 | 23.6 | | 21.2 | 22.1 | 23.0 | 23.4 | | LUMO+1 | 10.5 | 23.4 | | 23.5 | 24.8 | 23.8 | 24.1 | | LUMO+2 | 0.8 | 2.7 | | 3.1 | 2.3 | 3.1 | 2.6 | | LUMO+3 | 6.1 | 0 | | 13.0 | 65.2 | 3.2 | 1.9 | | LUMO+6 | 0 | 32.1 | | 2.6 | 5.3 | 3.5 | 0.3 |
|
Table 2. Contribution of electron densities of different segments to orbitals in HPI-TPA, HPI-PCz, HPI-DMAC, HPI-PTZ, HPI-PXZ and HPI-InPz
| Compound | HPI-TPA | HPI-PCz | HPI-DMAC | HPI-PTZ | HPI-PXZ | HPI-InPz |
|---|
| EHOMO | -5.27 | -5.56 | -5.24 | -5.29 | -4.98 | -4.81 | | ELUMO | -1.12 | -1.46 | -1.46 | -1.56 | -1.56 | -1.57 | | ΔEH‑L | 4.15 | 4.10 | 3.78 | 3.73 | 3.42 | 3.24 |
|
Table 3. HOMO energy, LUMO energy and HOMO-LUMO energy gap of studied compounds
| Compound | IP,v | IP,a | PHE | EA,v | EA,a | PEE | λhole | λelectron |
|---|
| HPI-TPA | 6.51 | 6.01 | 5.98 | 0.34 | 0.50 | 0.77 | 0.53 | 0.43 | | HPI-PCz | 6.40 | 6.35 | 6.29 | 0.50 | 0.76 | 0.99 | 0.11 | 0.49 | | HPI-DMAC | 6.09 | 6.01 | 5.98 | 0.49 | 0.76 | 0.99 | 0.11 | 0.50 | | HPI-PTZ | 6.14 | 5.95 | 5.75 | 0.58 | 0.85 | 1.09 | 0.39 | 0.51 | | HPI-PXZ | 5.87 | 5.81 | 5.76 | 0.58 | 0.85 | 1.09 | 0.11 | 0.51 | | HPI-InPz | 5.60 | 5.55 | 5.49 | 0.61 | 0.87 | 1.11 | 0.11 | 0.50 |
|
Table 4. Ionization potentials, electron affinities, extraction potentials and reorganization energies for studied compounds
| Compound | Electronic transition | Wavelength /nm | f | Main configuration |
|---|
| HPI-TPA | S0→S1 | 350(338)[16] | 0.9385 | HOMO-1→LUMO(75.8%) | | HOMO→LUMO+1(15.3%) | | S0→S2 | 342 | 0.2341 | HOMO-2→LUMO(11.8%) | | HOMO→LUMO(77.5%) | | S0→S3 | 340 | 0.1554 | HOMO-2→LUMO(83.4%) | | HOMO→LUMO(10.0%) | | HPI-PCz | S0→S1 | 358 | 0.0043 | HOMO→LUMO(98.7%) | | S0→S2 | 340(339)[12] | 0.1363 | HOMO-2→LUMO(5.3%) | | HOMO-1→LUMO(13.5%) | | HOMO→LUMO+1(73.5%) | | S0→S3 | 338 | 0.4745 | HOMO-2→LUMO(47.6%) | | HOMO-1→LUMO(16.0%) | | HOMO-1→LUMO+1(12.2%) | | HOMO→LUMO+1(18.9%) | | HPI-DMAC | S0→S1 | 398 | 0.0007 | HOMO→LUMO(81.3%) | | HOMO→LUMO+1(11.5%) | | S0→S2 | 381 | 0.0007 | HOMO-1→LUMO(71.3%) | | HOMO-1→LUMO+1(23.4%) | | S0→S3 | 358 | 0.0046 | HOMO-2→LUMO(98.8%) | | S0→S10 | 309 | 0.2398 | HOMO-2→LUMO+2(93.7%) | | HPI-PTZ | S0→S1 | 397 | 0.0002 | HOMO→LUMO(72.9%) | | HOMO→LUMO+1(21.7%) | | S0→S2 | 393 | 0.0001 | HOMO-1→LUMO(80.3%) | | HOMO-1→LUMO+1(12.7%) | | S0→S3 | 363 | 0.0038 | HOMO-2→LUMO(98.9%) | | S0→S10 | 310 | 0.1613 | HOMO-2→LUMO+2(61.1%) | | HOMO-1→LUMO+2(28.6%) | | HPI-PXZ | S0→S1 | 444 | 0.0186 | HOMO→LUMO(74.1%) | | S0→S2 | 442 | 0.0012 | HOMO-1→LUMO(79.4%) | | HOMO-1→LUMO+1(13.8%) | | S0→S3 | 391 | 0.0014 | HOMO→LUMO(16.5%) | | HOMO→LUMO+1(81.7%) | | S0→S14 | 310 | 0.2415 | HOMO-2→LUMO+2(92.7%) | | HPI-InPz | S0→S1 | 466 | 0.0043 | HOMO→LUMO(77.5%) | | HOMO→LUMO+1(18.5%) | | S0→S2 | 462 | 0.0049 | HOMO-1→LUMO(81.9%) | | HOMO-1→LUMO+1(12.6%) | | S0→S3 | 411 | 0.0006 | HOMO→LUMO(20.2%) | | HOMO→LUMO+1(78.2%) | | S0→S9 | 357 | 0.1086 | HOMO-1→LUMO+3(11.9%) | | HOMO-1→LUMO+6(66.7%) |
|
Table 5. Absorption spectra, oscillator strengths and main transition configurations obtained by TD PBE0/6-31G(d, p) method
| Compound | Wavelength /nm | f | Main configuration |
|---|
| HPI-TPA | 448(402)[16] | 0.1348 | HOMO→LUMO(96.8%) | | HPI-PCz | 498(490)[16] | 0.0022 | HOMO→LUMO(99.5%) | | HPI-DMAC | 502 | 0 | HOMO→LUMO(96.9%) | | HPI-PTZ | 605 | 0 | HOMO→LUMO(97.3%) | | HPI-PXZ | 600 | 0 | HOMO→LUMO(97.0%) | | HPI-InPz | 643 | 0 | HOMO→LUMO(97.3%) |
|
Table 6. Emission wavelengths, oscillator strengths, main transition orbitals and transition coefficients of studied compounds